10-K: Annual report pursuant to Section 13 and 15(d)
Published on March 1, 2019
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-K
(Mark One)
ý |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2018.
OR
o |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to .
Commission file number 001-33528
OPKO Health, Inc.
(Exact Name of Registrant as Specified in Its Charter)
Delaware |
75-2402409 |
|
|
(State or Other Jurisdiction of
Incorporation or Organization)
|
(I.R.S. Employer
Identification No.)
|
|
4400 Biscayne Blvd., Miami, FL 33137 (Address of Principal Executive Offices) (Zip Code) |
(Registrant’s Telephone Number, Including Area Code): (305) 575-4100
|
Securities registered pursuant to section 12(b) of the Act: | ||
Title of Each Class |
Name of Each Exchange on Which Registered |
|
Common Stock, $.01 par value per share |
NASDAQ Global Select Market |
|
Securities registered pursuant to section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ý No ¨
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No ý
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ý No ¨
Indicate by check mark whether the Registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ý No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer |
ý |
Accelerated filer |
o |
Non-accelerated filer |
o |
Smaller reporting company |
o |
Emerging growth company |
o |
||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No ý
The aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, as of the last business day of the registrant’s most recently completed second fiscal quarter was: $1,518,040,596.
As of February 15, 2019, the registrant had 615,600,775 shares of Common Stock outstanding.
Documents Incorporated by Reference
Portions of the registrant’s definitive proxy statement for its 2018 Annual Meeting of Stockholders are incorporated by reference in Items 10, 11, 12, 13, and 14 of Part III of this Annual Report on Form 10-K.
TABLE OF CONTENTS
Page |
|||
Part I. |
|||
Item 1. |
|||
Item 1A. |
|||
Item 1B. |
|||
Item 2. |
|||
Item 3. |
|||
Item 4. |
|||
Part II. |
|||
Item 5. |
|||
Item 6. |
|||
Item 7. |
|||
Item 7A. |
|||
Item 8. |
|||
Item 9. |
|||
Item 9A. |
|||
Item 9B. |
|||
Part III. |
|||
Item 10. |
Directors, Executive Officers and Corporate Governance |
||
Item 11. |
Executive Compensation |
||
Item 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
||
Item 13. |
Certain Relationships and Related Transactions and Director Independence |
||
Item 14. |
Principal Accounting Fees and Services |
||
Part IV. |
|||
Item 15. |
|||
Signatures |
|||
Certifications |
|||
EX-21 |
|||
EX-23.1 |
|||
EX-31.1 |
|||
EX-31.2 |
|||
EX-32.1 |
|||
EX-32.2 |
|||
EX-101. INS XBRL Instance Document |
|||
EX-101.SCH XBRL Taxonomy Extension Schema Document |
|||
EX-101.CAL XBRL Taxonomy Extension Calculation Linkbase Document |
|||
EX-101.DEF XBRL Taxonomy Extension Definition Linkbase Document |
|||
EX-101.LAB XBRL Taxonomy Extension Label Linkbase Document |
|||
EX-101.PRE XBRL Taxonomy Extension Presentation Linkbase Document |
|||
3
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains “forward-looking statements,” as that term is defined under the Private Securities Litigation Reform Act of 1995 (“PSLRA”), Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Forward-looking statements include statements about our expectations, beliefs or intentions regarding our product development efforts, business, financial condition, results of operations, strategies or prospects. You can identify forward-looking statements by the fact that these statements do not relate strictly to historical or current matters. Rather, forward-looking statements relate to anticipated or expected events, activities, trends or results as of the date they are made. Because forward-looking statements relate to matters that have not yet occurred, these statements are inherently subject to risks and uncertainties that could cause our actual results to differ materially from any future results expressed or implied by the forward-looking statements. Many factors could cause our actual activities or results to differ materially from the activities and results anticipated in forward-looking statements. These factors include those described below and in “Item 1A-Risk Factors” of this Annual Report on Form 10-K. We do not undertake an obligation to update forward-looking statements. We intend that all forward-looking statements be subject to the safe-harbor provisions of the PSLRA. These forward-looking statements are only predictions and reflect our views as of the date they are made with respect to future events and financial performance.
Risks and uncertainties, the occurrence of which could adversely affect our business, include the following:
• |
we have a history of losses and may not generate sustained positive cash flow sufficient to fund our operations and research and development programs; |
• |
our need for, and ability to obtain, additional financing when needed on favorable terms, or at all; |
• |
adverse results in material litigation matters or governmental inquiries, including, without limitation, recent lawsuits against the Company and its Chairman and Chief Executive Officer by the SEC, as well as related class action and derivative lawsuits; |
• |
the risks inherent in developing, obtaining regulatory approvals for and commercializing new, commercially viable and competitive products and treatments; |
• |
our research and development activities may not result in commercially viable products; |
• |
that earlier clinical results of effectiveness and safety may not be reproducible or indicative of future results; |
• |
the success of our relationship with Pfizer; |
• |
that we may fail to obtain regulatory approval for hGH-CTP or successfully commercialize Rayaldee and hGH-CTP;
|
• |
that we may not generate profits or cash flow from our laboratory operations or substantial revenue from Rayaldee and our pharmaceutical and diagnostic products;
|
• |
that currently available over-the-counter and prescription products, as well as products under development by others, may prove to be as or more effective than our products for the indications being studied; |
• |
our ability to build a successful pharmaceutical sales and marketing infrastructure; |
• |
our ability and our distribution and marketing partners’ ability to comply with regulatory requirements regarding the sales, marketing and manufacturing of our products and product candidates and the operation of our laboratories; |
• |
the performance of our third-party distribution partners, licensees and manufacturers over which we have limited control; |
• |
our success is dependent on the involvement and continued efforts of our Chairman and Chief Executive Officer; |
• |
integration challenges for Transition Therapeutics, BioReference, EirGen and other acquired businesses; |
• |
availability of insurance coverage with respect to material litigation matters; |
• |
changes in regulation and policies in the United States (“U.S.”) and other countries, including increasing downward pressure on healthcare reimbursement; |
• |
our ability to manage our growth and our expanded operations; |
• |
increased competition, including price competition; |
4
• |
changing relationships with payors, including the various state and multi-state Blues programs, suppliers and strategic partners; |
• |
efforts by third-party payors to reduce utilization and reimbursement for clinical testing services; |
• |
our ability to maintain reimbursement coverage for our products and services, including the 4Kscore test;
|
• |
failure to timely or accurately bill and collect for our services; |
• |
failure in our information technology systems, including cybersecurity attacks or other data security or privacy incidents; |
• |
failure to obtain and retain new clients and business partners, or a reduction in tests ordered or specimens submitted by existing clients; |
• |
failure to establish, and perform to, appropriate quality standards to assure that the highest level of quality is observed in the performance of our testing services; |
• |
failure to maintain the security of patient-related information; |
• |
our ability to obtain and maintain intellectual property protection for our products; |
• |
our ability to defend our intellectual property rights with respect to our products; |
• |
our ability to operate our business without infringing the intellectual property rights of others; |
• |
our ability to attract and retain key scientific and management personnel; |
• |
failure to obtain and maintain regulatory approval outside the U.S.; |
• |
legal, economic, political, regulatory, currency exchange, and other risks associated with international operations; and |
• |
our ability to finance and successfully complete construction of a research, development and manufacturing center in Waterford, Ireland. |
5
PART I
Unless the context otherwise requires, all references in this Annual Report on Form 10-K to the “Company”, “OPKO”, “we”, “our”, “ours”, and “us” refer to OPKO Health, Inc., a Delaware corporation, including our wholly-owned subsidiaries.
ITEM 1. |
BUSINESS |
OVERVIEW
We are a diversified healthcare company that seeks to establish industry-leading positions in large and rapidly growing medical markets. Our diagnostics business includes BioReference Laboratories (“BioReference”), the nation’s third-largest clinical laboratory with a core genetic testing business and an almost 300-person sales and marketing team to drive growth and leverage new products, including the 4Kscore prostate cancer diagnostic test and the Claros 1 in-office immunoassay platform. Our pharmaceutical business features Rayaldee, an FDA-approved treatment for secondary hyperparathyroidism (“SHPT”) in adults with stage 3 or 4 chronic kidney disease (“CKD”) and vitamin D insufficiency (launched in November 2016), OPK88004, a selective androgen receptor modulator which we have studied for benign prostatic hyperplasia but for which we are exploring other indications, and OPK88003, a once or twice weekly oxyntomodulin for type 2 diabetes and obesity which is a clinically advanced drug candidate among the new class of GLP-1 glucagon receptor dual agonists (phase 2b). Our pharmaceutical business also features hGH-CTP, a once-weekly human growth hormone injection (in phase 3 and partnered with Pfizer). We operate established pharmaceutical business operations in Spain, Ireland, Chile, and Mexico, which are generating revenue and from which we expect to generate positive cash flow and facilitate future market entry for our products currently in development. We have a development and commercial supply pharmaceutical company as well as a global supply chain operation and holding company in Ireland, which we expect will play an important role in the development, manufacturing, distribution and approval of a wide variety of drugs with an emphasis on high potency products. We also own a specialty active pharmaceutical ingredients (“APIs”) manufacturer in Israel, which we expect will facilitate the development of our pipeline of molecules and compounds for our proprietary molecular diagnostic and therapeutic products.
We have a highly experienced management team. Based on their experience in the industry, we believe that our management team has extensive development, regulatory and commercialization expertise and relationships that provide access to commercial opportunities.
All product or service marks appearing in type form different from that of the surrounding text are trademarks or service marks owned, licensed to, promoted or distributed by OPKO, its subsidiaries or affiliates, except as noted. All other trademarks or services marks are those of their respective owners.
GROWTH STRATEGY
We expect our future growth to come from leveraging our commercial infrastructure, proprietary technology and development strengths, and by opportunistically pursuing complementary, accretive, or strategic acquisitions and investments.
We launched our first pharmaceutical product, Rayaldee, in the U.S. market in the fourth quarter of 2016. We have under development a broad and diversified portfolio of diagnostic tests, small molecules, and biologics targeting a broad range of unmet medical needs. We also operate the third largest full service clinical laboratory in the U.S. We intend to continue to leverage our proprietary technology and our strengths in all phases of research and development to further develop and commercialize our portfolio of proprietary pharmaceutical and diagnostic products. In support of our strategy, we intend to:
• |
continue to enhance our commercialization capability in the U.S. and internationally; |
• |
develop and commercialize Rayaldee for new indications, including the treatment of SHPT in patients with vitamin D insufficiency and stage 5 CKD requiring regular hemodialysis;
|
• |
obtain requisite regulatory approval and compile clinical data for our most advanced product candidates; and |
• |
expand into other medical markets that provide significant opportunities and that we believe are complementary to and synergistic with our business. |
In addition, we expect to leverage the BioReference business and infrastructure to drive rapid and widespread uptake of our diagnostic products, including the 4Kscore test. We also intend to leverage the genetic and genomic data generated and accumulated through BioReference’s genetic sequencing laboratory to enhance drug discovery and clinical trial programs.
We have and expect to continue to be opportunistic and to pursue complementary or strategic acquisitions, licenses and investments. Our management team has significant experience in identifying, executing and integrating these transactions. We expect to use well-timed, carefully selected acquisitions, licenses and investments to continue to drive our growth, including:
6
• |
Products and technologies. We intend to continue to pursue product and technology acquisitions and licenses that will complement our existing businesses and provide new product and market opportunities, enhance our profitability, leverage our existing assets, and contribute to our own organic growth.
|
• |
Commercial businesses. We intend to continue to pursue acquisitions of commercial businesses that will both drive our growth and provide geographically diverse sales and distribution opportunities.
|
• |
Early stage investments. We have and may continue to make investments in early stage companies that we perceive to have valuable proprietary technology and significant potential to create value for OPKO as a shareholder.
|
CORPORATE INFORMATION
We were originally incorporated in Delaware in October 1991 under the name Cytoclonal Pharmaceutics, Inc., which was later changed to eXegenics, Inc. (“eXegenics”). On March 27, 2007, we were part of a three-way merger with Froptix Corporation (“Froptix”) and Acuity Pharmaceuticals, Inc. (“Acuity”), both research and development companies. On June 8, 2007, we changed our name to OPKO Health, Inc. Our shares are publicly traded on the NASDAQ Stock Market under the ticker “OPK” and on the Tel Aviv Stock Exchange. Our principal executive offices are located in leased office space in Miami, Florida.
We currently manage our operations in two reportable segments: diagnostics and pharmaceuticals. The pharmaceutical segment consists of the pharmaceutical operations we operate in Chile, Mexico, Ireland, Israel and Spain and our pharmaceutical research and development operations. The diagnostics segment primarily consists of the clinical laboratory operations we acquired through the acquisition of BioReference in 2015 and our point-of-care operations. There are no significant inter-segment sales. We evaluate the performance of each segment based on operating profit or loss. There is no inter-segment allocation of interest expense and income taxes. Refer to Note 17 for financial information about the segments and geographic areas.
CURRENT PRODUCTS AND SERVICES AND RELATED MARKETS
Diagnostics
BioReference Laboratories
Through BioReference, the third largest full service clinical laboratory in the U. S., we offer comprehensive laboratory testing services utilized by healthcare providers in the detection, diagnosis, evaluation, monitoring, and treatment of diseases, including esoteric testing, molecular diagnostics, anatomical pathology, genetics, women’s health and correctional healthcare. We market and sell these services to physician offices, clinics, hospitals, employers and governmental units nationally, with the largest concentration of business in the larger metropolitan areas across New York, New Jersey, Florida, Texas, Maryland, California, Pennsylvania, Delaware, Washington DC, Illinois and Massachusetts.
BioReference has an almost 300-person sales and marketing team and operates a network of approximately 200 patient service centers.
Our BioReference laboratory testing business consists of routine testing and esoteric testing. Routine tests measure various health parameters, such as the functions of the heart, kidney, liver, thyroid and other organs, including such tests as blood cell counts, cholesterol levels, pregnancy, substance abuse and urinalysis. We typically operate 24 hours per day, 365 days per year and perform and report most routine test results within 24 hours.
The esoteric tests we perform require sophisticated equipment and materials, highly skilled personnel and professional attention. Esoteric tests are ordered less frequently than routine tests and typically are priced higher than routine tests. Esoteric tests include tests related to endocrinology, genetics and genomics, immunology, microbiology, HIV tests, molecular diagnostics, next generation sequencing, oncology, serology, and toxicology.
Through BioReference, we operate in the following highly specialized laboratory divisions:
• |
BioReference Laboratories. BioReference constitutes our core clinical testing laboratory offering automated, high volume routine testing services, STAT testing, informatics, HIV, Hep C and other molecular tests.
|
• |
GenPath (Oncology). National oncology presence with expertise in cancer pathology and diagnostics, as well as molecular diagnostics. Core tests include FLOW, IHC, MicroArray, FISH, ISH, Morphology, and full service oncology.
|
7
• |
GenPath (Women’s Health). Innovative technology platform for sexually transmitted infections has enabled expansion nationally with specimens coming from 41 states, including Image Directed Paps analysis, HPV Plus, and STI Testing.
|
• |
GeneDx. Industry leading national laboratory for testing rare and ultra-rare genetic diseases with international reach, performing testing on specimens from more than 50 countries.
|
• |
Laboratorio Buena Salud. National testing laboratory dedicated to serving the Spanish-speaking population in the U. S., where all business is conducted in Spanish including patient and physician interaction.
|
We have one of the largest marketing staffs of any laboratory in the country with sales and marketing groups dedicated to urology, oncology, women’s health, genetic testing and correctional health, as well as cross-over groups selling to large institutions. All of our sales and marketing personnel operate in a dual capacity, as both marketing and client support representatives, which we believe provides better customer service and a strong connection with our customers.
We expect the clinical laboratory testing industry will continue to experience growth in testing volumes due to aging of the population in the U.S., patient awareness of the value of laboratory tests, a decrease in the cost of tests, the development of sophisticated and specialized tests for detection and management of disease, increased recognition of early detection and prevention as a means of reducing healthcare costs, and ongoing research and development in genetics and genomics and personalized medicine. Our mission is to be recognized by our clients as the premier provider of clinical laboratory testing, information and related services.
BioReference provides us with a significant diagnostics commercial infrastructure for marketing and sales that reached almost 11 million patients in 2018. In addition, its large team of managed care experts complement our efforts to ensure that payors recognize the value of our diagnostic and laboratory tests for reimbursement purposes. We continue to leverage the national marketing, sales and distribution resources of BioReference, along with its almost 300-person sales and marketing team, to enhance sales of and reimbursement for our 4Kscore test, a laboratory developed blood test that provides a personalized risk score for aggressive prostate cancer. We plan to leverage the BioReference commercial infrastructure and capabilities, as well as its extensive relationships with payors, to commercialize OPKO’s other diagnostic products under development.
4Kscore Test
We offer the 4Kscore test through our BioReference laboratory located in Elmwood Park, New Jersey. We began selling the 4Kscore test in the U.S. in March 2014 and in Europe and Mexico in September 2014 and January 2015, respectively. The 4Kscore test is a laboratory developed test that measures the blood plasma levels of four different prostate-derived kallikrein proteins: Total PSA, Free PSA, Intact PSA and Human Kallikrein-2 (“hK2”). These biomarkers are then combined with a patient’s age, Digital Rectal Exam (“DRE”) status (nodule / no nodule), and prior negative biopsy status (yes / no) using a proprietary algorithm to calculate the risk (probability) of finding a Gleason Score 7 or higher prostate cancer. The four kallikrein panel of biomarkers utilized in the 4Kscore test is based on decades of research conducted by scientists at Memorial Sloan-Kettering Cancer Center and leading European institutions. Investigators at the Lund University, Sweden, University of Turku, Finland and Memorial Sloan Kettering Cancer Center, New York, have also demonstrated that the 4Kscore test can risk stratify the 20-year risk for development of prostate metastases and mortality in men who present at age 50 or 60 years old with an elevated PSA.
The 4Kscore test was developed by OPKO and validated in two prospective, blinded studies of 1,012 and 366 men, respectively. The first study was done in collaboration with 26 urology centers across the U.S. and the second study was conducted at eight VA centers in the U.S. with a predominantly African American cohort. African Americans are 1.7 times more likely to be diagnosed with prostate cancer than Caucasian men and 2.2 times more likely to die from the disease. Results showed that the 4Kscore test was highly accurate for predicting the presence of high-grade cancer (Gleason score 7 or higher) prior to prostate biopsy, regardless of race. The full data from the blinded, prospective U.S. clinical validation studies have been published in peer reviewed medical journals.
The clinical data from both studies demonstrated the ability of the 4Kscore test to discriminate between men with high-grade, aggressive prostate cancer and those men who had no findings of cancer or had low-grade or indolent form of the disease. The discrimination, measured by Area Under the Curve (“AUC”) analysis, was greater than 0.80 and is significantly higher than previously developed tests. Furthermore, the 4Kscore test demonstrated excellent risk calibration, indicating the accuracy of the result for an individual patient, both Caucasian and African American. The high value of AUC and the excellent risk calibration make the 4Kscore test result valuable information for the shared decision-making between the urologist and patient on whether or not to perform a prostate biopsy.
8
A separate clinical utility study indicated that the 4Kscore test led to 64.6% fewer biopsies. The study, “The 4Kscore® Test Reduces Prostate Biopsy Rates in Community and Academic Urology Practices”, was published in a peer reviewed medical journal. The study, which included 611 patients seen by 35 academic and community urologists across the U.S., evaluated the influence of the 4Kscore test on urologist- patient decisions about whether to perform a biopsy in men who had an abnormal PSA and or DRE result. Test results for patients were stratified into low risk (< 7.5%), intermediate risk (7.5%-19.9%) and high risk (³20%) for developing aggressive prostate cancer. Nearly half (49.3%) of the men were categorized as low risk; 25.7% and 25.0% fell into the intermediate-risk and high-risk categories, respectively. Notably, the 4Kscore test results influenced biopsy decisions in 88.7% of the men. In the three risk groups, a biopsy was avoided in 94.0%, 52.9% and 19.0% of men in the low, intermediate and high-risk categories, respectively.
The National Comprehensive Cancer Network (“NCCN”) included the 4Kscore test as a recommended test in their 2015, 2016, 2017 and 2018 Guidelines for Prostate Cancer Early Detection. The panel making this recommendation concluded that the 4Kscore test is indicated for use prior to a first prostate biopsy, or after a negative biopsy, to assist patients and physicians in further defining the probability of high-grade cancer. In addition, the European Association of Urology (“EAU”) Prostate Cancer Guidelines Panel included the 4Kscore test in the 2018 EAU Guidelines for Prostate Cancer, concluding that the 4Kscore, as a blood test with greater specificity over the PSA test, is indicated for use prior to a first prostate biopsy or after a negative biopsy to assist patients and physicians in further defining the probability of high-grade cancer.
We have and will continue to commit substantial efforts to obtaining broad reimbursement coverage for the 4Kscore test. The 4Kscore test has been granted a Category I CPT® code by the AMA (CPT Code 81539). A CPT code is used by insurance companies and government payors to describe health care services and procedures. A Category I CPT code is critical to facilitate reimbursement in government programs such as Medicare and Medicaid, as well as private insurance programs. We have obtained a positive coverage decision from at least one national private payor and pricing agreements from several regional payors.
Novitas Solutions, the local Medicare Administrative Contractor (“MAC”) for our laboratory in New Jersey, issued a proposed non-coverage policy for the 4Kscore test in May 2018 subject to a public comment period ending July 5, 2018. We made oral presentations at a Novitas open meeting and submitted substantial evidence and data to address the comments raised in the draft non-coverage determination. In January 2019, Novitas issued a notice of a future non-coverage determination for the 4KScore test to be effective March 20, 2019. We are evaluating options to appeal the decision and undertake other steps with the Center for Medicare and Medicaid Services (“CMS”) in an effort to have this determination rescinded or reversed. We are also developing a strategy to obtain FDA approval for the 4Kscore test, among other efforts, to assist in securing broad reimbursement coverage.
Point-of-Care Diagnostics
OPKO Diagnostics, LLC (“OPKO Diagnostics”), formerly Claros Diagnostics, Inc., has developed a novel diagnostic instrument system to provide rapid, high performance blood test results in the point-of-care setting. The technology only requires a finger stick drop of blood introduced into the test cassette that can then run a quantitative test. The instrument performs the tests on a disposable, one time usable cassette that is a microfluidics-based diagnostic test system. The credit card-sized test cassette works with a sophisticated desktop analyzer to provide high performance quantitative blood test results within minutes and permits the transition of complex immunoassays from the centralized reference laboratory to the physician’s office, hospital nurses station, or other decentralized location.
We completed multiple in vitro analytical validation and field use tests for the PSA test in mid-2017 and filed the pre-marketing authorization (“PMA”) for the Claros Analyzer and Sangia Total PSA Test with the FDA in November 2017. The key clinical study with patients who were suspicious for prostate cancer found that the Sangia Total PSA test improved the sensitivity of a digital rectal exam (“DRE”) to 91%, detecting 2.9 times the prostate cancers compared to DRE alone.
The FDA approved the PMA for the Sangia Total PSA Test using the Claros Analyzer in January 2019. We also intend to commence a clinical trial of a testosterone diagnostic test for our point-of-care system. We are evaluating commercialization strategies for the Claros system, including expansion of the test menu prior to commercialization.
We are presently working to add additional tests for our point-of-care system, and we believe that there are many more applications for the technology, including infectious disease, cardiology, women’s health and companion diagnostics.
Pharmaceutical Business
We currently have one commercial stage pharmaceutical product and several pharmaceutical compounds and technologies in various stages of research and development for a broad range of indications and conditions, including the following:
9
Renal Products
We launched Rayaldee, our lead renal product, in the U.S. market in November 2016. In June 2016, the FDA approved Rayaldee extended release capsules for the treatment of secondary hyperparathyroidism (“SHPT”) in adults with stage 3 or 4 chronic kidney disease (“CKD”) and vitamin D insufficiency, defined as serum total 25-hydroxyvitamin D levels less than 30 ng/mL. Rayaldee is a patented extended release product containing 30 mcg of a prohormone called calcifediol (25-hydroxyvitamin D3).
We have a 79-person highly specialized sales, marketing and market access team dedicated to the launch and commercialization of Rayaldee as of December 31, 2018. As compared to the fourth quarter of 2017 and the third quarter of 2018, total Rayaldee prescriptions increased approximately 141% and 17%, respectively, in the fourth quarter of 2018. Efforts are underway to obtain broader commercial and Part D insurance coverage for Rayaldee. We have already contracted for commercial and Part D coverage for more than seventy percent (70%) of U.S. covered lives as of the end of 2018.
In connection with the launch of Rayaldee, we have also engaged in a comprehensive ongoing market education campaign highlighting the unmet need in treating SHPT, including by leveraging key opinion leaders in community outreach programs such as speakers’ bureaus and patient advocacy programs.
In May 2016, we entered into a collaboration with Vifor Fresenius Medical Care Renal Pharma (“VFMCRP”) for the development and commercialization of Rayaldee in Europe, Canada, Mexico, Australia, South Korea and certain other international markets for the treatment of SHPT in patients with stage 3, 4 or 5 CKD and vitamin D insufficiency. Under the terms of the agreement, OPKO received an upfront payment of $50 million. We also received a $2 million payment triggered by the marketing approval of Rayaldee in Canada and will receive up to $230 million in additional regulatory and sales-based milestones. In addition, VFMCRP will pay OPKO tiered, double digit royalties on sales of the product at percentage rates that range from the mid-teens to the mid-twenties or a minimum royalty, whichever is greater, upon commencement of sales of the product. OPKO and VFMCRP are also collaborating to develop and commercialize a new dosage form of Rayaldee for the treatment of SHPT in hemodialysis patients. OPKO granted VFMCRP an option to acquire rights to this dosage form for the U.S. market; if exercised, OPKO will receive up to $555 million in additional milestones and double digit royalties.
On October 12, 2017, we entered into a Development and License Agreement (the “JT Agreement”) with Japan Tobacco Inc. (“JT”) granting JT the exclusive rights for the development and commercialization of Rayaldee in Japan (the “JT Territory”). The license grant to JT covers the therapeutic and preventative use of the product for (i) SHPT in non-dialysis and dialysis patients with CKD, (ii) rickets and (iii) osteomalacia, as well as such additional indications as may be added to the scope of the license subject to the terms of the JT Agreement. Under the terms of the JT Agreement, OPKO received an initial upfront payment of $6 million and received another $6 million milestone payment triggered by the initiation of OPKO’s U.S. phase 2 study with Rayaldee in dialysis patients. OPKO is also eligible to receive up to an additional aggregate amount of $31 million upon the achievement of certain regulatory and development milestones by JT for Rayaldee in the JT Territory, and $75 million upon the achievement of certain sales based milestones by JT in the JT Territory. OPKO will also receive tiered, double digit royalty payments at rates ranging from low double digits to mid-teens on net product sales within the JT Territory. JT will, at its sole cost and expense, be responsible for performing all development activities necessary to obtain all regulatory approvals for Rayaldee in Japan and for all commercial activities pertaining to Rayaldee in Japan, except for certain preclinical expenses which OPKO has agreed to reimburse JT up to a capped amount.
The FDA approval of Rayaldee was supported by successful results from two identical randomized, double-blind, placebo-controlled, multi-site phase 3 studies which established the safety and efficacy of Rayaldee as a new treatment for SHPT in adults with stage 3 or 4 CKD and vitamin D insufficiency.
Vitamin D insufficiency arises in CKD due to the abnormal upregulation of CYP24A1, an enzyme that destroys vitamin D and its metabolites, and from many other causes as well.
Studies in CKD patients have demonstrated that currently available over-the-counter and prescription vitamin D supplements cannot reliably raise blood vitamin D prohormone levels and effectively treat SHPT, a condition commonly associated with CKD in which the parathyroid glands secrete excessive amounts of PTH. Prolonged elevation of blood PTH causes excessive calcium and phosphorus to be released from bone, leading to elevated serum calcium and phosphorus levels, softening of the bones (osteomalacia) and calcification of vascular and renal tissues. SHPT affects 40-82% of patients with stage 3 or 4 CKD and approximately 95% of patients with stage 5 CKD.
The completed phase 3 trials for Rayaldee successfully met all primary efficacy and safety endpoints. The primary efficacy endpoint was a responder analysis in which “responder” was defined as any treated subject who demonstrated an average decrease in PTH of at least 30% from pre-treatment baseline during the last six weeks of the 26-week treatment period. A significantly higher response rate was observed with Rayaldee compared to placebo treatment in both trials and safety and
10
tolerability data were comparable in both treatment groups. The PTH-lowering response rates with Rayaldee were similar in both stage 3 and 4 CKD. Patients completing the two pivotal trials were treated, at their election, for an additional six months with Rayaldee during an open-label extension study. Data from the extension study indicated that the PTH lowering response rate steadily increased with duration of Rayaldee treatment without deterioration in safety profile.
We also are developing Rayaldee for other indications, including for SHPT in patients with vitamin D insufficiency and stage 5 CKD requiring regular hemodialysis. A phase 2 study of a higher dose product commenced in this patient population during the third quarter of 2018. We expect to receive data from the study in the second half of 2020.
In August 2014, we also announced the submission of an Investigational New Drug Application (“IND”) to the FDA to evaluate Rayaldee as an adjunctive therapy for the prevention of skeletal-related events in patients with bone metastases undergoing anti-resorptive therapy. We commenced a phase 1 dose escalation study in the fourth quarter of 2014 in breast and prostate cancer patients with bone metastases who were receiving anti-resorptive therapy. The study, which has been completed, was designed to evaluate safety, markers of vitamin D and mineral metabolism and tumor progression. We are currently collecting the final data and will shortly complete a final analysis of the study.
We filed an IND for Rayaldee in January 2019 for the treatment of SHPT arising from vitamin D insufficiency in patients who have undergone bariatric surgery. We intend to commence a phase 2 study in this population in the first half of 2019.
Another renal product in our development pipeline, Alpharen (Fermagate Tablets), is a new and potent non-absorbed phosphate binder to treat hyperphosphatemia in stage 5 CKD patients requiring regular hemodialysis. Alpharen (Fermagate Tablets) has been shown to be safe and effective in treating hyperphosphatemia in phase 2 and 3 trials in stage 5 CKD patients undergoing chronic hemodialysis. Hyperphosphatemia, or elevated serum phosphorus, is common in dialysis patients and tightly linked to the progression of SHPT and vascular calcification, both of which drive morbidity and mortality. The kidneys provide the primary route of excretion for excess phosphorus absorbed from ingested food. As kidney function worsens, serum phosphorus levels increase and directly stimulate PTH secretion. Stage 5 CKD patients requiring dialysis must reduce their dietary phosphate intake and usually require regular treatment with orally administered phosphate binding agents to lower serum phosphorus to meet the recommendations of the Kidney Disease Improving Global Outcomes (“KDIGO”) Clinical Practice Guidelines that elevated serum phosphorus levels should be lowered. Hyperphosphatemia contributes to soft tissue mineralization and affects approximately 90% of dialysis patients. Dialysis patients require ongoing phosphate binder treatment to maintain controlled serum phosphorus levels. An additional phase 3 clinical trial is required to support marketing approvals for Alpharen in North America and in Europe, and the Company is evaluating development opportunities.
We believe the CKD patient population is large and growing as a result of obesity, hypertension and diabetes; therefore this patient population represents a significant global market opportunity. According to the National Kidney Foundation, CKD afflicts over 40 million people in the U.S., including more than 21 million patients with stage 3 or 4 CKD. In stage 5 CKD, kidney function is minimal to absent and most patients require regular dialysis or a kidney transplant for survival. An estimated 71-97% of CKD patients have vitamin D insufficiency which can lead to SHPT and its debilitating consequences. CKD continues to be associated with poor outcomes, reflecting the inadequacies of the current standard of care.
Vitamin D insufficiency, hyperphosphatemia and SHPT, when inadequately treated, are major contributors to poor CKD outcomes. We intend to develop and commercialize Rayaldee and other renal products to constitute part of the foundation for a new and markedly improved standard of care for CKD patients having SHPT and other renal products.
SARM
Through the acquisition of Transition Therapeutics, a Toronto-based biotechnology company, we acquired OPK88004, an orally administered selective androgen receptor modulator (SARM) which we have been developing for the treatment of Benign Prostatic Hypertrophy (BPH) and other urologic and metabolic conditions. The selective and antagonistic properties of OPK88004 on the prostate appear to be well suited to potentially reduce prostate hyperplasia and volume, as well as provide anabolic therapeutic benefits such as increased lean body mass and physical function, and decreased fat mass in specific patient populations. We believe that SARMs hold considerable promise as a new class of anabolic therapies for a variety of clinical indications, such as frailty and functional limitations associated with aging and chronic illnesses, cancer and osteoporosis.
A phase 2 study of 350 male subjects for another indication showed significantly increased lean body mass and muscle strength and significant fat mass reduction with no change in lower PSA levels. OPK88004 is currently being studied in a phase 2 study in prostate cancer patients who have undergone radical prostatectomy. The main objective of the study is to examine the effect of OPK88004 on sexual function and quality of life issues associated with this patient population.
An additional phase 2b study to determine the optimal dose to treat patients with BPH commenced in November 2017 and we completed enrollment and randomized 114 patients in the U.S. in December 2018. The main focus of the study is to
11
determine the optimal dose of OPK88004 that will reduce prostate volume and PSA levels, and increase anabolic effects such as lean body and decreased fat mass in BPH patients. As previously reported, blinded data from the phase 2b study have shown significant variability in the measurement of prostate volume, rendering the assessment of prostate volume from treatment impractical. Additionally, a small number of subjects have shown increased liver enzymes. We have suspended the current trial but continue to analyze data relating to the study’s other primary endpoint, the effect of OPK88004 on serum PSA levels, and the secondary endpoints, changes in lean body mass and fat mass. The results of this data analysis are expected in the second quarter of 2019. Additional indications including treatment of symptoms associated with androgen deprivation therapy in prostate cancer patients and low testosterone levels, muscle weakness and general frailty in kidney dialysis patients are being planned.
Oxyntomodulin
Our internal product development program is also currently focused on developing a once weekly administered oxyntomodulin for type 2 diabetes and obesity. Our most advanced oxyntomodulin product candidate, OPK88003, a once-weekly administered peptide for the treatment of type 2 diabetes and associated obesity, is a dual agonist of the Glucagon-Like Peptide-1 (GLP-1) and glucagon receptors. The receptors play an integral role in regulating appetite, food intake, satiety and energy utilization in the body. Stimulating both of the receptors, OPK88003 has the potential to regulate blood glucose.
OPK88003 has been evaluated in a phase 2 study enrolling 420 type 2 diabetes subjects in a 24 week study consisting of a 12-week randomized blinded stage followed by a 12-week open-label stage. The study included four once-weekly dose arms of OPK88003 (10mg, 15mg, 30mg, 50mg), a placebo arm, and an active comparator arm (exenatide extended release – 2mg). The study was completed in February, 2016.
Subjects receiving the highest dose of OPK88003 peptide once weekly in the study demonstrated significantly superior weight loss compared with currently approved extended release exenatide and placebo after 12 and 24 weeks of treatment. OPK88003 also provided a reduction in HbA1c, a marker of sugar metabolism, similar to exenatide at weeks 12 and 24.
We have evaluated OPK88003 in a dose escalation phase 2b trial in 110 type 2 diabetics where patients have been treated with a dose escalation regimen over 3 months intended to optimize dose levels, and increase body weight loss and reduce the adverse event profile, such as nausea and vomiting. The patients were treated for a total of 30 weeks in the study. We have completed the study and expect to have topline data from the study in March 2019. The key primary endpoint will be HbA1c and secondary endpoints such as weight loss, lipid profile and safety will also be analyzed.
We believe oxyntomodulin has potential to be a safe, long term therapy for obesity and diabetes type II patients, representing significant market opportunities. More than 380 million are living with diabetes worldwide, of which approximately 90% have type II diabetes. According to the World Health Organization, there are more than 500 million severely overweight or obese people.
Biologics
Our biologics business focuses on developing and commercializing longer-acting proprietary versions of already approved therapeutic proteins. One of our innovative platform technologies uses a short, naturally-occurring amino acid sequence, carboxl terminal peptide (“CTP”) which has the effect of slowing the removal from the body of the therapeutic protein to which it is attached. This CTP can be readily attached to a wide array of existing therapeutic proteins, stabilizing the therapeutic protein in the bloodstream and extending its life span without additional toxicity or loss of desired biological activity. We are using the CTP technology to develop new, proprietary versions of certain existing therapeutic proteins that have longer life spans than therapeutic proteins without CTP. We believe that our products will have greatly improved therapeutic profiles and distinct market advantages.
hGH-CTP
Our lead product candidate utilizing CTP, hGH-CTP, is a recombinant human growth hormone product under development for the treatment of growth hormone deficiency (“GHD”), which is a pituitary disorder resulting in short stature in children and other physical ailments in both children and adults.
In December 2014, we entered into an exclusive worldwide agreement with Pfizer for the development and commercialization of hGH-CTP for the treatment of GHD in adults (“Adult GHD”) and in children (“Pediatric GHD”), as well as for the treatment of growth failure in children born small for gestational age (“SGA”). In connection with the transaction, we granted Pfizer an exclusive license to commercialize hGH-CTP worldwide, and we received non-refundable and non-creditable upfront payments of $295 million and are eligible to receive up to an additional $275 million upon the achievement of certain regulatory milestones. In addition, we are eligible to receive initial tiered royalty payments associated with the commercialization of hGH-CTP for Adult GHD with percentage rates ranging from the high teens to mid-twenties. Upon the
12
launch of hGH-CTP for Pediatric GHD in certain major markets, the royalties will transition to regional, tiered gross profit sharing for both hGH-CTP and Pfizer’s Genotropin®.
Pursuant to our agreement with Pfizer, we agreed to lead the clinical development activities for the hGH-CTP program and are responsible for funding the development programs for the key indications, including Adult and Pediatric GHD and Pediatric SGA. Pfizer agreed to be responsible for all development costs for additional indications as well as all post-marketing studies. In addition, Pfizer agreed to fund the commercialization activities for all indications and lead the manufacturing activities covered by the global development plan. The agreement obligated us to fund development up to an agreed cap. We have exceeded the development cap and if we are unable to reach an agreement with Pfizer regarding cost sharing for the overruns, as well as other obligations, including development obligations, it could have a material adverse impact on the expected benefits of the Pfizer transaction.
GHD occurs when the production of growth hormone, secreted by the pituitary gland, is disrupted. Since growth hormone plays a critical role in stimulating body growth and development, and is involved in the production of muscle protein and in the breakdown of fats, a decrease in the hormone affects numerous body processes. hGH is used for the long-term treatment of children and adults with inadequate secretion of endogenous growth hormone. The primary indications it treats in children are GHD, SGA, kidney disease, Prader-Willi Syndrome and Turner’s Syndrome. In adults, the primary indications are replacement of endogenous growth hormone and the treatment of AIDS-induced weight loss. Patients using hGH receive daily injections six or seven times a week. This is particularly burdensome for pediatric patients. We believe a significant market opportunity exists for a longer-lasting version of hGH that would require fewer injections.
Our phase 3 trial of hGH-CTP in pediatric patients was initiated in December 2016 and patient enrollment was completed in August 2018. The global study is a 225-patient study in Pediatric GHD patients designed to evaluate weekly treatment with hGH-CTP versus daily injections of Genotropin. The hGH-CTP is delivered in a pen device in this multi-regional study in over 21 countries. The GHD subjects will be treated weekly for 12 months. We expect to perform top-line data analysis from the study in the fourth quarter of 2019. In addition to the phase 3 pediatric study, we have continued without interruption our ongoing phase 2 pediatric open label extension study for hGH-CTP. The phase 2 pediatric patients have been treated with hGH-CTP for over four years, and some patients for over five years. We have switched all of the pediatric patients in this study to the disposable pen device. We have also initiated a 44-patient study in Pediatric GHD patients in Japan which has completed enrollment. hGH-CTP has orphan drug designation in the U.S. and Europe for both adults and children with GHD.
In December 2016, we announced preliminary topline data from our phase 3, double blind, placebo controlled study of hGH-CTP in adults with GHD. The multinational, multi-center study, which utilized a 2:1 randomization between hGH-CTP and placebo, enrolled 203 subjects, 198 of whom received at least one dose of study treatment. Treatment was administered through a weekly injection. The topline results showed:
•The active group had a mean change in trunk fat mass of -0.4kg and placebo group was 0;
•There was no statistically significant difference (≤ 0.05 (p value)) between the active and placebo group;
•97% of hGH-CTP vs 6% of placebo group showed IGF-1 normalization; and
•The safety profile of hGH-CTP is consistent with that observed with those treated with daily growth hormone
Although there was no statistically significant difference between hGH-CTP and placebo on the primary endpoint of change in trunk fat mass from baseline to 26 weeks, after unblinding the study, we identified an exceptional value of trunk fat mass reduction in the placebo group that may have affected the primary outcome. We have completed post-hoc sensitivity analyses to evaluate the influence of outliers on the primary endpoint results using multiple statistical approaches. Analyses that excluded outliers showed a statistically significant difference between hGH-CTP and placebo on the change in trunk fat mass. Additional analyses that did not exclude outliers showed mixed results. Following completion of the analyses, OPKO and Pfizer have agreed that OPKO may communicate with the FDA regarding a potential biologics license application (“BLA”) submission.
Factor VII
In addition to hGH-CTP, we are developing a product to extend the life span of Factor VIIa (hemophilia) using the CTP technology. In February 2013, the FDA granted orphan drug designation to our longer-acting version of clotting Factor VIIa, Factor VIIa-CTP, for the treatment of bleeding episodes in patients with hemophilia A or B with inhibitors to Factor VIII or Factor IX. Currently, Factor VIIa therapy is available only as an intravenous (IV) formulation which, due to Factor VIIa’s short half-life, requires multiple infusions to treat a bleeding episode. In addition, frequent infusions are onerous when used as preventative prophylactic therapy, especially for children.
13
We have conducted a phase 1/2a dose escalation study and a phase 1 dose escalating subcutaneous study in healthy volunteers to determine safety of our long acting Factor VIIa-CTP for the treatment of bleeding episodes in hemophilia A or B patients with inhibitors to Factor VIII or Factor IX. These two studies are completed, and data assessment is ongoing. Further regulatory and development strategies will be planned.
We believe that the CTP technology may also be broadly applicable to other therapeutic proteins in the market and provide a reduction in the number of injections.
APIs
FineTech Pharmaceutical, Ltd. (“FineTech”), is our Israeli-based subsidiary that develops and produces high value, high potency specialty APIs. Through its FDA registered facility in Nesher, Israel, FineTech currently manufactures commercial APIs for sale or license to pharmaceutical companies in the U.S., Canada, Europe and Israel. We believe that FineTech’s significant know-how and experience with analytical chemistry and organic syntheses, together with its production capabilities, may play a valuable role in the development of our pipeline of proprietary molecules and compounds for diagnostic and therapeutic products, while providing revenues and profits from its existing API business.
Oligonucleotide Therapeutics
OPKO CURNA, LLC (“CURNA”), previously CURNA Inc., is engaged in the discovery of new drugs for the treatment of a wide variety of illnesses, including cancer, heart disease, metabolic disorders and a range of genetic anomalies. CURNA’s platform technology utilizes a short, single strand oligonucleotide and is based on the up-regulation of protein production through interference with non-coding RNA’s or natural antisense. This strategy contrasts with established approaches which down-regulate protein production. CURNA has designed a novel type of therapeutic modality, termed AntagoNAT, and has initially demonstrated this approach for up-regulation of several therapeutically relevant proteins in in vitro and animal models.
CURNA has identified and developed potential active compounds which increase the production of over 80 key proteins involved in a large number of individual diseases. We have ongoing pre-clinical studies for several of these compounds. A lead compound has been identified for the treatment of Dravet Syndrome. Orphan disease designations are granted by FDA and EMA.
NK-1 Program
We acquired rolapitant and other neurokinin-1 (“NK-1”) assets from Merck & Co. In December 2010, we exclusively out-licensed the development, manufacture and commercialization of our lead NK-1 candidate, VARUBI™ (rolapitant), to TESARO, Inc. (“TESARO”). VARUBI™, a potent and selective competitive antagonist of the NK-1 receptor, had successfully completed clinical testing for prevention of chemotherapy induced nausea and vomiting (“CINV”) and post-operative induced nausea and vomiting. TESARO’s NDA for oral VARUBI™ was approved by the FDA in September 2015, and in November 2015, TESARO commenced the commercial launch of oral VARUBI™ in the U.S. TESARO’s IV formulation of VARUBI™ was approved by the FDA in October 2017 and commercial sales commenced in November 2017. In January 2018, the package insert for VARUBI™ was updated to include mention of new adverse effects, including anaphylaxis, anaphylactic shock and other serious hypersensitivity reactions which were reported following its introduction to the market in November 2017. In late February 2018, TESARO announced it would suspend distribution of VARUBI™ IV, but would continue to support the oral product.
Under the terms of the license, we received a $6.0 million upfront payment from TESARO and we received $30.0 million of milestone payments upon achievement of certain regulatory and commercial sale milestones. We are eligible to receive additional commercial milestone payments of up to $85.0 million if specified levels of annual net sales are achieved. TESARO is also obligated to pay us tiered royalties on annual net sales achieved in the U.S. and Europe at percentage rates that range from the low double digits to the low twenties, and outside of the U.S. and Europe at low double-digit percentage rates. TESARO assumed responsibility for clinical development and commercialization of licensed products at its expense. Under the agreement, we will continue to receive royalties on a county-by-country and product-by-product basis until the later of the date that all of the patents rights licensed from us and covering rolapitant expire, are invalidated or are not enforceable, and 12 years from the date of the first commercial sale of the product.
If TESARO elects to develop and commercialize VARUBI™ in Japan through a third-party licensee, TESARO will share equally with us all amounts it receives in connection with such activities, subject to certain exceptions and deductions. The term of the license will remain in force until the expiration of the royalty term unless we terminate the license earlier for TESARO’s material breach of the license or bankruptcy. TESARO has a right to terminate the license during the term for any reason on three month’s written notice.
14
In June 2018, TESARO assigned its rights and obligations under the agreement to TerSera Therapeutics LLC (“TerSera”) pursuant to an asset purchase agreement. Under the asset purchase agreement, TerSera is responsible for VARUBI in the U.S.and Canada and TESARO can continue to commercialize VARUBY® in Europe and the rest of the world though a sublicense with TerSera.
Commercial Operations
We also intend to continue to leverage our global commercialization expertise to pursue acquisitions of commercial businesses that will both drive our growth and provide geographically diverse sales and distribution opportunities. During 2015, we acquired EirGen Pharma Ltd. (“EirGen”), a specialty pharmaceutical company based in Ireland. EirGen is focused on the development and commercial supply of high potency, high barrier to entry, pharmaceutical products. Through its facility in Waterford, Ireland, EirGen currently manufactures high potency pharmaceutical products and exports to over 50 countries. High potency drugs such as those used for cancer chemotherapy are typically unsuitable for manufacture in normal multi-product facilities due to cross contamination risks.
To date, EirGen and its commercial partners have filed several product applications with the FDA in Europe and in Japan. EirGen has a strong research and development portfolio of high barrier to entry drugs and we expect to rapidly expand its drug portfolio. We believe EirGen will play an important role in the development, manufacturing, distribution and approval of a wide variety of drugs in a variety of dosage forms with an emphasis on high potency products.
OPKO Health Europe (previously Farmadiet Group Holding, S.L.) operates primarily in Spain and has more than 20 years of experience in the development, manufacture, marketing and sale of pharmaceutical, nutraceutical and veterinary products in Europe.
OPKO Mexico (previously Pharmacos Exakta S.A. de C.V.), is engaged in the manufacture, marketing, sale and distribution of ophthalmic and other pharmaceutical products to private and public customers in Mexico. OPKO Mexico is commercializing food supplements and over the counter products, and manufactures and sells products primarily in the generics market in Mexico, although it also has some proprietary products as well.
OPKO Chile (previously Pharma Genexx, S.A.) markets, sells and distributes pharmaceutical products to the private, hospital, pharmacy and public institutional markets in Chile for a wide range of indications, including, cardiovascular products, vaccines, antibiotics, gastro- intestinal products and hormones, among others. ALS Distribuidora Limitada (“ALS”) is engaged in the business of importation, commercialization and distribution of pharmaceutical products for private markets in Chile. ALS started operations in 2009 as the exclusive product distributor of Arama Laboratorios y Compañía Limitada (“Arama”), a company with more than 20 years of experience in the pharmaceutical products market. In connection with the acquisition of ALS, OPKO acquired all of the product registrations and trademarks previously owned by Arama, as well as the Arama name. We distribute food supplements and over the counter products through Arama.
Strategic Investments
We have and may continue to make investments in other early stage companies that we perceive to have valuable proprietary technology and significant potential to create value for OPKO as a shareholder.
RESEARCH AND DEVELOPMENT EXPENSES
During the years ended December 31, 2018, 2017, and 2016, we incurred $125.6 million, $126.4 million, and $113.9 million, respectively, of research and development expenses related to our various product candidates. During the years ended December 31, 2018, 2017, and 2016, our research and development expenses primarily consisted of hGH-CTP and Rayaldee development programs, including expenses related to the development of hGH-CTP and phase 3 clinical trials for Rayaldee.
INTELLECTUAL PROPERTY
We believe that technology innovation is driving breakthroughs in healthcare. We have adopted a comprehensive intellectual property strategy which blends the efforts to innovate in a focused manner with the efforts of our business development activities to strategically in-license intellectual property rights. We develop, protect, and defend our own intellectual property rights as dictated by the developing competitive environment. We value our intellectual property assets and believe we have benefited from early and insightful efforts at understanding diagnostics, as well as the disease and the molecular basis of potential pharmaceutical intervention.
We actively seek, when appropriate and available, protection for our products and proprietary information by means of U.S. and foreign patents, trademarks, trade secrets, copyrights, and contractual arrangements. Patent protection in the
15
pharmaceutical and diagnostic fields, however, can involve complex legal and factual issues. There can be no assurance that any steps taken to protect such proprietary information will be effective.
We own or license-in thousands of U.S. and foreign patents and applications for our products, product candidates and our outlicensed product candidates. These patents cover pharmaceuticals, diagnostics and other products and their uses, pharmaceutical and diagnostic compositions and formulations and product manufacturing processes. Our patents are filed in various locations worldwide as is appropriate to the particular patent and its use.
Rayaldee
We have multiple U.S. patent families relating to Rayaldee. These patents are also filed in multiple countries worldwide. One patent family claims a sustained release oral dosage formulation and a method of treating 25-hydroxyvitamin D insufficiency or deficiency and will not expire until at least February 2027. A second patent family claims a method of administering 25-hydroxyvitamin D3 by controlled release, a formulation for controlled release of a vitamin D compound, a controlled release oral dosage formulation of a vitamin D compound and a method of treatment, and will not expire until at least April 2028. We also have additional patents and patent applications pending relating to the sustained release formulation and its use which will expire in 2034 and have licensed patents covering the capsule shell. The patents issued in the U.S. covering Rayaldee are listed in the Approved Drug Products with Therapeutic Equivalence Evaluations, or the Orange Book. OPKO and/or its affiliates have entered into two exclusive license agreements with respect to Rayaldee patents in certain territories outside of North America with VFMCRP (Europe plus) and JT (Japan).
Rolapitant
The rolapitant line of patents, exclusively licensed to TESARO and TerSera includes multiple patent families that cover anti-nausea treatment for chemotherapy patients. These U.S. patents are also filed and granted in many countries around the world. One patent family covers the chemical composition of rolapitant and related compounds and expires in December 2023 (with the patent term adjustment). A patent term extension request was submitted to the USPTO in October 2015 to obtain an additional 1,716 days which will, upon approval, extend the rolapitant compound patent expiration date to August 2028.The second patent family covers pharmaceutical formulations, including a capsule formulation with a related method of use and expires in April of 2027. The third patent family covers particular aspects of the chemical composition of rolapitant as well as certain methods of treating delayed onset nausea and expires in April 2027. The fourth patent family covers a powdered pharmaceutical composition of a crystalline salt of rolapitant and expires in March 2028. The current line of rolapitant patents are approved for oral treatment. Patents and applications directed towards the IV formulation of rolapitant are granted and/or currently pending in multiple jurisdictions.
hGH-CTP
The hGH-CTP line of patents, which is currently licensed to Pfizer, Inc., includes two main patent families that cover modified human grown hormone treatment. These U.S. patents are also filed in multiple countries around the world. One patent family covers certain CTP modified hGH polypeptides relating to growth hormones and their method of use and expires in February of 2027 (with the exception of two U.S. patents, namely US 8304386 and US 8097435, which expire in January 2028 and April 2027, respectively, due to Patent Term Adjustment for each). The second patent family covers cytokine-based polypeptides relating to human growth hormone treatment and expires in 2027. In addition to the CTP patents and applications licensed to Pfizer, OPKO has multiple patent families covering similar biologicals with patents and applications pending in the U.S. and internationally.
OPK88003 and OPK88004
In 2016, we acquired Transition Therapeutics, Inc. which is developing multiple drug candidates that include OPK88003 (a long acting oxyntomodulin) and OPK88004 (a selective androgen receptor modulator (SARM)), each of which are licensed from Eli Lilly and have granted patents worldwide covering the compounds and their use in their respective indications. U.S. Pat. No. 8367607 covers OPK88003 and expires in December 2030, without extension. U.S. Pat. No. 7968587 covers OPK88004 and expires, without extension, in November 2027. In addition to the molecule patent covering the selective androgen receptor modulator, Transition Therapeutics exclusively licensed a method of use patent family covering its use in treating androgen deprivation therapy associated symptoms. These patents expire in 2035. In addition, Transition and its affiliates have patented compounds (scyllo-inositol) for the treatment of Alzheimer’s disease. The patents are pending or granted in many countries of the world. We and/or our affiliates will seek all available patent term extensions for our product candidates and products.
Because the patent positions of pharmaceutical, biotechnology, and diagnostics companies are highly uncertain and involve complex legal and factual questions, the patents owned and licensed by us, or any future patents, may not prevent other
16
companies from developing similar or therapeutically equivalent products or ensure that others will not be issued patents that may prevent the sale of our products or require licensing and the payment of significant fees or royalties. Furthermore, to the extent that any of our future products or methods are not patentable, that such products or methods infringe upon the patents of third parties, or that our patents or future patents fail to give us an exclusive position in the subject matter claimed by those patents, we will be adversely affected. We may be unable to avoid infringement of third party patents and may have to obtain a license, defend an infringement action, or challenge the validity of the patents in court. A license may be unavailable on terms and conditions acceptable to us, if at all. Patent litigation is costly and time consuming, and we may be unable to prevail in any such patent litigation or devote sufficient resources to even pursue such litigation.
LICENSES AND COLLABORATIVE RELATIONSHIPS
Our strategy is to develop a portfolio of product candidates through a combination of internal development, acquisition, and external partnerships. Collaborations are key to our strategy and we continue to build relationships and forge partnerships in various areas where unmet medical need and commercial opportunities exist. In October 2017, we entered into a license and development agreement with JT for the development and commercialization of Rayaldee in Japan for the treatment of SHPT in non-dialysis and dialysis patients with CKD. In May 2016, we entered into a license and collaboration with VFMCRP for the development and commercialization of Rayaldee in Europe, Canada, Mexico, Australia, South Korea and certain other international markets for the treatment of SHPT in patients with CKD and vitamin D insufficiency. In December 2014, we entered into an exclusive agreement with Pfizer for the development and commercialization of our long-acting hGH-CTP for the treatment of GHD in adults and children, as well as for the treatment of growth failure in children born small for gestational age. Previously, we (or entities we have acquired) have completed strategic licensing transactions with the President and Fellows of Harvard College, Academia Sinica, The Scripps Research Institute, TESARO, INEOS Healthcare, and Arctic Partners, among others.
COMPETITION
The pharmaceutical and diagnostic testing industries are highly competitive and require an ongoing, extensive search for technological innovation. The industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. They also require, among other things, the ability to effectively discover, develop, test and obtain regulatory approvals for products, as well as the ability to effectively commercialize, market and promote approved products.
Numerous companies, including major pharmaceutical companies, specialty pharmaceutical companies and specialized biotechnology companies, are engaged in the development, manufacture and marketing of pharmaceutical products competitive with those that we are or intend to commercialize ourselves and through our partners. Competitors to our diagnostics business include major diagnostic companies, reference laboratories, molecular diagnostic firms, universities and research institutions. Most of these companies have substantially greater financial and other resources, larger research and development staffs and more extensive marketing and manufacturing organizations than ours. This enables them, among other things, to make greater research and development investments and efficiently utilize their research and development costs, as well as their marketing and promotion costs, over a broader revenue base. This also provides our competitors with a competitive advantage in connection with the highly competitive product acquisition and product in-licensing process, which may include auctions in which the highest bidder wins. Our competitors may also have more experience and expertise in obtaining marketing approvals from the FDA and other regulatory authorities. In addition to product development, testing, approval, and promotion, other competitive factors in the pharmaceutical and diagnostics industry include industry consolidation, product quality and price, product technology, reputation, customer service, and access to technical information.
In our clinical laboratory operations, we compete with three types of providers in a highly fragmented and competitive industry: hospital laboratories, physician-office laboratories and other independent clinical laboratories. Our major competitors in the New York metropolitan area are two of the largest national laboratories, Quest Diagnostics and Laboratory Corporation of America. Although we are much smaller than these national laboratories, we believe that we compete successfully with them in our region due to our innovative testing services and our level of service. We believe our responses to medical consultation are faster and more personalized than those of the national laboratories. Our client service staff deals only with basic technical questions and those that have medical or scientific significance are referred directly to our senior scientists and medical staff.
We are commercializing our 4Kscore product in the U.S., Europe and Mexico in a laboratory setting and seek to capitalize on near-term commercialization opportunities for our proprietary diagnostic point-of-care system by transitioning laboratory-based tests, including the PSA, and testosterone and other tests to our point-of-care system. We expect to leverage BioReference’s national marketing, sales and distribution resources, along with its almost 300-person sales and marketing team
17
to support commercialization of the 4Kscore and other diagnostic products. Competitors to our diagnostics business are many and include major diagnostic companies, molecular diagnostic firms, universities, and research institutions.
Pricing and reimbursement coverage positions, including the recent future non-coverage determination by Novitas, could substantially impact the competitiveness of the 4Kscore test and our other diagnostic products. See “Risk Factors - If the 4Kscore test is not covered and eligible for reimbursement from government and third party payors, we may not be able to generate significant revenue for the product.”
Our ability to commercialize our pharmaceutical and diagnostic test product candidates and compete effectively will depend, in large part, on:
• |
our ability to meet all necessary regulatory requirements to advance our product candidates through clinical trials and the regulatory approval process in the U.S. and abroad; |
• |
the perception by physicians and other members of the health care community of the safety, efficacy, and benefits of our products compared to those of competing products or therapies; |
• |
our ability to manufacture products we may develop on a commercial scale; |
• |
the effectiveness of our sales and marketing efforts; |
• |
the willingness of physicians to adopt a new diagnostic or treatment regimen represented by our technology; |
• |
our ability to secure reimbursement for our product candidates; |
• |
the price of the products we may develop and commercialize relative to competing products; |
• |
our ability to accurately forecast and meet demand for our product candidates if regulatory approvals are achieved; |
• |
our ability to develop a commercial scale infrastructure either on our own or with a collaborator, which would include expansion of existing facilities, including our manufacturing facilities, development of a sales and distribution network, and other operational and financial systems necessary to support our increased scale; |
• |
our ability to maintain a proprietary position in our technologies; and |
• |
our ability to rapidly expand the existing information technology infrastructure and configure existing operational, manufacturing, and financial systems (on our own or with third party collaborators) necessary to support our increased scale, which would include existing or additional facilities and or partners. |
GOVERNMENT REGULATION
The U.S. government regulates healthcare through various agencies, including but not limited to the following: (i) the FDA, which administers the Federal Food, Drug and Cosmetic Act (“FDCA”), as well as other relevant laws; (ii) the Centers for Medicare & Medicaid Services (“CMS”), which administers the Medicare and Medicaid programs; (iii) the Office of Inspector General (“OIG”), which enforces various laws aimed at curtailing fraudulent or abusive practices, including by way of example, the Anti-Kickback Statute, the Physician Self-Referral Law, commonly referred to as the Stark law, the Civil Monetary Penalty Law (including the beneficiary inducement prohibition) (“CMP”), and the laws that authorize the OIG to exclude healthcare providers and others from participating in federal healthcare programs; and (iv) the Office of Civil Rights, which administers the privacy aspects of the Health Insurance Portability and Accountability Act of 1996. All of the aforementioned are agencies within the Department of Health and Human Services (“HHS”). Healthcare is also provided or regulated, as the case may be, by the Department of Defense through its TRICARE program, the Department of Veterans Affairs, especially through the Veterans Health Care Act of 1992, the Public Health Service within HHS under Public Health Service Act § 340B (42 U.S.C. § 256b), the Department of Justice through the Federal False Claims Act and various criminal statutes, and state governments under the Medicaid and other state sponsored or funded programs and their internal laws regulating all healthcare activities.
The testing, manufacture, distribution, advertising, and marketing of drug and diagnostic products and medical devices, as well as the performance of clinical testing services, are subject to extensive regulation by federal, state, and local governmental authorities in the U.S., including the FDA, and by similar agencies in other countries. Any drug, diagnostic, or device product that we develop must receive all relevant regulatory approvals or clearances, as the case may be, before it may be marketed in a particular country.
Clinical Laboratory Operations
18
Our clinical laboratory operations are subject to regulations, which are designed to ensure the quality and reliability of clinical laboratories by mandating specific standards in the areas of personnel qualifications, administration and participation in proficiency testing, patient test management, quality control, quality assurance and inspections. Laboratories must undergo on-site surveys at least every two years, which may be conducted by CMS under the Federal Clinical Laboratory Improvement Amendments (“CLIA”) program or by a private CMS approved accrediting agency. The sanction for failure to comply with CLIA requirements may be suspension, revocation or limitation of a laboratory’s CLIA certificate, which is necessary to conduct business, as well as significant fines and/or criminal penalties. We are also subject to regulation of laboratory operations under state clinical laboratory laws. State clinical laboratory laws may require that laboratories and/or laboratory personnel meet certain qualifications, specify certain quality controls or require maintenance of certain records. Certain states, such as New York, California, Maryland, Pennsylvania, and Rhode Island, each require that we obtain licenses to test specimens from patients residing in those states and additional states may require similar licenses in the future. Only Washington and New York State are exempt under CLIA, as these states have established laboratory quality standards at least as stringent as CLIA’s. Potential sanctions for violation of these statutes and regulations include significant fines and the suspension or loss of various licenses, certificates and authorizations.
Our clinical laboratory operations are subject to complex laws, regulations and licensure requirements relating to billing and payment for laboratory services, sales and marketing interactions with ordering physicians and other health care providers, security and confidentiality of health information, and environmental and occupational safety, among others. Changes in regulations often increase the cost of testing or processing claims. Also, these laws may be interpreted or applied by a prosecutorial, regulatory or judicial authority in a manner that could require us to make changes in our operations, including in our pricing, billing and/or marketing practices in a manner that could adversely affect operations.
Drug Development
The regulatory process, which includes overseeing preclinical studies and clinical trials of each pharmaceutical compound to establish its safety and efficacy and confirmation by the FDA that good laboratory, clinical, and manufacturing practices were maintained during testing and manufacturing, can take many years, requires the expenditure of substantial resources, and gives larger companies with greater financial resources a competitive advantage over us. Delays or terminations of clinical trials that we undertake would likely impair our development of product candidates. Delays or terminations could result from a number of factors, including stringent enrollment criteria, slow rate of enrollment, size of patient population, having to compete with other clinical trials for eligible patients, geographical considerations, failure to meet anticipated clinical success, patient safety concerns, and others.
Although accelerated pathways for approval exist for certain drugs, generally, FDA review processes can be lengthy and unpredictable, and we may encounter delays or rejections of our applications when submitted. Generally, in order to gain FDA approval, we must first conduct preclinical studies in a laboratory and in animal models to obtain preliminary information on a compound and to identify any safety problems. The results of these studies are submitted as part of an IND application that the FDA must review before human clinical trials of an investigational drug can commence.
Clinical trials are normally done in three sequential phases and generally take two to five years or longer to complete. phase 1 consists of testing the drug product in a small number of humans, normally healthy volunteers, to determine preliminary safety and tolerable dose range. Phase 2 usually involves studies in a limited patient population to evaluate the effectiveness of the drug product in humans having the disease or medical condition for which the product is indicated, determine dosage tolerance and optimal dosage, and identify possible common adverse effects and safety risks. Phase 3 consists of additional controlled testing at multiple clinical sites to establish clinical safety and effectiveness in an expanded patient population of geographically dispersed test sites to evaluate the overall benefit-risk relationship for administering the product and to provide an adequate basis for product labeling. Phase 4 clinical trials may be conducted- and are sometimes required - after approval to gain additional experience from the treatment of patients in the intended therapeutic indication. There are also certain situations when drugs and biologics are eligible for one of FDA’s expedited approval programs, designed to shorten review and development time.
After completion of clinical trials of a new drug product, FDA and foreign regulatory authority marketing approval must be obtained. Assuming that the clinical data support the product’s safety and effectiveness for its intended use, a Biologics License Application (BLA) or an NDA is submitted to the FDA for its review. Since the early 1990s, the FDA has managed a user fee program whereby sponsors of drug applications pay a fee to the agency and the agency commits to meeting a series of performance goals designed to reduce drug review times. Generally, it takes one to three years to obtain approval. If questions arise during the FDA review process, approval may take a significantly longer period of time. The testing and approval processes require substantial time and effort and we may not receive approval on a timely basis, if at all, or the approval that we receive may be for a narrower indication than we had originally sought, potentially undermining the commercial viability of the product. Even if regulatory approvals are obtained, a marketed product is subject to continual review, and later discovery of
19
previously unknown problems or failure to comply with the applicable regulatory requirements may result in restrictions on the marketing of a product or withdrawal of the product from the market as well as possible civil or criminal sanctions. For marketing outside the U.S., we also will be subject to foreign regulatory requirements governing human clinical trials and marketing approval for pharmaceutical products. The requirements governing the conduct of clinical trials, product licensing, pricing, and reimbursement vary widely from country to country.
In addition to clinical trial rules, FDA imposes other requirements on applicants including obligations related to Good Manufacturing Practices (GMPs), proper labeling, and other issues related to manufacturing and marketing a drug.
Other than Rayaldee, none of our pharmaceutical products under development have been approved for marketing in the U.S. or elsewhere. We may not be able to obtain regulatory approval for any such products under development in a timely manner, if at all. Failure to obtain requisite governmental approvals or failure to obtain approvals of the scope requested will delay or preclude us, or our licensees or marketing partners, from marketing our products, or limit the commercial use of our products, and thereby would have a material adverse effect on our business, financial condition, and results of operations. See “Risk Factors — The results of pre-clinical trials and previous clinical trials for our products may not be predictive of future results, and our current and planned clinical trials may not satisfy the requirements of the FDA or other non-U.S. regulatory authorities.”
Device Development
Medical devices are subject to varying levels of premarket regulatory control, the most comprehensive of which requires human clinical trials be conducted before a device receives approval for commercial distribution. The FDA classifies medical devices into one of three classes based upon their risk profile (both to the patient and provider): Class I devices are relatively simple “low risk” technologies, and can be manufactured and distributed with general controls without a premarket clearance or approval from the FDA; Class II devices are somewhat more complex “moderate risk” devices, and require greater scrutiny from the agency, requiring a premarket clearance from the FDA before market entry; Class III devices are “high risk” technologies inserted or implanted in the body, intended to treat life sustaining functions. These Class III technologies require a premarket approval from the FDA before market entry.
In the U.S., a company generally can obtain permission to distribute a new device in one of two ways. The first applies to a Class II device that is substantially equivalent to a device first marketed prior to May 1976, or to another device marketed after that date, but which was substantially equivalent to a pre-May 1976 device. To obtain FDA permission to distribute the device, a company generally must submit a section 510(k) premarket notification, and receive an FDA order finding substantial equivalence to a predicate device (pre-May 1976 or post-May 1976 device that was substantially equivalent to a pre-May 1976 device) and permitting commercial distribution of that device for its intended use. A 510(k) submission must provide information supporting a claim of substantial equivalence to the predicate device. If clinical data from human experience are required to support the 510(k) submission, these data must be gathered in compliance with investigational device exemption (“IDE”), regulations for investigations performed in the U.S. The 510(k) process is normally used for products of the type that the Company proposes distributing. The FDA review process for premarket notifications submitted pursuant to section 510(k) takes, on average, about 90 days, but it can take substantially longer if the FDA has concerns, and there is no guarantee that the FDA will “clear” the device for marketing, in which case the device cannot be distributed in the U.S. There is also no guarantee that the FDA will deem the applicable device subject to the 510(k) process, as opposed to the more time-consuming, resource-intensive and problematic, PMA process described below.
The second, more comprehensive, PMA process, which can take a year or longer, applies to a new device that is not substantially equivalent to a pre-1976 product or that is to be used in supporting or sustaining life or preventing impairment. These devices are normally Class III devices. For example, most implantable devices are subject to the approval process. Two steps of FDA approval are generally required before a company can market a product in the U.S. that is subject to approval, as opposed to clearance. First, a company must comply with IDE regulations in connection with any human clinical investigation of the device. These regulations permit a company to undertake a clinical study of a “non-significant risk” device without formal FDA approval. Prior express FDA approval is required if the device is a significant risk device. Second, the FDA must review the company’s PMA application, which contains, among other things, clinical information acquired under the IDE. The FDA will approve the PMA application if it finds there is reasonable assurance that the device is safe and effective for its intended use. The PMA process takes substantially longer than the 510(k) process and it is conceivable that the FDA would not agree with our assessment that a device that we propose to distribute should be a Class I or Class II device. If that were to occur we would be required to undertake the more complex and costly PMA process. However, for either the 510(k) or the PMA process, the FDA could require us to run clinical trials, which would pose all of the same risks and uncertainties associated with the clinical trials of drugs, described above.
20
In December of 2016, Congress enacted the 21st Century Cures Act (P.L. 114-255) which contained provisions establishing a new Breakthrough Device pathway to allow faster patient access to devices and breakthrough technologies that provide for more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases, for which no approved or cleared treatment exists or that offer significant advantages over existing approved or cleared alternatives. FDA has just begun to implement this program and it is not clear if any of our products would be eligible.
Even when a clinical study has been approved by the FDA or deemed approved, the study is subject to factors beyond a manufacturer’s control, including, but not limited to the fact that the institutional review board at a given clinical site might not approve the study, might decline to renew approval which is required annually, or might suspend or terminate the study before the study has been completed. Also, the interim results of a study may not be satisfactory, leading the sponsor to terminate or suspend the study on its own initiative or the FDA may terminate or suspend the study. There is no assurance that a clinical study at any given site will progress as anticipated; there may be an insufficient number of patients who qualify for the study or who agree to participate in the study or the investigator at the site may have priorities other than the study. Also, there can be no assurance that the clinical study will provide sufficient evidence to assure the FDA that the product is safe and effective, a prerequisite for FDA approval of a PMA, or substantially equivalent in terms of safety and effectiveness to a predicate device, a prerequisite for clearance under 510(k). Even if the FDA approves or clears a device, it may limit its intended uses in such a way that manufacturing and distributing the device may not be commercially feasible. For marketing outside the U.S., we also will be subject to foreign regulatory requirements governing clinical trials and marketing approval for medical devices. The requirements governing the conduct of clinical trials, device clearance/approval, pricing, and reimbursement vary widely from country to country. In addition to the regulatory clearance and approval processes described herein, the FDA periodically issues draft guidance documents designed to provide additional detail on or reform aspects of the 510(k) and PMA clearance and approval processes. To the extent the FDA finalizes and implements these documents, the average 510(k) and PMA submission requirements and review times may change and devices that might previously have been cleared under the 510(k) process may require approval under the PMA process (and vice-versa). Additionally, since 2012, the FDA has collected user fees for the review of certain premarket submissions received on or after October 1, 2012, including 510(k) and PMA applications. These fees are intended to improve the device review process, but it is still too early to assess the actual impact on the industry.
After clearance or approval to market is given, the FDA and foreign regulatory agencies, upon the occurrence of certain events, are authorized under various circumstances to withdraw the clearance or approval or require changes to a device, its manufacturing process or its labeling or additional proof that regulatory requirements have been met.
A manufacturer of a device approved through the PMA is not permitted to make changes to the device, which affects its safety or effectiveness without first submitting a supplement application to its PMA and obtaining FDA approval for that supplement. In some instances, the FDA may require clinical trials to support a supplement application. A manufacturer of a device cleared through the 510(k) process must submit another premarket notification if it intends to make a change or modification in the device that could significantly affect the safety or effectiveness of the device, such as a significant change or modification in design, material, chemical composition, energy source or manufacturing process. Any change in the intended uses of a PMA device or a 510(k) device requires an approved PMA supplement or a cleared premarket notification. Exported devices are subject to the regulatory requirements of each country to which the device is exported, as well as certain FDA export requirements.
A company that intends to manufacture medical devices is required to register with the FDA before it begins to manufacture the device for commercial distribution. As a result, we and any entity that manufactures products on our behalf will be subject to periodic inspection by the FDA for compliance with the FDA’s Quality System Regulation requirements and other regulations. In the European Community, we will be required to maintain certain International Organization for Standardization (“ISO”), certifications in order to sell products and we or our manufacturers undergo periodic inspections by notified bodies to obtain and maintain these certifications. These regulations require us or our manufacturers to manufacture products and maintain documents in a prescribed manner with respect to design, manufacturing, testing and control activities. Further, we are required to comply with various FDA and other agency requirements for labeling and promotion. The Medical Device Reporting regulations require that we provide information to the FDA whenever there is evidence to reasonably suggest that a device may have caused or contributed to a death or serious injury or, if a malfunction were to occur, could cause or contribute to a death or serious injury. In addition, the FDA prohibits us from promoting a medical device for unapproved indications.
21
Diagnostic Products
Certain of our diagnostic products in development are subject to regulation by the FDA and similar international health authorities. For these products, we have an obligation to adhere to the FDA’s cGMP regulations. Additionally, we are subject to periodic FDA inspections, quality control procedures, and other detailed validation procedures. If the FDA finds deficiencies in the validation of our manufacturing and quality control practices, they may impose restrictions on marketing specific products until corrected.
Regulation by governmental authorities in the U.S. and other countries may be a significant factor in how we develop, test, produce and market our diagnostic test products. Diagnostic tests like ours may not fall squarely within the regulatory approval process for pharmaceutical or device products as described above, and the regulatory pathway is not as clear. Although the FDA regulates in vitro diagnostic devices, some companies have successfully commercialized diagnostic tests for various conditions and disease states without seeking clearance or approval for such tests through a 510(k) or PMA approval process. These tests are known as laboratory developed tests (“LDTs”) and are designed, manufactured, and used within a single laboratory that is certified under the Clinical Laboratory Improvement Amendments of 1988 (“CLIA”). CLIA is a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for diagnostic, preventative or treatment purpose. Such LDT testing is currently under the purview of CMS and state agencies that provide oversight of the safe and effective use of LDTs.
However, the FDA has consistently asserted that it has the regulatory authority to regulate LDTs despite historically exercising enforcement discretion. In furtherance of that position, the FDA issued two draft guidance documents in October 2014: (1) Framework for Regulatory Oversight of Laboratory Developed Tests (the “Framework Guidance”); and (2) FDA Notification and Medical Device Reporting for Laboratory Developed Tests (the “Notification Guidance”). The Framework Guidance outlines the FDA’s plan to adopt over time a risk-based approach to regulating LDTs whereby different classifications of LDTs would be subject to different levels of FDA oversight and enforcement, including, for example, prohibitions on adulteration and misbranding, establishment registration and device listing, premarket notification, banned devices, records and reports, good manufacturing practices, adverse event reporting, premarket review of safety, effectiveness, and clinical validity, and quality system requirements. The Notification Guidance is intended to explain how clinical laboratories should notify the FDA of the LDTs they develop and how to satisfy Medical Device Reporting requirements. However, the FDA indicated in November 2016 that it would delay implementation of the Framework Guidance and the Notification Guidance, and seek additional input from industry. In addition, on January 13, 2017, the FDA published a synthesis of feedback on the Framework Guidance and Notification Guidance titled, Discussion Paper on Laboratory Developed Tests (the “Discussion Paper”). The Discussion Paper provided notice that the FDA would not issue a final guidance on the oversight of LDTs to allow for further public discussion on appropriate oversight approach, and to give congressional authorizing committees the opportunity to develop a legislative solution.
If finalized in the October 2014 format, the Framework Guidance and the Notification Guidance may have a materially adverse effect on the time, cost, and risk associated with the Company’s development and commercialization of LDTs for the U.S. market, and there can be no assurance that clearances or approvals sought by the Company will be granted and maintained. However, the FDA’s authority to regulate LDTs continues to be challenged and the regulatory situation remains fluid. The FDA has indicated that it will continue dialogue with the industry, and the timeline and process for finalizing the draft guidance documents is unknown. We will continue to monitor changes to all domestic and international LDT regulatory policy so as to ensure compliance with the current regulatory scheme.
Impact of Regulation
The FDA in the course of enforcing the FDCA may subject a company to various sanctions for violating FDA regulations or provisions of the FDCA, including requiring recalls, issuing Warning Letters, seeking to impose civil money penalties, seizing devices that the agency believes are non-compliant, seeking to enjoin distribution of a specific drug or device seeking to revoke a clearance or approval, seeking disgorgement of profits and seeking to criminally prosecute a company and its officers and other responsible parties.
The levels of revenues and profitability of biopharmaceutical companies may be affected by the continuing efforts of government and third party payors to contain or reduce the costs of health care through various means. For example, in certain foreign markets, pricing or profitability of therapeutic and other pharmaceutical products is subject to governmental control. In the U.S., there have been, and we expect that there will continue to be, a number of federal and state proposals to implement similar governmental control. In addition, in the U.S. and elsewhere, sales of therapeutic and other pharmaceutical products are dependent in part on the availability and adequacy of reimbursement from third party payors, such as the government or private insurance plans. Third party payors are increasingly challenging established prices, and new products that are more expensive than existing treatments may have difficulty finding ready acceptance unless there is a clear therapeutic benefit. On April 1,
22
2014, the Protecting Access to Medicare Act of 2014 (“PAMA”) was enacted into law. Under PAMA, Medicare payment for clinical diagnostic laboratory tests are established by calculating a weighted mean of private payor rates with new rates. Effective January 1, 2018, clinical laboratory fee schedule rates will be based on weighted median private payor rates as required by PAMA. We cannot assure you that any of our products will be considered cost effective, or that reimbursement will be available or sufficient to allow us to sell them competitively and profitably.
State and Federal Security and Privacy Regulations
The privacy and security regulations under the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 ( the “HITECH Act”, and collectively, “HIPAA”), establish comprehensive federal standards with respect to the uses and disclosures of protected health information, or PHI, by health plans and health care providers, in addition to setting standards to protect the confidentiality, integrity and availability of electronic PHI. The regulations establish a complex regulatory framework on a variety of subjects, including:
• |
the circumstances under which uses and disclosures of PHI are permitted or required without a specific authorization by the patient, including but not limited to treatment purposes, to obtain payments for services and health care operations activities; |
• |
a patient’s rights to access, amend and receive an accounting of certain disclosures of PHI; |
• |
the content of notices of privacy practices for PHI; and |
• |
administrative, technical and physical safeguards required of entities that use or receive PHI electronically. |
The final omnibus rule implementing the HITECH Act took effect on March 26, 2013. The rule is broad in scope, but certain provisions are particularly significant in light of our business operations. For example, the final “omnibus” rule implementing the HITECH Act:
• |
Makes clear that situations involving impermissible access, acquisition, use or disclosure of protected health information are now presumed to be a breach unless the covered entity or business associate is able to demonstrate that there is a low probability that the information has been compromised; |
• |
Defines the term “business associate” to include subcontractors and agents that receive, create, maintain or transmit protected health information on behalf of the business associate; |
• |
Establishes new parameters for covered entities and business associates on uses and disclosures of PHI for fundraising and marketing; and |
• |
Establishes clear restrictions on the sale of PHI without patient authorization. |
As a provider of clinical laboratory services and as we launch commercial diagnostic tests, we must continue to implement policies and procedures related to compliance with the HIPAA privacy and security regulations, as required by law. The privacy and security regulations provide for significant fines and other penalties for wrongful use or disclosure of PHI, including potential civil and criminal fines and penalties.
Additionally, as we operate in Europe, we may be subject to laws governing the collection, use, disclosure and transmission of personal and/or patient information. In December 2015, the European Union approved a General Data Protection Regulation (“GDPR”) to replace the current data protection directive, Directive 95/46/EC, which took effect May 25, 2018. The GDPR governs the use and transfer of personal data and imposes enhanced penalties for noncompliance. We have made, and will continue to make, certain adjustments to our operations so as to comply with the GDPR.
Anti-Kickback Laws, Physician Self-Referral Laws, False Claims Act, Civil Monetary Penalties
We are also subject to various federal, state, and international laws pertaining to health care “fraud and abuse,” including anti-kickback laws and false claims laws. The federal Anti-Kickback Statute prohibits anyone from knowingly and willfully soliciting, receiving, offering, or paying any remuneration with the intent to refer, or to arrange for the referral or order of, services or items payable under a federal health care program, including the purchase or prescription of a particular drug or the use of a service or device. Recognizing that the Anti-Kickback Statute is broad and may technically prohibit many innocuous or beneficial arrangements, Congress authorized the U.S. Department of Health and Human Services Office of Inspector General, or OIG, to issue a series of regulations, known as “safe harbors.” These safe harbors set forth requirements that, if met in their entirety, will assure health care providers and other parties that they will not be prosecuted under the Anti-Kickback Statute. The failure of a transaction or arrangement to fit precisely within one or more safe harbors does not necessarily mean that it is illegal, or that prosecution will be pursued. However, conduct and business arrangements that do not fully satisfy each applicable safe harbor may result in increased scrutiny by government enforcement authorities, such as the OIG.
23
Violations of the Anti-Kickback Statute are punishable by the imposition of criminal fines, civil money penalties, treble damages, and/or exclusion from participation in federal health care programs. Many states have also enacted similar anti-kickback laws. The Anti-Kickback Statute and similar state laws and regulations are expansive. If the government were to allege against or convict us of violating these laws, there could be a material adverse effect on our business, results of operations, financial condition, and our stock price. Even an unsuccessful challenge could cause adverse publicity and be costly to respond to, which could have a materially adverse effect on our business, results of operations and financial condition. We will consult counsel concerning the potential application of these and other laws to our business and our sales, marketing and other activities and will make good faith efforts to comply with them. However, given the broad reach of federal and state anti-kickback laws and the increasing attention given by law enforcement authorities, we are unable to predict whether any of our activities will be challenged or deemed to violate these laws.
We are also subject to the physician self-referral laws, commonly referred to as the Stark law, which is a strict liability statute that generally prohibits physicians from referring Medicare patients to providers of “designated health services,” including clinical laboratories, with whom the physician or the physician’s immediate family member has an ownership interest or compensation arrangement, unless an applicable exception applies. Moreover, many states have adopted or are considering adopting similar laws, some of which extend beyond the scope of the Stark law to prohibit the payment or receipt of remuneration for the prohibited referral of patients for designated healthcare services and physician self-referrals, regardless of the source of the payment for the patient’s care. If it is determined that certain of our practices or operations violate the Stark law or similar statutes, we could become subject to civil and criminal penalties, including exclusion from the Medicare programs and loss of government reimbursement. The imposition of any such penalties could harm our business.
Another development affecting the health care industry is the increased use of the federal civil False Claims Act and, in particular, actions brought pursuant to the False Claims Act’s “whistleblower” or “qui tam” provisions. The False Claims Act, as amended by the Fraud Enforcement and Recovery Act of 2009 and the Patient Protection and Affordable Care Act of 2010 (“Affordable Care Act”), imposes liability on any person or entity who, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal health care program. We submit claims for services performed at our laboratories. The qui tam provisions of the False Claims Act allow a private individual to bring actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In recent years, the number of suits brought by private individuals has increased dramatically. In addition, various states have enacted false claim laws analogous to the False Claims Act. Many of these state laws apply where a claim is submitted to any third-party payor and not merely a federal health care program. When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties for each separate false claim. There are many potential bases for liability under the False Claims Act. Liability arises, primarily, when an entity knowingly submits, or causes another to submit, a false claim for reimbursement to the federal government. The False Claims Act has been used to assert liability on the basis of inadequate care, kickbacks and other improper referrals, improper use of Medicare numbers when detailing the provider of services, and allegations as to misrepresentations with respect to the services rendered. Our activities relating to the sale and marketing of our products may be subject to scrutiny under these laws. We are unable to predict whether we would be subject to actions under the False Claims Act or a similar state law, or the impact of such actions. However, the costs of defending such claims, as well as any sanctions imposed, could significantly adversely affect our financial performance.
Further, the beneficiary inducement prohibition of the federal Civil Monetary Penalty Law prohibits any entity from offering or transferring to a Medicare or Medicaid beneficiary any remuneration that the entity knows or should know is likely to influence the beneficiary’s selection of a particular provider, practitioner or supplier of Medicare or Medicaid payable items or services, including waivers of copayments and deductible amounts (or any part thereof) and transfers of items or services for free or for other than fair market value. On December 7, 2016, the OIG released amendments to the CMP. Some of the amendments may impact our business, such as allowing certain remuneration to financially needy individuals. Entities found in violation may be liable for civil monetary penalties of up to $10,000 for each wrongful act. Although we believe that our sales and marketing practices are in material compliance with all applicable federal and state laws and regulations, relevant regulatory authorities may disagree and violation of these laws, or, our exclusion from such programs as Medicaid and other governmental programs as a result of a violation of such laws, could have a material adverse effect on our business, results of operations, financial condition and cash flows.
Open Payments Program
With the launch of Rayaldee, part of our business is now subject to the federal Physician Payments Sunshine Act under the Affordable Care Act, and its implementing regulations, which is implemented though the physicians Open Payments Program (the “Open Payments Program”). The Open Payments Program requires manufacturers of drugs, devices, biological and medical supplies covered by Medicare, Medicaid or the Children’s Health Insurance Program, to report information related to certain payments or other transfers of value made or distributed to physicians and teaching hospitals, or to entities or
24
individuals at the request of, or designated on behalf of, the physicians and teaching hospitals. Manufacturers must also report, on an annual basis, certain ownership and investment interests held by physicians and their immediate family members and payments or other “transfers of value” made to such physician owners. A failure to report each payment, other transfer of value, or ownership/investment interest in a timely, accurate, and complete manner may result in civil monetary penalties of up to $150,000 annually. Further, the “knowing” failure to report each payment, other transfer of value, or ownership/investment interest may result in a one million dollar annual penalty. Several other states and a number of countries worldwide have adopted or are considering the adoption of similar transparency laws. Any failure by us to implement proper procedures to track and report on a timely basis transfers of value to physicians and teaching hospitals could result in substantial penalties.
Foreign Corrupt Practices Act
We are also subject to the U.S. Foreign Corrupt Practices Act (“FCPA”), which prohibits corporations and individuals from paying, offering to pay, or authorizing the payment of anything of value to any foreign government official, government staff member, political party, or political candidate in an attempt to obtain or retain business or to otherwise influence a person working in an official capacity. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect their transactions and to devise and maintain an adequate system of internal accounting controls. Our international activities create the risk of unauthorized payments or offers of payments by our employees, consultants, sales agents or distributors, even though they may not always be subject to our control. We discourage these practices by our employees and agents. However, our existing safeguards and any future improvements may prove to be less than effective, and our employees, consultants, sales agents or distributors may engage in conduct for which we might be held responsible. Any failure by us to adopt appropriate compliance procedures and ensure that our employees and agents comply with the FCPA and applicable laws and regulations in foreign jurisdictions could result in substantial penalties or restrictions on our ability to conduct business in certain foreign jurisdictions.
MANUFACTURING AND QUALITY
Other than our facilities in Waterford, Ireland, Guadalajara, Mexico, Nesher, Israel, and Banyoles, Spain, we currently have no pharmaceutical manufacturing facilities. We have entered into agreements with various third parties for the formulation and manufacture of our pharmaceutical clinical supplies. These suppliers and their manufacturing facilities must comply with FDA regulations, current good laboratory practices (“cGLPs”) and current good manufacturing practices (“cGMPs”). We plan to continue to outsource the manufacturing and formulation of our clinical supplies.
The FDA and similar regulatory bodies may inspect our facilities and the facilities of those who manufacture on our behalf worldwide. If the FDA or similar regulatory bodies inspecting our facilities or the facilities of our suppliers find regulatory violations in manufacturing and quality control practices or procedures they may require us to cease partial or complete manufacturing operations until the violations are corrected. They may also impose restrictions on distribution of specific products until the violations are corrected.
Our point-of-care diagnostic system consists of a disposable test cassette and an analyzer. We prepare all necessary test reagents and assemble and package the disposable cassettes at our facility in Woburn, Massachusetts. We rely on third parties for the manufacture of the analyzer.
We are committed to providing high quality products to our customers, and we plan to meet this commitment by working diligently to continue implementing updated and improved quality systems and concepts throughout our organization.
SALES & MARKETING
Our diagnostics business includes BioReference’s almost 300-person sales and marketing team in the U.S. to drive growth and leverage new products, including the 4Kscore prostate cancer test. We have a highly specialized, field based 79-person sales and marketing team in the U.S. dedicated to the launch and commercialization of Rayaldee. We also have limited sales and marketing personnel in Ireland, Chile, Spain, Mexico and Israel.
EMPLOYEES
As of December 31, 2018, we had 5,690 full-time employees worldwide. None of our employees are represented by a collective bargaining agreement.
Code of Ethics
We have adopted a Code of Business Conduct and Ethics. We require all employees, including our principal executive officer and principal accounting officer and other senior officers and our employee directors, to read and to adhere to the Code of Business Conduct and Ethics in discharging their work-related responsibilities. Employees are required to report any
25
conduct that they believe in good faith to be an actual or apparent violation of the Code of Business Conduct and Ethics. The Code of Business Conduct and Ethics is available on our website at http://www.OPKO.com.
Available Information
We are required to file annual, quarterly and current reports, proxy statements and other information with the SEC. Information that we file with the SEC is available at the SEC’s web-site at www.sec.gov. We also make available free of charge on or through our web site, at www.opko.com, our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and all amendments to those reports as soon as reasonably practicable after such material is electronically filed with the SEC. The information on our website is not, and shall not be deemed to be, a part hereof or incorporated into this or any of our other filings with the SEC.
ITEM 1A. RISK FACTORS.
You should carefully consider the risks described below, as well as other information contained in this report, including the consolidated financial statements and the notes thereto and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” The occurrence of any of the events discussed below could significantly and adversely affect our business, prospects, results of operations, financial condition, and cash flows.
RISKS RELATED TO OUR BUSINESS
We have a history of operating losses and may not become profitable in the near future.
We are not profitable and have incurred losses since our inception. We may not generate substantial revenue from the sale of proprietary pharmaceutical products or certain of our diagnostic products for some time and we have generated only limited revenue from our pharmaceutical operations in the U. S., Chile, Mexico, Israel, Spain, and Ireland, and from sale of the 4Kscore test. We may not successfully leverage the national marketing, sales and distribution resources of BioReference to enhance sales of, and reimbursement for, our 4Kscore test and our other diagnostic products under development, which would adversely impact our ability to generate substantial revenue from the sale of these products for some time. Rayaldee is our only pharmaceutical product that has been approved for marketing, other than those products sold by our Chilean, Mexican, Israeli, Spanish, and Irish subsidiaries. We continue to incur substantial research and development and general and administrative expenses related to our operations and, to date, we have devoted most of our financial resources to research and development, including our pre-clinical development activities and clinical trials. We may incur losses from our operations for the foreseeable future and these losses could increase as we continue our research activities and conduct development of, and seek regulatory approvals and clearances for, our product candidates, and prepare for and begin to commercialize any approved or cleared products, particularly if we are unable to generate profits and cash flow from BioReference and our other commercial businesses. If we are unable to generate profits and cash flow from BioReference and our other commercial businesses, our product candidates fail in clinical trials or do not gain regulatory approval or clearance, or if our approved products and product candidates do not achieve market acceptance, we may never become profitable. In particular, if we are unable to successfully commercialize Rayaldee, we may never generate substantial revenues from Rayaldee or achieve profitability. In addition, if we are required by the U.S. Food and Drug Administration (“FDA”), to perform studies in addition to those we currently anticipate, our expenses will increase beyond current expectations and the timing of any potential product approval may be delayed. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods.
We will continue to require additional funding, which may not be available to us on acceptable terms, or at all.
As of December 31, 2018, we have cash and cash equivalents of $96.5 million. We have not generated sustained positive cash flows sufficient to offset our operating and research and development expenses and our primary source of cash has been from the public and private placement of stock, the issuance of the 2033 Senior Notes, 2023 Convertible Notes and the 2025 Convertible Notes (each as defined below) and credit facilities available to us. On November 8, 2018, we entered into stock purchase agreements with certain investors pursuant to which we agreed to sell to such investors in private placements (the “Private Placements”) an aggregate of approximately 26.5 million shares of our common stock at a purchase price of $3.49 per share, which was the closing bid price of our common stock on the Nasdaq Global Select Market on such date, for an aggregate purchase price of $92.5 million. On February 7, 2019, we issued $200 million aggregate principal amount of its 4.50% Convertible Senior Notes due 2025 (the “2025 Convertible Notes”) in a registered public offering under the Securities Act. In addition, we granted the underwriter in the offering a 30-day option to purchase up to an additional $30 million aggregate principal amount of the 2025 Convertible Notes to cover over-allotments, if any.
We believe that the cash and cash equivalents on hand or available to us from operations or through our lines of credit, together with the proceeds of the 2025 Convertible Notes offering, are sufficient to meet our anticipated cash requirements for operations and debt service beyond the next 12 months. We have based this estimate on assumptions that may prove to be
26
wrong or subject to change, and we may be required to use our available capital resources sooner than we currently expect or curtail aspects of our operations in order to preserve our capital. Because of the numerous risks and uncertainties associated with the development and commercialization of our products and product candidates, the success of our relationships with Pfizer, VFMCRP and JT and the success of our integration of BioReference and other acquisitions, we are unable to estimate the amounts of increased capital outlays and operating expenditures associated with our current and anticipated clinical trials and our expanded commercial operations. Our future capital requirements will depend on a number of factors, including the successful commercialization of Rayaldee, our continued relationships with Pfizer, VFMCRP, and JT, cash flow generated by BioReference and costs associated with the integration of the BioReference and other acquisitions, the continued progress of our research and development of product candidates, the timing and outcome of clinical trials and regulatory approvals, the costs involved in preparing, filing, prosecuting, maintaining, defending, and enforcing patent claims and other intellectual property rights, the status of competitive products, the availability of financing, and our success in developing markets for our products and product candidates.
Until we can generate a sufficient amount of product and service revenue to finance our cash requirements for research, development and operations, we will need to finance future cash needs primarily through public or private equity offerings, debt financings, or strategic collaborations. Our ability to obtain additional capital may depend on prevailing economic conditions and financial, business and other factors beyond our control. Disruptions in the U.S. and global financial markets may adversely impact the availability and cost of credit, as well as our ability to raise money in the capital markets. Economic conditions have been, and continue to be, volatile. Continued instability in these market conditions may limit our ability to replace, in a timely manner, maturing liabilities and access the capital necessary to fund and grow our business. Additionally, our continuing operating losses and the recent lawsuits involving us and our Chief Executive Officer (“CEO”) and Chairman of our Board of Directors (“Chairman”) by the SEC and other parties increase the difficulty in obtaining additional capital.
There can be no assurance that additional capital will be available to us on acceptable terms, or at all, which could adversely impact our business, results of operations, liquidity, capital resources and financial condition. If we are not able to secure additional funding when needed, we may have to delay, reduce the scope of, or eliminate one or more of our clinical trials or research and development programs or cease operations altogether. To the extent that we raise additional funds by issuing equity securities, our stockholders may experience additional significant dilution, and debt financing, if available, may involve restrictive covenants and other onerous terms. To the extent that we raise additional funds through collaboration and licensing arrangements, it may be necessary to relinquish some rights to our technologies or our products and product candidates or grant licenses on terms that may not be favorable to us.
Our research and development activities may not result in commercially viable products.
Many of our product candidates are in the early stages of development and are prone to the risks of failure inherent in drug, diagnostic, and medical device product development. These risks further include the possibility that such products would:
• |
be found to be ineffective, unreliable, or otherwise inadequate or otherwise fail to receive regulatory approval; |
• |
be difficult or impossible to manufacture on a commercial scale; |
• |
be uneconomical to market or otherwise not be effectively marketed; |
• |
fail to be successfully commercialized if adequate reimbursement from government health administration authorities, private health insurers, and other organizations for the costs of these products is unavailable; |
• |
be impossible to commercialize because they infringe on the proprietary rights of others or compete with products marketed by others that are superior; or |
• |
fail to be commercialized prior to the successful marketing of similar products by competitors. |
The results of pre-clinical trials and previous clinical trials for our products may not be predictive of future results, and our current and planned clinical trials may not satisfy the requirements of the FDA or other non-U.S. regulatory authorities.
Positive results from pre-clinical studies and early clinical trial experience should not be relied upon as evidence that later-stage or large-scale clinical trials will succeed. Likewise, there can be no assurance that the results of studies conducted by collaborators or other third parties will be viewed favorably or are indicative of our own future study results. We may be required to demonstrate with substantial evidence through well-controlled clinical trials that our product candidates are either (i) with respect to drugs or Class III devices, safe and effective for use in a diverse population of their intended uses or (ii) with respect to Class I or Class II devices, are substantially equivalent in terms of safety and effectiveness to devices that are already marketed under section 510(k) of the Food, Drug and Cosmetic Act. Success in early clinical trials does not mean that future clinical trials will be successful because product candidates in later-stage clinical trials may fail to demonstrate sufficient safety
27
and efficacy to the satisfaction of the FDA and other non-U.S. regulatory authorities despite having progressed through initial clinical trials.
Further, our drug candidates may not be approved or cleared even if they achieve their primary endpoints in phase 3 clinical trials or registration trials. In addition, our diagnostic test candidates may not be approved or cleared, as the case may be, even though clinical or other data are, in our view, adequate to support an approval or clearance. The FDA or other non-regulatory authorities may disagree with our trial design and our interpretation of data from pre-clinical studies and clinical trials. In addition, any of these regulatory authorities may change requirements for the approval or clearance of a product candidate even after reviewing and providing comment on a protocol for a pivotal clinical trial that has the potential to result in FDA and other non-U.S. regulatory authorities’ approval. Any of these regulatory authorities may also approve or clear a product candidate for fewer or more limited indications or uses than we request or may grant approval or clearance contingent on the performance of costly post-marketing clinical trials. The FDA or other non-U.S. regulatory authorities may not approve the labeling claims necessary or desirable for the successful commercialization of our product candidates.
The results of our clinical trials may show that our product candidates may cause undesirable side effects, which could interrupt, delay or halt clinical trials, resulting in the denial of regulatory approval by the FDA and other non-U.S. regulatory authorities.
Safety concerns with drug products over the years have resulted in the withdrawal of drug products, revisions to drug labeling that further limit use of the drug products, and establishment of risk management programs that may, for instance, restrict distribution of drug products. Attention to drug safety issues may result in a more cautious approach by the FDA to clinical trials. Data from clinical trials may receive greater scrutiny with respect to safety, which may make the FDA or other regulatory authorities more likely to terminate clinical trials before completion, or require longer or additional clinical trials that may result in substantial additional expense and a delay or failure in obtaining approval or approval for a more limited indication than originally sought.
The failure to successfully commercialize Rayaldee would have a material adverse effect on our business.
In June 2016, the FDA approved the Company’s New Drug Application for Rayaldee (calcifediol) extended release capsules for the treatment of secondary hyperparathyroidism (SHPT) in adults with stage 3 or 4 chronic kidney disease (CKD) and serum total 25-hydroxyvitamin D levels less than 30 ng/mL. The commercial launch for Rayaldee began in November 2016. Rayaldee is our only pharmaceutical product approved for marketing in the U.S. and our ability to generate revenue from product sales and achieve profitability is substantially dependent on our ability to effectively commercialize Rayaldee. Our failure to successfully commercialize Rayaldee would have a material adverse effect on our business, financial condition, cash flows and results of operations.
Additionally, the market perception and reputation of Rayaldee and its safety and efficacy are important to our business and the continued acceptance of our product candidates and products. Any negative publicity about Rayaldee, such as the discovery of safety issues, adverse events, or even public rumors about such events, could have a material adverse effect on our business. Levels of market acceptance for Rayaldee could be impacted by several factors, some of which are not within our control, including but not limited to the:
• |
safety, efficacy, convenience and cost-effectiveness of our product compared to products of our competitors; |
• |
scope of approved uses and marketing approvals; |
• |
availability of patent or regulatory exclusivity; |
• |
timing of market approvals and market entries; |
• |
ongoing regulatory obligations following approval; |
• |
any restrictions or “black box” warnings required on the product labeling: |
• |
availability of alternative products from our competitors; |
• |
acceptance of the price of our product; |
• |
effectiveness of our sales force and promotional efforts; |
• |
the level of reimbursement of our product; |
• |
acceptance of our product on government and private formularies; |
• |
ability to market our product effectively at the retail level or in the appropriate setting of care; and |
28
• |
the reputation of our product. |
If Rayaldee fails to gain, or loses, market acceptance, our revenues would be adversely impacted and we may be required to take material impairment charges, all of which could have a material adverse effect on our business, financial condition, cash flows and results of operations.
We rely on licensing agreements with Vifor Fresenius Medical Renal Care Pharma Ltd (“VFMCRP”) and Japan Tobacco (“JT”) for the international development and marketing of Rayaldee. Failure to maintain these license agreements could prevent us from successfully developing and commercializing Rayaldee worldwide.
In May 2016, EirGen, our wholly-owned subsidiary, partnered with VFMCRP through a Development and License Agreement for the development and marketing of Rayaldee in Europe, Canada, Mexico, Australia, South Korea and certain other international markets. The license to VFMCRP potentially covers all therapeutic and prophylactic uses of the product in human patients, provided that initially the license is for the use of the product for the treatment or prevention of secondary hyperparathyroidism related to patients with stage 3 or 4 chronic kidney disease and vitamin D insufficiency/deficiency. We received a non-refundable and non-creditable upfront payment of $50 million and a $2.0 million payment triggered by the approval of Rayaldee in Canada for the treatment of SHPT in adults with stage 3 or 4 CKD and vitamin D insufficiency. EirGen is also eligible to receive up to an additional $35 million in regulatory milestones and $195 million in launch and sales-based milestones. In addition, we are eligible to receive tiered, double digit royalty payments or a minimum royalty, whichever is greater, upon commencement of sales of the product. The success of the Development and License Agreement with VFMCRP is dependent in part on, among other things, the skills, experience and efforts of VFMCRP’s employees responsible for the project, VFMCRP’s commitment to the arrangement, and the financial condition of VFMCRP, all of which are beyond our control. In the event that VFMCRP, for any reason, including but not limited to early termination of the agreement, fails to devote sufficient resources to successfully develop and market Rayaldee internationally, our ability to earn milestone payments or receive royalty payments would be adversely affected, which would have a material adverse effect on our financial condition and prospects.
In October 2017, we entered into a Development and License Agreement (the “JT Agreement”) with JT under which JT was granted the exclusive rights for the development and commercialization of Rayaldee in Japan. The license grant to JT covers the therapeutic and preventative use of the product for (i) SHPT in non-dialysis and dialysis patients with CKD, (ii) rickets, and (iii) osteomalacia, as well as such additional indications as may be added to the scope of the license subject to the terms of the JT Agreement. Under the terms of the JT Agreement, we received an initial upfront payment of $6 million and received another $6 million upon the initiation of our phase 2 study for Rayaldee in dialysis patients in the U.S. We are also eligible to receive up to an additional aggregate amount of $31 million upon the achievement of certain regulatory and development milestones by JT for Rayaldee in Japan, and $75 million upon the achievement of certain sales based milestones by JT. We will also receive tiered, double digit royalty payments at rates ranging from low double digits to mid-teens on net sales within Japan. JT will, at its sole cost and expense, be responsible for performing all development activities necessary to obtain all regulatory approvals for Rayaldee in Japan and for all commercial activities pertaining to Rayaldee in Japan, except for certain preclinical expenses which we have agreed to reimburse JT up to a capped amount. If JT, for any reason, including but not limited to early termination of the JT Agreement, fails to devote sufficient resources to successfully develop and market Rayaldee in Japan, our ability to earn milestone payments or receive royalty payments would be adversely affected, which could have a material adverse effect on our financial condition and prospects.
We currently have a seventy-nine person specialized sales and marketing team for Rayaldee in the U.S. If we are unable to develop or maintain a strong sales, marketing and distribution capability on our own or through collaborations with marketing partners, we will not be successful in commercializing Rayaldee or our other pharmaceutical products or product candidates in the U.S.
Other than our 79-person specialized sales and marketing team dedicated to Rayaldee, we currently have no pharmaceutical marketing, sales or distribution capabilities in the U.S. Any failure or inability to maintain adequate sales, marketing and distribution capabilities would adversely impact the commercialization of Rayaldee or our other pharmaceutical products or candidates. If we are not successful in commercializing our existing and future pharmaceutical products and product candidates, either on our own or through collaborations with one or more third parties, our product revenue will suffer and we may incur significant additional losses.
29
Our exclusive worldwide agreement with Pfizer Inc. is important to our business. If we do not successfully develop hGH- CTP and/or Pfizer Inc. does not successfully commercialize hGH-CTP, our business could be adversely affected.
In December 2014, we entered into a development and commercialization agreement with Pfizer relating to our long-acting hGH- CTP for the treatment of growth hormone deficiency in adults and children (the “Pfizer Agreement”). Under the Pfizer Agreement, we received non-refundable and non-creditable upfront payments of $295 million and are eligible to receive up to an additional $275 million upon the achievement of certain regulatory milestones. In addition, we are eligible to receive initial royalty payments associated with the commercialization of hGH-CTP for Adult GHD. Upon the launch of hGH-CTP for Pediatric GHD, the royalties will transition to a regional, tiered gross profit sharing for both hGH-CTP and Pfizer’s Genotropin®. We are responsible for the development program and are obligated to pay for the development up to an agreed cap, which may be exceeded under certain circumstances. We have exceeded development cap and if we are unable to reach an agreement with Pfizer regarding cost sharing for the overruns as well as other obligations, including development obligations, it could have a material adverse impact on the expected benefits to us from the Pfizer transaction and our overall financial condition. In the event that the parties are able to obtain regulatory approvals to market a product covered by the Pfizer Agreement, we will be substantially dependent on Pfizer for the successful commercialization of such product. The success of the collaboration arrangement with Pfizer is dependent in part on, among other things, the skills, experience and efforts of Pfizer’s employees responsible for the project, Pfizer’s commitment to the arrangement, and the financial condition of Pfizer, all of which are beyond our control. The Pfizer Agreement is terminable for any reason by Pfizer upon ninety days written notice to OPKO. In the event that Pfizer, for any reason, including but not limited to early termination of the Pfizer Agreement, fails to devote sufficient resources to successfully develop and commercialize any product resulting from the collaboration arrangement, our ability to earn milestone payments or receive royalty or profit sharing payments would be adversely affected, which would have a material adverse effect on our financial condition and prospects and the trading prices of our securities.
Our business is substantially dependent on the success of clinical trials for hGH-CTP and our ability to achieve regulatory approval for the marketing of this product.
There is no assurance that clinical trials for hGH-CTP will be successful or support marketing approval, or that we will be able to obtain marketing approval for the product, or any other product candidate we are developing. Before they can be marketed, our products in development must be approved by the FDA or similar foreign governmental agencies. The process for obtaining FDA approval is both time-consuming and costly, with no certainty of a successful outcome. Before obtaining regulatory approval for the sale of any drug candidate, we must conduct extensive preclinical tests and clinical trials to demonstrate the safety and efficacy in humans of our product candidates. Although the safety profile for hGH-CTP has been consistent with that observed with those treated with daily growth hormone, further testing or patient use may undermine those determinations or unexpected side effects may arise. A failure of any preclinical study or clinical trial can occur at any stage of testing. The results of preclinical and initial clinical testing of these products may not necessarily indicate the results that will be obtained from later or more extensive testing. It also is possible to suffer significant setbacks in advanced clinical trials, even after obtaining promising results in earlier trials. In December 2016, we announced preliminary topline data from our phase 3, double blind, placebo controlled study of hGH-CTP in adults with GHD. Although there was no statistically significant difference between hGH-CTP and placebo on the primary endpoint of change in trunk fat mass from baseline to 26 weeks, after unblinding the study, we identified an exceptional value of trunk fat mass reduction in the placebo group that may have affected the primary outcome. We completed post-hoc sensitivity analyses to evaluate the influence of outliers on the primary endpoint results using multiple statistical approaches. Analyses that excluded outliers showed a statistically significant difference between hGH-CTP and placebo on the change in trunk fat mass. Additional analyses that did not exclude outliers showed mixed results. There can be no assurance that a BLA will be submitted or that the FDA will consider the sensitivity analysis or consider the product for approval for adults with GHD. If phase 3 clinical trials for hGH-CTP are not successful or we are unable to achieve regulatory approval for this product, our business will be significantly adversely impacted, which could have a materially adverse effect on our business, financial condition and results of operations.
Our business is substantially dependent on our ability to develop, launch and generate revenue from our diagnostic products.
Our business is dependent on our ability to successfully commercialize the 4Kscore test and other diagnostic products, including the Claros 1. We are committing significant resources to the development and commercialization of these products, and there is no guarantee that we will be able to successfully commercialize these tests. We have limited experience in developing, manufacturing, selling, marketing and distributing diagnostic tests. If we are not able to successfully develop, market or sell diagnostic tests we develop for any reason, including the failure to obtain any required regulatory approvals, or obtain reimbursement, we will not generate any meaningful revenue from the sale of such tests. Even if we are able to develop effective diagnostic tests for sale in the marketplace, a number of factors could impact our ability to sell such tests or generate any significant revenue from the sale of such tests, including without limitation:
30
▪ |
our ability to establish and maintain adequate infrastructure to support the commercial launch and sale of our diagnostic tests, including establishing adequate laboratory space, information technology infrastructure, sample collection and tracking systems, electronic ordering and reporting systems and other infrastructure and hiring adequate laboratory and other personnel; |
▪ |
the success of the validation studies for our diagnostic tests under development and our ability to publish study results in peer-reviewed journals; |
▪ |
the availability of alternative and competing tests or products and technological innovations or other advances in medicine that cause our technologies to be less competitive; |
▪ |
the accuracy rates of such tests, including rates of false-negatives and/or false-positives; |
▪ |
concerns regarding the safety or effectiveness or clinical utility of our diagnostic tests; |
▪ |
changes in the regulatory environment affecting health care and health care providers, including changes in laws regulating laboratory testing and/or device manufacturers; |
▪ |
the extent and success of our sales and marketing efforts and ability to drive adoption of our diagnostic tests; |
▪ |
coverage and reimbursement levels by government payors and private insurers; |
▪ |
pricing pressures and changes in third-party payor reimbursement policies; and |
▪ |
intellectual property rights held by others or others infringing our intellectual property rights. |
Our business is substantially dependent on our ability to generate profits and cash flow from our laboratory operations.
We have made a significant investment in our laboratory operations through the acquisition of BioReference. We compete in the clinical laboratory market primarily on the basis of the quality of testing, reporting and information systems, reputation in the medical community, the pricing of services and ability to employ qualified personnel. Our failure to successfully compete on any of these factors could result in the loss of clients and a reduction in our revenues and profits. To offset efforts by payors to reduce the cost and utilization of clinical laboratory services, we will need to obtain and retain new clients and business partners and grow the laboratory operations. A reduction in tests ordered, specimens submitted by existing clients, or payment rates, without offsetting growth in our client base, could impact our ability to successfully grow our business and could have a material adverse impact on our ability to generate profits and cash flow from the laboratory operations.
Discontinuation or recalls of existing testing products, failure to develop, or acquire, licenses for new or improved testing technologies or our clients using new technologies to perform their own tests could adversely affect our business.
From time to time, manufacturers discontinue or recall reagents, test kits or instruments used by us to perform laboratory testing. Such discontinuations or recalls could adversely affect our costs, testing volume and revenue.
The clinical laboratory industry is subject to changing technology and new product introductions. Our success in maintaining a leadership position in genomic and other advanced testing technologies will depend, in part, on our ability to develop, acquire or license new and improved technologies on favorable terms and to obtain appropriate coverage and reimbursement for these technologies. We may not be able to negotiate acceptable licensing arrangements and it cannot be certain that such arrangements will yield commercially successful diagnostic tests. If we are unable to license these testing methods at competitive rates, our research and development costs may increase as a result. In addition, if we are unable to license or develop new or improved technologies to expand our esoteric testing operations, our testing methods may become outdated when compared with our competition and testing volume and revenue may be materially and adversely affected.
Currently, most clinical laboratory testing is categorized as “high” or “moderate” complexity, and thereby is subject to extensive and costly regulation under CLIA. The cost of compliance with CLIA makes it impractical for most physicians to operate clinical laboratories in their offices, and other laws limit the ability of physicians to have ownership in a laboratory and to refer tests to such a laboratory. Manufacturers of laboratory equipment and test kits could seek to increase their sales by marketing point-of-care laboratory equipment to physicians and by selling test kits approved for home or physician office use to both physicians and patients. Diagnostic tests approved for home use are automatically deemed to be “waived” tests under CLIA and may be performed in physician office laboratories as well as by patients in their homes with minimal regulatory oversight. Other tests meeting certain FDA criteria also may be classified as “waived” for CLIA purposes. The FDA has regulatory responsibility over instruments, test kits, reagents and other devices used by clinical laboratories and has taken responsibility from the Centers for Disease Control for classifying the complexity of tests for CLIA purposes. Increased approval of “waived” test kits could lead to increased testing by physicians in their offices or by patients at home, which could affect our market for laboratory testing services and negatively impact our revenues. If our competitors develop and market
31
products that are more effective, safer or less expensive than our products and product candidates, our net revenues, profitability and commercial opportunities will be negatively impacted.
If our competitors develop and market products or services that are more effective, safer or less expensive than our current and future products or services, our revenues, profitability and commercial opportunities will be negatively impacted.
The pharmaceutical, diagnostic, and laboratory testing industries are highly competitive and require an ongoing, extensive search for technological innovation. The industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. They also require, among other things, the ability to effectively discover, develop, test and obtain regulatory approvals for products, as well as the ability to effectively commercialize, market and promote approved products.
Numerous companies, including major pharmaceutical companies, specialty pharmaceutical companies and specialized biotechnology companies, are engaged in the development, manufacture and marketing of pharmaceutical products competitive with those that we intend to commercialize ourselves and through our partners. Competitors to our diagnostics business include major diagnostic companies, reference laboratories, molecular diagnostic firms, universities and research institutions. Most of these companies have substantially greater financial and other resources, larger research and development staffs and more extensive marketing and manufacturing organizations than ours. This enables them, among other things, to make greater research and development investments and efficiently utilize their research and development costs, as well as their marketing and promotion costs, over a broader revenue base. This also provides our competitors with a competitive advantage in connection with the highly competitive product acquisition and product in-licensing process, which may include auctions in which the highest bidder wins. Our competitors may also have more experience and expertise in obtaining marketing approvals from the FDA and other regulatory authorities. We cannot predict with accuracy the timing or impact of the introduction of potentially competitive products or their possible effect on our sales. In addition to product development, testing, approval, and promotion, other competitive factors in the pharmaceutical and diagnostics industry include industry consolidation, product quality and price, product technology, reputation, customer service, and access to technical information.
In our clinical laboratory operations, we compete with three types of providers in a highly fragmented and competitive industry: hospital laboratories, physician-office laboratories and other independent clinical laboratories. Our major competitors in the New York metropolitan area are two of the largest national laboratories, Quest Diagnostics and Laboratory Corporation of America. We are much smaller than these national laboratories.
The clinical laboratory business is intensely competitive both in terms of price and service. Pricing of laboratory testing services is often one of the most significant factors used by health care providers and third-party payors in selecting a laboratory. As a result of the clinical laboratory industry undergoing significant consolidation, larger clinical laboratory providers are able to increase cost efficiencies afforded by large-scale automated testing. This consolidation results in greater price competition. We may be unable to increase cost efficiencies sufficiently, if at all, and as a result, our net earnings and cash flows could be negatively impacted by such price competition. Additionally, we may also face changes in contracting with third party payors, fee schedules, competitive bidding for laboratory services or other actions or pressures reducing payment schedules as a result of increased or additional competition.
If our competitors market products that are more effective, safer, easier to use or less expensive than our current products and product candidates, or that reach the market sooner than our products and product candidates, we may not achieve commercial success. In addition, the biopharmaceutical, diagnostic, medical device, and laboratory industries are characterized by rapid technological change. Because our research approach integrates many technologies, it may be difficult for us to stay abreast of the rapid changes in each technology. If we fail to stay at the forefront of technological change, we may be unable to compete effectively. Technological advances or products developed by our competitors may render our technologies, products or product candidates obsolete or less competitive.
Our product development activities could be delayed or stopped.
We do not know whether our current or planned pre-clinical and clinical studies will be completed on schedule, or at all. Furthermore, we cannot guarantee that our planned pre-clinical and clinical studies will begin on time or at all. The commencement of our planned clinical trials could be substantially delayed or prevented by several factors, including:
▪ |
a limited number of, and competition for, suitable patients with the particular types of disease required for enrollment in our clinical trials or that otherwise meet the protocol’s inclusion criteria and do not meet any of the exclusion criteria; |
▪ |
a limited number of, and competition for, suitable serum or other samples from patients with particular types of disease required for our validation studies; |
32
▪ |
a limited number of, and competition for, suitable sites to conduct our clinical trials; |
▪ |
delay or failure to obtain FDA or other non-U.S. regulatory authorities’ approval or agreement to commence a clinical trial; |
▪ |
delay or failure to obtain sufficient supplies of the product candidate for our clinical trials; |
▪ |
requirements to provide the drugs, diagnostic tests, or medical devices required in our clinical trial protocols or clinical trials at no cost or cost, which may require significant expenditures that we are unable or unwilling to make; |
▪ |
delay or failure to reach agreement on acceptable clinical trial agreement terms or clinical trial protocols with prospective sites or investigators; |
▪ |
delay or failure to obtain institutional review board (“IRB”) approval to conduct or renew a clinical trial at a prospective site; and |
▪ |
insufficient liquidity to fund our preclinical and clinical studies. |
The completion of our clinical trials could also be substantially delayed or prevented by several factors, including:
▪ |
slower than expected rates of patient recruitment and enrollment; |
▪ |
failure of patients to complete the clinical trial; |
▪ |
unforeseen safety issues; |
▪ |
lack of efficacy evidenced during clinical trials; |
▪ |
termination of our clinical trials by one or more clinical trial sites; |
▪ |
inability or unwillingness of patients or medical investigators to follow our clinical trial protocols; |
▪ |
inability to monitor patients adequately during or after treatment; and |
▪ |
insufficient liquidity to fund ongoing studies. |
Our clinical trials may be suspended or terminated at any time by the FDA, other regulatory authorities, the IRB for any given site, or us. Additionally, changes in regulatory requirements and guidance may occur and we may need to amend clinical trial protocols to reflect these changes with appropriate regulatory authorities. Amendments may require us to resubmit our clinical trial protocols to IRBs for re-examination, which may impact the costs, timing, or successful completion of a clinical trial. Any failure or significant delay in commencing or completing clinical trials for our product candidates could materially harm our results of operations and financial condition, as well as the commercial prospects for our product candidates.
Our approved products or product candidates may have undesirable side effects and cause our products to be taken off the market.
If we or others identify undesirable side effects caused by our products:
• |
regulatory authorities may require the addition of labeling statements, specific warnings, a contraindication, or field alerts to physicians and pharmacies; |
• |
regulatory authorities may withdraw their approval of the product and require us to take our approved product off the market; |
• |
we may be required to change the way the product is administered, conduct additional clinical trials, or change the labeling of the product; |
• |
we may have limitations on how we promote our products; |
• |
sales of products may decrease significantly; |
• |
we may be subject to litigation or product liability claims; and |
• |
our reputation may suffer. |
Any of these events could prevent us from achieving or maintaining market acceptance of the affected product or could substantially increase our commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from its sale.
33
Our inability to meet regulatory quality standards applicable to our manufacturing and quality processes and to address quality control issues in a timely manner could delay the production and sale of our products or result in recalls of products.
Manufacturing or design defects, unanticipated use of our products, or inadequate disclosure of risks relating to the use of our products could lead to injury or other adverse events. These events could lead to recalls or safety alerts relating to our products (either voluntary or required by governmental authorities) and could result, in certain cases, in the removal of a product from the market. Any recall could result in significant costs as well as negative publicity that could reduce demand for our products. Personal injuries relating to the use of our products can also result in product liability claims being brought against us. In some circumstances, such adverse events could also cause delays in new product approvals.
We are committed to providing high quality products to our customers, and we plan to meet this commitment by working diligently to continue implementing updated and improved quality systems and concepts throughout our organization. We cannot assure you that we will not have quality control issues in the future, which may result in warning letters and citations from the FDA. If we receive any warning letters from the FDA in the future, there can be no assurances regarding the length of time or cost it will take us to resolve such quality issues to our satisfaction and to the satisfaction of the FDA. If our remedial actions are not satisfactory to the FDA, we may have to devote additional financial and human resources to our efforts, and the FDA may take further regulatory actions against us including, but not limited to, assessing civil monetary penalties or imposing a consent decree on us, which could result in further regulatory constraints, including the governance of our quality system by a third party. Our inability to resolve these issues or the taking of further regulatory action by the FDA may weaken our competitive position and have a material adverse effect on our business, results of operations and financial condition.
We manufacture pharmaceutical products in Ireland, Mexico, Spain, and Israel. We also prepare necessary test reagents and assemble and package the cassettes for our point-of-care diagnostic system at our facility in Woburn, Massachusetts. Any quality control issues at our facilities may weaken our competitive position and have a material adverse effect on our business results of operations and financial condition.
As a medical device manufacturer, we are required to register with the FDA and are subject to periodic inspection by the FDA for compliance with its Quality System Regulation (“QSR”) requirements, which require manufacturers of medical devices to adhere to certain regulations, including testing, quality control and documentation procedures. Compliance with applicable regulatory requirements is subject to continual review and is monitored rigorously through periodic inspections by the FDA. In addition, most international jurisdictions have adopted regulatory approval and periodic renewal requirements for medical devices, and we must comply with these requirements in order to market our products in these jurisdictions. In the European Community, we are required to maintain certain ISO certifications in order to sell our products and must undergo periodic inspections by notified bodies to obtain and maintain these certifications. Further, some emerging markets rely on the FDA’s Certificate for Foreign Government (“CFG”) in lieu of their own regulatory approval requirements. Our failure, or our manufacturers’ failure to meet QSR, ISO, or any other regulatory requirements or industry standards could delay production of our products and lead to fines, difficulties in obtaining regulatory clearances, recalls or other consequences, which could, in turn, have a material adverse effect on our business, results of operations, and our financial condition.
Failure to establish, and perform to, appropriate quality standards to assure that the highest level of quality is observed in the performance of our testing services could adversely affect the results of our operations and adversely impact our reputation.
The provision of clinical testing services, including anatomic pathology services, and related services, and the design, manufacture and marketing of diagnostic products involve certain inherent risks. The services that we provide and the products that we design, manufacture and market are intended to provide information for healthcare providers in providing patient care. Therefore, users of our services and products may have a greater sensitivity to errors than the users of services or products that are intended for other purposes.
Similarly, negligence in performing our services can lead to injury or other adverse events. We may be sued under physician liability or other liability law for acts or omissions by our pathologists, laboratory personnel and other employees. We are subject to the attendant risk of substantial damages awards and risk to our reputation.
Even after we receive regulatory approval or clearance to market our product candidates, the market may not be receptive to our products.
Our products may not gain market acceptance among physicians, patients, health care payors and/or the medical community. We believe that the degree of market acceptance will depend on a number of factors, including:
• |
timing of market introduction of competitive products; |
• |
safety and efficacy of our product compared to other products; |
34
• |
prevalence and severity of any side effects; |
• |
potential advantages or disadvantages over alternative treatments; |
• |
strength of marketing and distribution support; |
• |
price of our products, both in absolute terms and relative to alternative treatments; |
• |
availability of coverage and reimbursement from government and other third-party payors; |
• |
potential product liability claims; |
• |
limitations or warnings contained in a product’s regulatory authority-approved labeling; and |
• |
changes in the standard of care for the targeted indications for any of our products or product candidates, which could reduce the marketing impact of any claims that we could make following applicable regulatory authority approval. |
In addition, our efforts to educate the medical community and health care payors on the benefits of our products and product candidates may require significant resources and may never be successful. If our products do not gain market acceptance, it would have a material adverse effect on our business, results of operations, and financial condition.
If our products are not covered and eligible for reimbursement from government and third party payors, we may not be able to generate significant revenue or achieve or sustain profitability.
The coverage and reimbursement status of newly approved or cleared drugs, diagnostic and laboratory tests is uncertain, and failure of our pharmaceutical products, diagnostic tests or laboratory tests to be adequately covered by insurance and eligible for adequate reimbursement could limit our ability to market any future product candidates we may develop and decrease our ability to generate revenue from any of our existing and future product candidates that may be approved or cleared. The commercial success of our existing and future products in both domestic and international markets will depend in part on the availability of coverage and adequate reimbursement from third-party payors, including government payors, such as the Medicare and Medicaid programs, managed care organizations, and other third-party payors, as well as our ability to obtain in network status with such payors. The government and other third-party payors are increasingly attempting to contain health care costs by limiting both insurance coverage and the level of reimbursement for new drugs and diagnostic tests and restricting in network status of laboratory providers. As a result, they may not cover or provide adequate payment for our product candidates. These payors may conclude that our products are less safe, less effective, or less cost-effective than existing or later-introduced products. These payors may also conclude that the overall cost of the procedure using one of our devices exceeds the overall cost of the competing procedure using another type of device, and third-party payors may not approve our products for insurance coverage and adequate reimbursement or approve our laboratory for in network status.
The failure to obtain coverage and adequate or any reimbursement for our products, or health care cost containment initiatives that limit or restrict reimbursement for our products, may reduce any future product revenue. Even though a drug (not administered by a physician) may be approved by the FDA, this does not mean that a Prescription Drug Plan (“PDP”), a private insurer operating under Medicare Part D, will list that drug on its formulary or will set a reimbursement level. PDPs are not required to make every FDA-approved drug available on their formularies. If our drug products are not listed on sufficient number of PDP formularies or if the PDPs’ levels of reimbursement are inadequate, our business, results of operations and financial condition could be materially adversely affected. Private health plans, such as managed care plans and pharmacy benefit management (“PBM”) programs may also not include our products on formularies, and may use other techniques that restrict access to our products or set a lower reimbursement rate than anticipated.
A significant portion of our revenues come from government subsidized healthcare programs such as Medicaid and Medicare. Our failure to comply with applicable Medicare, Medicaid and other governmental payor rules could result in our inability to participate in a governmental payor program, our returning funds already paid to us, civil monetary penalties, criminal penalties and/or limitations on the operational function of our laboratory. If we were unable to receive reimbursement under a governmental payor program, a substantial portion of our revenues would be lost, which would adversely affect our results of operations and financial condition. In addition, if a federal government shutdown were to occur for a prolonged period of time, federal government payment obligations, including its obligations under Medicaid and Medicare, may be delayed. Similarly, if state government shutdowns were to occur, state payment obligations may be delayed. If the federal or state governments fail to make payments under these programs on a timely basis, our business could suffer, and our financial position, results of operations or cash flows may be materially affected.
35
If the 4Kscore test is not covered and eligible for reimbursement from government and third party payors, we may not be able to generate significant revenue for the product.
On May 18, 2018, Novitas, the MAC for a jurisdiction that includes the State of New Jersey, where our 4Kscore test samples are processed, issued a draft non-coverage determination (“LCD”) that proposed no coverage for our 4Kscore test. We submitted comments to the draft LCD during the public comment period, which ended on July 5, 2018. In January 2019, Notivas issued a notice of future non-coverage determination for the 4Kscore test to be effective March 20, 2019. We are currently evaluating options to appeal the decision and undertake other steps with CMS in an effort to have this determination rescinded or reversed, however, there can be no assurance that we will be successful. We are also developing a strategy to obtain FDA approval for the 4Kscore test, among other efforts, to assist in securing broad reimbursement coverage. If we are not able to successfully appeal Novitas’ decision, we may not be able to obtain Medicare reimbursement for the 4Kscore test, which could result in a loss of revenues and could have a material adverse effect on our cash flows, results of operations, net income, and financial condition.
As we evolve from a company primarily involved in development to a company also involved in commercialization of our pharmaceutical and diagnostic products as well as our laboratory testing services, we may encounter difficulties in managing our growth and expanding our operations successfully.
As we advance our product candidates and expand our business, we will need to expand our development, regulatory and commercial infrastructure. As our operations expand, we expect that we will need to manage additional relationships with various third parties, collaborators and suppliers. Maintaining these relationships and managing our future growth will impose significant added responsibilities on members of our management. We must be able to: manage our development efforts and operations effectively; manage our clinical trials effectively; hire, train and integrate additional management, administrative and sales and marketing personnel; improve our managerial, development, operational and finance systems; implement and manage an effective marketing strategy; and expand our facilities, all of which may impose a strain on our administrative and operational infrastructure.
Furthermore, we may acquire additional businesses, products or product candidates that complement or augment our existing business. Integrating any newly acquired business or product could be expensive and time-consuming. We may not be able to integrate any acquired business or product successfully or operate any acquired business profitably. Our future financial performance will depend, in part, on our ability to manage any future growth effectively and our ability to integrate any acquired businesses. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing our company, which would have a material adverse effect on our business, results of operations and financial condition.
Our success is dependent to a significant degree upon the involvement,efforts and reputation of our Chairman and Chief Executive Officer, Phillip Frost, M.D.
Our success is dependent to a significant degree upon the efforts of our Chairman and CEO, Phillip Frost, M.D., who is essential to our business. The departure of our CEO for whatever reason or the inability of our CEO to continue to serve in his present capacity could have a material adverse effect upon our business, financial condition and results of operations. Our CEO has a highly regarded reputation in the pharmaceutical and medical industry and attracts business opportunities and assists both in negotiations with acquisition targets, investment targets and potential joint venture partners. Our CEO has also provided financing to us, both in terms of a credit agreement and equity investments. If we lost his services or if his reputation was damaged for whatever reason, including, but not limited to, as a result of the allegations underlying various SEC and shareholder lawsuits against us and Dr. Frost, our relationships with acquisition and investment targets, joint ventures, customers and investors, as well as our ability to obtain additional funding on acceptable terms, or at all, may suffer and could cause a material adverse impact on our operations, financial condition and the value of our common stock.
If we fail to attract and retain key management and scientific personnel, we may be unable to successfully operate our business and develop or commercialize our products and product candidates.
We will need to expand and effectively manage our managerial, operational, sales, financial, development, and other resources in order to successfully operate our business and pursue our research, development, and commercialization efforts for our products and product candidates. Our success depends on our continued ability to attract, retain, and motivate highly qualified management and pre-clinical and clinical personnel. The loss of the services or support of any of our senior management, particularly Dr. Phillip Frost, our Chairman and CEO, could delay or prevent the development and commercialization of our products and product candidates.
36
If the FDA or other applicable regulatory authorities approve generic products that compete with any of our products or product candidates, the sale of our products or product candidates may be adversely affected.
Once an NDA is approved, the product covered thereby becomes a “listed drug” which, in turn can be relied upon by potential competitors in support of an approval of an abbreviated new drug application, or ANDA, or 505(b)(2) application. U.S. laws and other applicable policies provide incentives to manufacturers to create modified, non-infringing versions of a drug to facilitate the approval of an ANDA or other application for a generic substitute. These manufacturers might only be required to conduct a relatively inexpensive study to show that their product has the same active ingredient(s), dosage form, strength, route of administration, and conditions of use, or labeling, as our product or product candidate and that the generic product is bioequivalent to ours, meaning it is absorbed in the body at the same rate and to the same extent as our product or product candidate. These generic equivalents, which must meet the same quality standards as branded pharmaceuticals, would be significantly less costly than ours to bring to market and companies that produce generic equivalents are generally able to offer their products at lower prices. Thus, after the introduction of a generic competitor, a significant percentage of sales of any branded product is typically lost to the generic product. Accordingly, competition from generic equivalents to our products or product candidates would materially adversely impact our revenues, profitability and cash flows and substantially limit our ability to obtain a return on the investments that we have made in our products and product candidates.
In 2017, Congress reauthorized the Generic Drug User Fee Act (GDUFA). The generic drug user fee program, established in 2012, is designed to speed the approval of new generic drugs. In addition, over the past few months, the FDA has used its regulatory authority to enact other programs to streamline the path to market for generic drugs. In addition, a regulatory pathway for biosimilars was established in 2012 including a new user fee program to promote the development of these products that show no clinically meaningful differences from innovator biologics. Though they have their own statutory market pathway, like generic drugs, biosimilars can receive FDA approval by providing less clinical data than the innovator product. Biosimilars are expected to be less expensive competitors to innovator biologics reducing prices overall. We anticipate several new biosimilars reaching the market over the next year.
If we fail to acquire and develop other products or product candidates at all or on commercially reasonable terms, we may be unable to diversify or grow our business.
We intend to continue to rely on acquisitions and in-licensing as a source of our products and product candidates for development and commercialization. The success of this strategy depends upon our ability to identify, select, and acquire pharmaceutical and diagnostic products, drug delivery technologies, and medical device product candidates. Proposing, negotiating, and implementing an economically viable product acquisition or license is a lengthy and complex process. We compete for partnering arrangements and license agreements with pharmaceutical, biotechnology and medical device companies, and academic research institutions. Our competitors may have stronger relationships with third parties with whom we are interested in collaborating and/or may have more established histories of developing and commercializing products.
Most of our competitors also have substantially greater financial and other resources than us. As a result, our competitors may have a competitive advantage in entering into partnering arrangements with such third parties, as such partnering arrangements are often decided in an auction process in which the highest bidder wins. In addition, even if we find promising products and product candidates, and generate interest in a partnering or strategic arrangement to acquire such products or product candidates, we may not be able to acquire rights to additional product candidates or approved products on terms that we find acceptable, or at all.
We expect that any product candidate to which we acquire rights will require additional development efforts prior to commercial sale, including extensive clinical testing and approval or clearance by the FDA and other non-U.S. regulatory authorities. All product candidates are subject to the risks of failure inherent in pharmaceutical, diagnostic test or medical device product development, including the possibility that the product candidate will not be shown to be sufficiently safe and effective for approval by regulatory authorities. Even if the product candidates are approved or cleared for marketing, we cannot be sure that they would be capable of economically feasible production or commercial success. If we fail to acquire or develop other product candidates that are capable of economically feasible production and commercial success, our business, results of operations and financial condition and cash flows may be materially adversely affected.
We rely on third parties to manufacture and supply our pharmaceutical and diagnostic products and product candidates.
If our manufacturing partners are unable to produce our products in the amounts that we require, we may not be able to establish a contract and obtain a sufficient alternative supply from another supplier on a timely basis and in the quantities we require. We expect to continue to depend on third-party contract manufacturers for the foreseeable future.
Our products and product candidates require precise, high quality manufacturing. Any of our contract manufacturers will be subject to ongoing periodic unannounced inspection by the FDA and other non-U.S. regulatory authorities to ensure strict
37
compliance with QSR regulations for devices or cGMPs for drugs, and other applicable government regulations and corresponding standards relating to matters such as testing, quality control, and documentation procedures. If our contract manufacturers fail to achieve and maintain high manufacturing standards in compliance with QSR or cGMPs, we may experience manufacturing errors resulting in patient injury or death, product recalls or withdrawals, delays or interruptions of production or failures in product testing or delivery, delay or prevention of filing or approval of marketing applications for our products, cost overruns, or other problems that could seriously harm our business.
Any performance failure on the part of our contract manufacturers could delay clinical development or regulatory approval or clearance of our product candidates or commercialization of our products and product candidates, depriving us of potential product revenue and resulting in additional losses. In addition, our dependence on a third party for manufacturing may adversely affect our future profit margins. Our ability to replace an existing manufacturer may be difficult because the number of potential manufacturers is limited and the FDA must approve any replacement manufacturer before it can begin manufacturing our products or product candidates. Such approval would result in additional non-clinical testing and compliance inspections. It may be difficult or impossible for us to identify and engage a replacement manufacturer on acceptable terms in a timely manner, or at all.
Independent clinical investigators and contract research organizations that we engage to conduct our clinical trials may not be diligent, careful or timely.
We depend on independent clinical investigators to conduct our clinical trials. Contract research organizations may also assist us in the collection and analysis of data. These investigators and contract research organizations will not be our employees, and we will not be able to control, other than by contract, the amount of resources, including time, that they devote to products that we develop. If independent investigators fail to devote sufficient resources to the development of product candidates or clinical trials, or if their performance is substandard, it will delay the marketing approval or clearance and commercialization of any products that we develop. Further, the FDA requires that we comply with standards, commonly referred to as good clinical practice, for conducting, recording and reporting clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity, and confidentiality of trial subjects are protected. If our independent clinical investigators and contract research organizations fail to comply with good clinical practice, the results of our clinical trials could be called into question and the clinical development of our product candidates could be delayed.
Failure of clinical investigators or contract research organizations to meet their obligations to us or comply with federal regulations and good clinical practice procedures could adversely affect the clinical development of our product candidates and harm our business, results of operations, and financial condition.
If the validity of an informed consent from a subject was to be challenged, it may negatively impact our product development efforts.
We take steps to ensure that all clinical data and genetic and other biological samples are collected from subjects who provide informed consent for the data and samples as required by applicable laws and we work to ensure that the subjects from whom our data and samples are collected do not retain any proprietary or commercial rights to the data or samples or any discoveries derived from them. However, because we may collect data and samples from countries that are governed by a number of different regulatory regimes, there are many complex legal questions relating to the adequacy of informed consent that we must continually address. The adequacy of any given subject’s informed consent may be challenged in the future, and any given informed consent may prove unlawful or otherwise inadequate for our purposes. Any findings against us, or our clinical collaborators, could obligate us to stop using some of our clinical samples, which in turn may hinder our product development efforts. Such a result would also likely involve legal challenges that may consume our management and financial resources.
Failure to timely or accurately bill and collect for our services could have a material adverse effect on our revenues and our business.
Billing for laboratory testing services is extremely complicated and is subject to extensive and non-uniform rules and administrative requirements. Depending on the billing arrangement and applicable law, we bill various payors, such as patients, insurance companies, Medicare, Medicaid, physicians, hospitals and employer groups. Changes in laws and regulations and payor practices increase the complexity and cost of our billing process. Additionally, in the U.S., third-party payors generally require billing codes on claims for reimbursement that describe the services provided. For laboratory services, the American Medical Association establishes most of the billing codes using a data code set called Current Procedural Terminology, or CPT, codes and the World Health Organization establishes diagnostic codes using a data set called International Statistical Classification of Diseases, or ICD-10, codes. Each third-party payor generally develops payment amounts and coverage policies for their beneficiaries or members that ties to the CPT code established for the laboratory test and the ICD-10 code selected by the ordering or performing physician. Therefore, coverage and reimbursement may differ by
38
payor even if the same billing code is reported for claims filing purposes. For laboratory tests without a specific billing code, payors often review claims on a claim-by-claim basis and there are increased uncertainties as to coverage and eligibility for reimbursement.
In addition to the items described above, third-party payors, including government programs, may decide to deny payment or recoup payments for testing that they contend was improperly billed or not medically necessary, against their coverage determinations, or for which they believe they have otherwise overpaid (including as a result of their own error), and we may be required to refund payments already received. Our revenues may be subject to retroactive adjustment as a result of these factors among others, including without limitation, differing interpretations of billing and coding guidance and changes by government agencies and payors in interpretations, requirements, and “conditions of participation” in various programs.
We implemented a new billing system for our laboratory business in the third quarter of 2016. The adoption of the new billing system, which replaced the old billing system, poses several challenges relating to, among other things, training of personnel, communication of new rules and procedures, changes in corporate culture, migration of data, and the potential instability of the new system. As an integral part of our billing compliance program, we assess our billing and coding practices in the ordinary course of business, respond to payor audits on a routine basis, and investigate reported failures or suspected failures to comply with federal and state healthcare reimbursement requirements, as well as overpayment claims which may arise from time to time without fault on our part. We have in the ordinary course of business been the subject of recoupments by payors and have from time to time identified and reimbursed payors for overpayments.
Incorrect or incomplete documentation and billing information, as well as the other items described above, among other factors, could result in non-payment for services rendered or having to pay back amounts incorrectly billed and collected. Further, the failure to timely or correctly bill could lead to various penalties, including: (1) exclusion from participation in the CMS and other government programs; (2) asset forfeitures; (3) civil and criminal fines and penalties; and (4) the loss of various licenses, certificates and authorizations necessary to operate our business, any of which could have a material adverse effect on our results of operations or cash flows.
Failure in our information technology systems, including by cybersecurity attacks or other data security incidents, could significantly increase testing turn-around time or billing processes and otherwise disrupt our operations.
Our operations depend, in part, on the continued performance of our information technology systems. Our information technology systems are potentially vulnerable to physical or electronic break-ins, computer viruses and similar disruptions. In addition, we are in the process of integrating the information technology systems of our subsidiaries, and we may experience system failures or interruptions as a result of this process. Sustained system failures or interruption of our systems in one or more of our laboratory operations could disrupt our ability to process laboratory requisitions, perform testing, provide test results in a timely manner and/or bill the appropriate party. Failure of our information technology systems could adversely affect our business, profitability and financial condition.
A successful cybersecurity attack or other data security incident could result in the misappropriation and/or loss of confidential or personal information, create system interruptions, or deploy malicious software that attacks our systems. It is possible that a cybersecurity attack might not be noticed for some period of time. The occurrence of a cybersecurity attack or incident could result in business interruptions from the disruption of our information technology systems, or negative publicity resulting in reputational damage with our customers, shareholders and other stakeholders and/or increased costs to prevent, respond to or mitigate cybersecurity events. In addition, the unauthorized dissemination of sensitive personal information or proprietary or confidential information could expose us or other third-parties to regulatory fines or penalties, litigation and potential liability, or otherwise harm our business.
Healthcare plans have taken steps to control the utilization and reimbursement of healthcare services, including clinical test services.
We also face efforts by non-governmental third-party payors, including healthcare plans, to reduce utilization and reimbursement for clinical testing services.
The healthcare industry has experienced a trend of consolidation among healthcare insurance plans, resulting in fewer but larger insurance plans with significant bargaining power to negotiate fee arrangements with healthcare providers, including clinical testing providers. These healthcare plans, and independent physician associations, may demand that clinical testing providers accept discounted fee structures or assume all or a portion of the financial risk associated with providing testing services to their members through capped payment arrangements. In addition, some healthcare plans limit the laboratory network to only a single national or regional laboratory to obtain improved fee-for-service pricing. There is also an increasing number of patients enrolling in consumer driven products and high deductible plans that involve greater patient cost-sharing.
39
The increased consolidation among healthcare plans also has increased the potential adverse impact of ceasing to be a contracted provider with any such insurer.
We expect continuing efforts to limit the number of participating laboratories in payor networks, reduce reimbursements, to impose more stringent cost controls and to reduce utilization of clinical test services. These efforts, including future changes in third-party payor rules, practices and policies, or failing to become a contracted provider or ceasing to be a contracted provider to a healthcare plan, may have a material adverse effect on our business.
The success of our business may be dependent on the actions of our collaborative partners.
We have entered into and expect in the future to enter into collaborative arrangements with established multi-national pharmaceutical, diagnostic, and medical device companies, which will finance or otherwise assist in the development, manufacture and marketing of products incorporating our technology. We anticipate deriving some revenues from research and development fees, license fees, milestone payments, and royalties from collaborative partners. Our prospects, therefore, may depend to some extent upon our ability to attract and retain collaborative partners and to develop technologies and products that meet the requirements of prospective collaborative partners. In addition, our collaborative partners may have the right to abandon research projects, guide strategy regarding prosecution of relevant patent applications and terminate applicable agreements, including funding obligations, prior to or upon the expiration of the agreed-upon research terms. There can be no assurance that we will be successful in establishing collaborative arrangements on acceptable terms or at all, that collaborative partners will not terminate funding before completion of projects, that our collaborative arrangements will result in successful product commercialization, or that we will derive any revenues from such arrangements. To the extent that we are unable to develop and maintain collaborative arrangements, we would need substantial additional capital to undertake research, development, and commercialization activities on our own.
If we are unable to obtain and enforce patent protection for our products, our business could be materially harmed.
Our success depends, in part, on our ability to protect proprietary methods and technologies that we develop or license under the patent and other intellectual property laws of the U.S. and other countries, so that we can prevent others from unlawfully using our inventions and proprietary information. However, we may not hold proprietary rights to some patents required for us to commercialize our products and product candidates. Because certain U.S. patent applications are confidential, third parties may have filed patent applications for technology covered by our pending patent applications without our being aware of those applications, and our patent applications may not have priority over those applications. For this and other reasons, we or our third-party collaborators may be unable to secure desired patent rights, thereby losing desired exclusivity. If licenses are not available to us on acceptable terms, we may not be able to market the affected products or conduct the desired activities, unless we challenge the validity, enforceability, or infringement of the third-party patent or otherwise circumvent the third-party patent.
Our strategy depends on our ability to rapidly identify and seek patent protection for our discoveries. In addition, we will rely on third-party collaborators to file patent applications relating to proprietary technology that we develop jointly during certain collaborations. The process of obtaining patent protection is expensive and time-consuming. If our present or future collaborators fail to file and prosecute all necessary and desirable patent applications at a reasonable cost and in a timely manner, our business will be adversely affected. Unauthorized parties may be able to obtain and use information that we regard as proprietary.
The issuance of a patent does not guarantee that it is valid or enforceable. Any patents we have obtained, or obtain in the future, may be challenged, invalidated, unenforceable, or circumvented. Moreover, the U.S. Patent and Trademark Office (the “USPTO”) may commence interference proceedings involving our patents or patent applications. In addition, court decisions may introduce uncertainty in the enforceability or scope of patents owned by biotechnology, pharmaceutical, and medical device companies. Any challenge to, finding of unenforceability or invalidation or circumvention of, our patents or patent applications would be costly, would require significant time and attention of our management, and could have a material adverse effect on our business, results of operations and financial condition.
Our pending patent applications may not result in issued patents. The patent position of pharmaceutical, biotechnology, diagnostic, and medical device companies, including ours, is generally uncertain and involves complex legal and factual considerations. The standards that the USPTO and its foreign counterparts use to grant patents are not always applied predictably or uniformly and can change. There is also no uniform, worldwide policy regarding the subject matter and scope of claims granted or allowable in pharmaceutical, biotechnology, diagnostic, or medical device patents. Accordingly, we do not know the degree of future protection for our proprietary rights or the breadth of claims that will be allowed in any patents issued to us or to others. The legal systems of certain countries do not favor the aggressive enforcement of patents, and the laws of foreign countries may not protect our rights to the same extent as the laws of the U.S. Therefore, the enforceability or scope of our owned or licensed patents in the U.S. or in foreign countries cannot be predicted with certainty, and, as a result,
40
any patents that we own or license may not provide sufficient protection against competitors. We may not be able to obtain or maintain patent protection for our pending patent applications, those we may file in the future, or those we may license from third parties.
We cannot assure you that any patents that have issued, that may issue, or that may be licensed to us will be enforceable or valid, or will not expire prior to the commercialization of our products and product candidates, thus allowing others to more effectively compete with us. Therefore, any patents that we own or license may not adequately protect our products and product candidates or our future products, which could have a material adverse effect on our business, results of operations, and financial condition.
If we are unable to protect the confidentiality of our proprietary information and know-how, the value of our technology and products could be adversely affected.
In addition to patent protection, we also rely on other proprietary rights, including protection of trade secrets, know-how, and confidential and proprietary information. To maintain the confidentiality of trade secrets and proprietary information, we will seek to enter into confidentiality agreements with our employees, consultants, and collaborators upon the commencement of their relationships with us. These agreements generally require that all confidential information developed by the individual or made known to the individual by us during the course of the individual’s relationship with us be kept confidential and not disclosed to third parties. Our agreements with employees also generally provide that any inventions conceived by the individual in the course of rendering services to us shall be our exclusive property.
However, we may not obtain these agreements in all circumstances, and individuals with whom we have these agreements may not comply with their terms. In the event of unauthorized use or disclosure of our trade secrets or proprietary information, these agreements, even if obtained, may not provide meaningful protection, particularly for our trade secrets or other confidential information. To the extent that our employees, consultants, or contractors use technology or know-how owned by third parties in their work for us, disputes may arise between us and those third parties as to the rights in related inventions.
Adequate remedies may not exist in the event of unauthorized use or disclosure of our confidential information. The disclosure of our trade secrets would impair our competitive position and may materially harm our business, financial condition, and results of operations.
We will rely heavily on licenses from third parties. Failure to comply with the provisions of these licenses could result in the loss of our rights under the license agreements.
Many of the patents and patent applications in our patent portfolio are not owned by us, but are licensed from third parties. Such license agreements give us rights for the commercial exploitation of the patents resulting from the respective patent applications, subject to certain provisions of the license agreements. Failure to comply with these provisions could result in the loss of our rights under these license agreements. Our inability to rely on these patents and patent applications, which are the basis of our technology, would have a material adverse effect on our business, results of operations and financial condition.
We license patent rights to certain of our technology from third-party owners. If such owners do not properly maintain or enforce the patents underlying such licenses, our competitive position and business prospects will be harmed.
We have obtained licenses from, among others, INEOS Healthcare, the President and Fellows of Harvard College, The Scripps Research Institute, Arctic Partners, and Academia Sinica, that are necessary or useful for our business. In addition, we intend to enter into additional licenses of third-party intellectual property in the future. We cannot guarantee that no third parties will step forward and assert inventorship or ownership in our in-licensed patents. In some cases, we may rely on the assurances of our licensors that all ownership rights have been secured and that all necessary agreements are intact or forthcoming.
Our success will depend in part on our ability or the ability of our licensors to obtain, maintain, and enforce patent protection for our licensed intellectual property and, in particular, those patents to which we have secured exclusive rights in our field. We or our licensors may not successfully prosecute the patent applications which are licensed to us. Even if patents issue in respect of these patent applications, we or our licensors may fail to maintain these patents or may determine not to pursue litigation against other companies that are infringing these patents. Without protection for the intellectual property we have licensed, other companies might be able to offer substantially identical products for sale, which could adversely affect our competitive business position and harm our business, results of operations and financial condition.
41
Our commercial success depends significantly on our ability to operate without infringing the patents and other proprietary rights of third parties.
Other entities may have or obtain patents or proprietary rights that could limit our ability to develop, manufacture, use, sell, offer for sale or import products, or impair our competitive position. In addition, other entities may have or obtain patents or proprietary rights that cover our current research and preclinical studies. The U.S. case law pertaining to statutory exemptions to patent infringement for those who are using third party patented technology in the process of pursuing FDA regulatory approval changes over time. Lawsuits involving such exemptions are very fact intensive and it is currently unclear under U.S. case law whether preclinical studies would always qualify for such an exemption, and whether such exemptions would apply to research tools. To the extent that our current research and preclinical studies may be covered by the patent rights of others, the risk of suit may continue after such patents expire because the statute of limitations for patent infringement runs for six years. To the extent that a third party develops and patents technology that covers our products, we may be required to obtain licenses to that technology, which licenses may not be available or may not be available on commercially reasonable terms, if at all. If licenses are not available to us on acceptable terms, we will not be able to market the affected products or conduct the desired activities, unless we challenge the validity, enforceability or infringement of the third-party patent, or circumvent the third-party patent, which would be costly and would require significant time and attention of our management. Third parties may have or obtain by license or assignment valid and enforceable patents or proprietary rights that could block us from developing products using our technology. Our failure to obtain a license to any technology that we require may materially harm our business, financial condition, and results of operations.
If we become involved in patent litigation or other proceedings related to a determination of rights, we could incur substantial costs and expenses, substantial liability for damages or be required to stop our product development and commercialization efforts.
Third parties may sue us for infringing their patent rights. Likewise, we may need to resort to litigation to enforce a patent issued or licensed to us or to determine the scope and validity of proprietary rights of others. In addition, a third-party may claim that we have improperly obtained or used its confidential or proprietary information. Furthermore, in connection with our third-party license agreements, we generally have agreed to indemnify the licensor for costs incurred in connection with litigation relating to intellectual property rights. The cost to us of any litigation or other proceeding relating to intellectual property rights, even if resolved in our favor, could be substantial, and the litigation would divert our management’s efforts. Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. Uncertainties resulting from the initiation and continuation of any litigation could limit our ability to continue our operations. Our involvement in patent litigation and other proceedings could have a material adverse effect on our business, results of operations, and financial condition.
If any parties successfully claim that our creation or use of proprietary technologies infringes upon their intellectual property rights, we might be forced to pay damages, potentially including treble damages, if we are found to have willfully infringed on such parties’ patent rights. In addition to any damages we might have to pay, a court could require us to stop the infringing activity or obtain a license. Any license required under any patent may not be made available on commercially acceptable terms, if at all. In addition, such licenses are likely to be non-exclusive and, therefore, our competitors may have access to the same technology licensed to us. If we fail to obtain a required license and are unable to design around a patent, we may be unable to effectively market some of our technology and products, which could limit our ability to generate revenues or achieve profitability and possibly prevent us from generating revenue sufficient to sustain our operations.
We have faced, and may in the future face, intellectual property infringement claims that could be time-consuming and costly to defend, and could result in our loss of significant rights and the assessment of treble damages.
We may from time to time receive notices of claims of infringement and misappropriation or misuse of other parties’ proprietary rights. Some of these additional claims may also lead to litigation. We cannot assure you that we will prevail in such actions, or that other actions alleging misappropriation or misuse by us of third-party trade secrets, infringement by us of third-party patents and trademarks or the validity of our patents, will not be asserted or prosecuted against us.
We may also initiate claims to defend our intellectual property or to seek relief on allegations that we use, sell, or offer to sell technology that incorporates third party intellectual property. Intellectual property litigation, regardless of outcome, is expensive and time-consuming, could divert management’s attention from our business and have a material negative effect on our business, operating results or financial condition. If there is a successful claim of infringement against us, we may be required to pay substantial damages (including treble damages if we were to be found to have willfully infringed a third party’s patent) to the party claiming infringement, develop non-infringing technology, stop selling our tests or using technology that contains the allegedly infringing intellectual property or enter into royalty or license agreements that may not be available on
42
acceptable or commercially practical terms, if at all. Our failure to develop non-infringing technologies or license the proprietary rights on a timely basis could harm our business.
It is possible that a third party or patent office might take the position that one or more patents or patent applications constitute prior art in the field of genomic-based diagnostics. In such a case, we might be required to pay royalties, damages and costs to firms who own the rights to these patents, or we might be restricted from using any of the inventions claimed in those patents.
We may become subject to product liability for our diagnostic tests, clinical trials, pharmaceutical products and medical device products.
Our success depends on the market’s confidence that we can provide reliable, high-quality pharmaceuticals, medical devices, and diagnostics tests. Our reputation and the public image of our products or technologies may be impaired if our products fail to perform as expected or our products are perceived as difficult to use. Our products are complex and may develop or contain undetected defects or errors. Furthermore, if product or future product candidate harms people, or is alleged to be harmful, we may be subject to costly and damaging product liability claims brought against us by clinical trial participants, consumers, health care providers, corporate partners or others. We have product liability insurance covering commercial sales of current products and our ongoing clinical trials. Any defects or errors could lead to the filing of product liability claims, which could be costly and time-consuming to defend and result in substantial damages. If we experience a sustained material defect or error, this could result in loss or delay of revenues, delayed market acceptance, damaged reputation, diversion of development resources, legal claims, increased insurance costs or increased service and warranty costs, any of which could materially harm our business. We cannot assure you that our product liability insurance would protect our assets from the financial impact of defending a product liability claim. A product liability claim could have a serious adverse effect on our business, financial condition and results of operations.
We are the subject of pending civil litigation which could require us to pay substantial damages or could otherwise have a material adverse effect on us.
On September 7, 2018, the SEC filed a lawsuit in the Southern District of New York (the “Complaint”), against a number of individuals and entities (each a “Defendant” and, collectively, the “Defendants”) including us and our CEO and Chairman, Dr. Phillip Frost. The SEC alleged that we (i) aided and abetted a purported “pump and dump” scheme in connection with one company perpetrated by a number of the Defendants, and (ii) failed to file required Schedules 13D or 13G with the SEC. The Complaint also alleged that Dr. Frost (i) participated in the alleged market manipulation in connection with two companies, (ii) failed to file required Schedule 13Ds with the SEC, and (iii) sold unregistered securities without an applicable exemption. Following the SEC’s announcement of the Complaint, a number of class action and derivative suits were filed against us and our directors and officers concerning the allegations in the Complaint and related matters.
In December 2018, we and Dr. Frost entered into settlements with the SEC, which, upon approval by the court in January 2019, resolved the claims against us and Dr. Frost raised in the Complaint. Pursuant to the settlement between us and the SEC, and without admitting or denying any of the allegations of the Complaint, we agreed to an injunction from violations of Section 13(d) of the Securities Exchange Act of 1934 (the “Exchange Act”), a strict liability claim, and to pay a $100,000 penalty, which has been paid. We also agreed to, within certain stipulated time periods: (i) establish a Management Investment Committee (“MIC”) that will make recommendations to an Independent Investment Committee (“IIC”) of our Board of Directors in connection with existing and future strategic minority investments; and (ii) retain an Independent Compliance Consultant (“ICC”) to (a) advise us on whether filings pursuant to Section 13(d) of the Exchange Act for previous strategic investments made at the suggestion of or in tandem with Dr. Frost should be amended or made to reflect group membership with Dr. Frost and his related entities; (b) review our existing policies and procedures relating to compliance with Section 13(d) of the Exchange Act; and (c) review the independence of the MIC and IIC of our Board of Directors solely for purposes of the handling of strategic minority investments. The ICC is required to report its findings (including recommendations as to filings, amendments, improvements to policies and procedures, and improvement to the composition of the MIC and the IIC to our Board of Directors) to the SEC within 15 days of completion of its work, and we are required to implement the ICC’s recommendations, and to certify our compliance with these undertakings in writing.
Under the terms of the settlement between the SEC and Dr. Frost, and without admitting or denying any of the allegations in the Complaint, Dr. Frost agreed to injunctions from violations of Sections 5(a) and (c) and 17(a)(2) of the Securities Act, claims which may be satisfied by strict liability and negligence, respectively, and Section 13(d) of the Exchange Act, also a strict liability claim; to pay approximately $5.5 million in penalty, disgorgement and pre-judgment interest, which has been paid; and to be prohibited, with certain exceptions, from trading in penny stocks.
The settlements include no restriction on Dr. Frost’s ability to continue to serve as our CEO and Chairman.
43
We are separately evaluating our strategic minority investments and reporting under Section 13(d) of the Exchange Act. In connection with this evaluation, we may make additional or amended filings pursuant to Section 13(d) of the Exchange Act reflecting group membership.
Although the SEC matter against us and Dr. Frost is resolved, there can be no assurance that additional charges from other governmental authorities will not be brought against one or more parties named in the Complaint.
We also continue to face a number of class actions and derivative suits concerning the allegations in the SEC Complaint. We cannot predict with certainty the outcome or effect of the class actions or derivative suits, which could require us to pay substantial damages or could otherwise have a material adverse effect on us.
Our primary and side A directors and officers liability insurance carrier has denied coverage for the class action and derivative suits filed against us and our directors and officers concerning the allegations in the Complaint. We believe that this denial is in error and are in the process of appealing this coverage determination. If we are unsuccessful in this appeal, or if other third-party insurers deny, cancel, or refuse coverage, which we are not able to successfully appeal, or are otherwise unable to provide us with adequate insurance coverage for all or any of the aforementioned lawsuits, then our overall risk exposure and operational expenses could increase and the management of our business operations could be disrupted, which could cause a material adverse impact on our business, operations and financial condition. Further, an unusually large liability claim or a string of claims, like these lawsuits, could potentially exceed our available insurance coverage if any. In addition, the availability of, and our ability to collect on, insurance coverage can be subject to factors beyond our control.
As our current insurance policies expire, increased premiums for renewed or new coverage, if such coverage can be secured at all, may increase our insurance expense and/or require us to increase our self-insured retention or deductibles. If the number of claims or the dollar amounts of any such claims rise in any policy year, we could suffer additional costs associated with accessing excess coverage policies. Also, an increase in the loss amounts attributable to such claims could expose us to uninsured damages if we are unable or elect not to insure against certain claims because of increased premiums or other reasons. These lawsuits or the resolution of such lawsuits may affect the availability or cost of some of our insurance coverage, which could materially adversely impact our business, results of operations and cash flows and potentially expose us to increased risks that would be uninsured.
Adverse results in material litigation matters or governmental inquiries could have a material adverse effect upon our business and financial condition.
We may from time to time become subject in the ordinary course of business to material legal action related to, among other things, intellectual property disputes, professional liability, contractual and employee-related matters, as well as inquiries from governmental agencies and Medicare or Medicaid carriers requesting comment and information on allegations of billing irregularities and other matters that are brought to their attention through billing audits, third parties or other sources. The health care industry is subject to substantial federal and state government regulation and audit. Additionally, we are subject to pending legal proceedings with respect to alleged violations of securities laws. See “We are the subject of pending civil litigation which could require us to pay substantial damages or could otherwise have a material adverse effect on us” above.
From time to time, we may receive inquiries, document requests, Civil Investigative Demands (“CIDs”) or subpoenas from the Department of Justice, the Office of Inspector General and Office for Civil Rights (“OCR”) of the Department of Health and Human Services, the Centers for Medicare and Medicaid Services, various payors and fiscal intermediaries, and other state and federal regulators regarding investigations, audits and reviews. We are currently responding to CIDs, subpoenas or document requests for various matters relating to our laboratory operations. Some pending or threatened proceedings against us may involve potentially substantial amounts as well as the possibility of civil, criminal, or administrative fines, penalties, or other sanctions, which could be material. Settlements of suits involving the types of issues that we routinely confront may require monetary payments as well as corporate integrity agreements. Additionally, qui tam or “whistleblower” actions initiated under the civil False Claims Act may be pending but placed under seal by the court to comply with the False Claims Act’s requirements for filing such suits. The Company generally has cooperated, and intends to continue to cooperate, with appropriate regulatory authorities as and when investigations, audits and inquiries arise.
Legal actions and government investigations could result in substantial monetary damages, negatively impact our ability to obtain additional funding on acceptable terms, or at all, and damage to our reputation with customers, business partners and other third parties, all of which could have a material adverse effect upon our results of operations and financial position. Further, the legal actions and government investigations could damage our reputation with investors and adversely affect the trading prices of our securities.
44
RISKS RELATED TO REGULATORY COMPLIANCE
Our ability to successfully operate our laboratories and develop and commercialize certain of our diagnostic tests and LDTs will depend on our ability to maintain required regulatory licensures and comply with all the CLIA requirements.
In order to successfully operate our laboratory business and offer certain of our diagnostic tests and LDTs, we must maintain our CLIA certification and comply with all the CLIA requirements. CLIA is designed to ensure the quality and reliability of clinical laboratories by mandating specific standards in the areas of personnel qualifications, administration and participation in proficiency testing, patient test management, quality control, quality assurance and inspections. The sanction for failure to comply with CLIA requirements may be suspension, revocation or limitation of a laboratory’s CLIA certificate, which is necessary to conduct business, as well as significant fines and/or criminal penalties. Laboratories must undergo on-site surveys at least every two years, which may be conducted by the Federal CLIA program or by a private CMS approved accrediting agency such as CAP, among others. Our laboratories are also subject to regulation of laboratory operations under state clinical laboratory laws as will be any new CLIA-certified laboratory that we establish or acquire. State clinical laboratory laws may require that laboratories and/or laboratory personnel meet certain qualifications, specify certain quality controls or require maintenance of certain records. Certain states, such as California, Florida, Maryland, New York, Pennsylvania and Rhode Island, require that laboratories obtain licenses to test specimens from patients residing in those states and additional states may require similar licenses in the future. If we are unable to obtain and maintain licenses from states where required, we will not be able to process any samples from patients located in those states. Only Washington and New York States are exempt under CLIA, as these states have established laboratory quality standards at least as stringent as CLIA’s. Potential sanctions for violation of these statutes and regulations include significant fines and the suspension or loss of various licenses, certificates and authorizations, which could adversely affect our business and results of operations.
If we fail to comply with CLIA requirements, HHS or state agencies could require us to cease diagnostic testing. Even if it were possible for us to bring our laboratories back into compliance after failure to comply with such requirements, we could incur significant expenses and potentially lose revenues in doing so. Moreover, new interpretations of current regulations or future changes in regulations under CLIA may make it difficult or impossible for us to comply with the CLIA classification, which would significantly harm our business and materially adversely affect our financial condition.
The regulatory approval process is expensive, time consuming and uncertain and may prevent us or our collaboration partners from obtaining approvals for the commercialization of some or all of our product candidates.
The research, testing, manufacturing, labeling, approval, selling, marketing, and distribution of drug products, diagnostic products, or medical devices are subject to extensive regulation by the FDA and other non-U.S. regulatory authorities, which regulations differ from country to country. In general, we are not permitted to market our product candidates in the U.S. until we receive approval of a Biologics License Application (BLA), an approval of a NDA, a clearance letter under the premarket notification process, or 510(k) process, or an approval of a PMA from the FDA. To date, we have only submitted one NDA which was approved in June 2016. We have received FDA approval of the PMA for our Sangia Total PSA Test using the Claros Analyzer but we have not received marketing approval or clearance for any of our other diagnostic product candidates, Obtaining approval of a NDA or PMA can be a lengthy, expensive, and uncertain process. With respect to medical devices, while the FDA reviews and clears a premarket notification in as little as three months, there is no guarantee that our products will qualify for this more expeditious regulatory process, which is reserved for Class I and II devices, nor is there any assurance that even if a device is reviewed under the 510(k) process that the FDA will review it expeditiously or determine that the device is substantially equivalent to a lawfully marketed non-PMA device. If the FDA fails to make this finding, then we cannot market the device. In lieu of acting on a premarket notification, the FDA may seek additional information or additional data which would further delay our ability to market the product. Furthermore, we are not permitted to make changes to a device approved through the PMA or 510(k) which affects the safety or efficacy of the device without first submitting a supplement application to the PMA and obtaining FDA approval or cleared premarket notification for that supplement. In some cases, the FDA may require clinical trials to support a supplement application. In addition, failure to comply with FDA, non-U.S. regulatory authorities, or other applicable U.S. and non-U.S. regulatory requirements may, either before or after product approval or clearance, if any, subject our company to administrative or judicially imposed sanctions, including, but not limited to the following:
▪ |
restrictions on the products, manufacturers, or manufacturing process; |
▪ |
adverse inspectional observations (Form 483), warning letters, or non-warning letters incorporating inspectional observations; |
▪ |
civil and criminal penalties; |
▪ |
injunctions; |
45
▪ |
suspension or withdrawal of regulatory approvals or clearances; |
▪ |
product seizures, detentions, or import bans; |
▪ |
voluntary or mandatory product recalls and publicity requirements; |
▪ |
total or partial suspension of production; |
▪ |
imposition of restrictions on operations, including costly new manufacturing requirements; and |
▪ |
refusal to approve or clear pending NDAs or supplements to approved NDAs, applications or pre-market notifications. |
Regulatory approval of an NDA or NDA supplement, BLA, PMA, PMA supplement or clearance pursuant to a pre-market notification is not guaranteed, and the approval or clearance process, as the case may be, is expensive and may, especially in the case of an NDA or PMA application, take several years. The FDA also has substantial discretion in the drug and medical device approval and clearance process. Failure can occur at any stage, and we could encounter problems that cause us to abandon clinical trials or to repeat or perform additional pre-clinical studies and clinical trials. The number of pre-clinical studies and clinical trials that will be required for FDA approval or clearance varies depending on the drug or medical device candidate, the disease or condition that the drug or medical device candidate is designed to address, and the regulations applicable to any particular drug or medical device candidate. The FDA can delay, limit or deny approval or clearance of a drug or medical device candidate for many reasons, including:
▪ |
a drug candidate may not be deemed safe or effective; |
▪ |
a medical device candidate may not be deemed to be substantially equivalent to a lawfully marketed non-PMA device, in the case of a premarket notification; |
▪ |
the FDA may not find the data from pre-clinical studies and clinical trials sufficient; |
▪ |
the FDA may not approve our or our third-party manufacturer’s processes or facilities; or |
▪ |
the FDA may change its approval or clearance policies or adopt new regulations. |
Beyond these risks, there is also a possibility that our licensees or collaborators could decide to discontinue a study at any time for commercial, scientific or other reasons.
Regulation by governmental authorities in the U.S. and other countries may be a significant factor in how we develop, test, produce and market our diagnostic test products. Diagnostic tests like ours may not fall squarely within the regulatory approval process for pharmaceutical or device products as described above, and the regulatory pathway is not as clear. It is possible that the diagnostic products developed by us or our collaborators will be regulated as medical devices by the FDA and comparable agencies of other countries and require either PMA or 510(k) clearance from the FDA prior to marketing. Some companies that have successfully commercialized diagnostic tests for various conditions and disease states have not sought clearance or approval for such tests through the traditional 510(k) or PMA processes, and have instead utilized a process involving LDTs through a CLIA- certified laboratory. CLIA is a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for diagnostic, preventative or treatment purpose. In such instances, the CLIA lab is solely responsible for the development, validation and commercialization of the assay.
Such LDT testing is currently under the purview of CMS and state agencies that provide oversight of the safe and effective use of LDTs. However, the FDA has consistently asserted that it has the regulatory authority to regulate LDTs despite historically exercising enforcement discretion. In furtherance of that position, the FDA issued two draft guidance documents in October 2014: (1) Framework for Regulatory Oversight of Laboratory Developed Tests (the “Framework Guidance”); and (2) FDA Notification and Medical Device Reporting for Laboratory Developed Tests (the “Notification Guidance”). The Framework Guidance outlines the FDA’s plan to adopt over time a risk-based approach to regulating LDTs whereby different classifications of LDTs would be subject to different levels of FDA oversight and enforcement, including, for example, prohibitions on adulteration and misbranding, establishment registration and device listing, premarket notification, banned devices, records and reports, good manufacturing practices, adverse event reporting, premarket review of safety, effectiveness, and clinical validity, and quality system requirements. The Notification Guidance is intended to explain how clinical laboratories should notify the FDA of the LDTs they develop and how to satisfy Medical Device Reporting requirements. On January 13, 2017, the FDA published a synthesis of feedback on the Framework Guidance and Notification Guidance titled, Discussion Paper on Laboratory Developed Tests (the “Discussion Paper”). The Discussion Paper provided notice that the FDA would not issue a final guidance on the oversight of LDTs to allow for further public discussion on appropriate oversight
46
approach, and to give congressional authorizing committees the opportunity to develop a legislative solution. The outcome and ultimate impact of such proposals on the business is difficult to predict at this time. However, the FDA’s authority to regulate LDTs continues to be challenged and the regulatory situation is fluid. The timeline and process for finalizing the draft guidance documents is unknown. We will continue to monitor changes to all domestic and international LDT regulatory policy so as to ensure compliance with the current regulatory scheme.
The terms of approvals and ongoing regulation of our products may limit how we manufacture and market our products and product candidates, which could materially impair our ability to generate anticipated revenues.
We, our approved or cleared products, and the manufacturers of our products are subject to continual review. Our approved or cleared products may only be promoted for their indicated uses. Marketing, labeling, packaging, adverse event reporting, storage, advertising, and promotion for our approved products will be subject to extensive regulatory requirements. We train our marketing and sales force against promoting our products for uses outside of the cleared or approved indications for use, known as “off-label uses.” If the FDA determines that our promotional materials or training constitute promotion of unsupported claims or an off-label use, it could request that we modify our training or promotional materials or subject us to regulatory or enforcement actions, including the issuance of an untitled letter, a warning letter, injunction, seizure, civil fine or criminal penalties. It is also possible that other federal, state or foreign enforcement authorities might take action if they consider our business activities to constitute promotion of an off-label use, which could result in significant penalties, including, but not limited to, criminal, civil and/or administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs, and the curtailment of our operations.
We and the manufacturers of our products are also required to comply with current Good Manufacturing Practices (“cGMP”) regulations or the FDA’s QSR regulations, which include requirements relating to quality control and quality assurance as well as the corresponding maintenance of records and documentation. Moreover, device manufacturers are required to report adverse events by filing Medical Device Reports with the FDA, which reports are publicly available.
Further, regulatory agencies must approve manufacturing facilities before they can be used to manufacture our products, and these facilities are subject to ongoing regulatory inspection. If we fail to comply with the regulatory requirements of the FDA and other non-U.S. regulatory authorities, or if previously unknown problems with our products, manufacturers, or manufacturing processes are discovered, we could be subject to administrative or judicially imposed sanctions. Furthermore, any limitation on indicated uses for a product or product candidate or our ability to manufacture and promote a product or product candidate could significantly and adversely affect our business, results of operations, and financial condition.
In addition, the FDA and other non-U.S. regulatory authorities may change their policies and additional regulations may be enacted that could prevent or delay marketing approval or clearance of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the U.S. or abroad. If we are not able to maintain regulatory compliance, we would likely not be permitted to market our products or product candidates and we may not achieve or sustain profitability, which would materially impair our ability to generate anticipated revenues.
If we fail to comply with complex and rapidly evolving laws and regulations, we could suffer penalties, be required to pay substantial damages or make significant changes to our operations.
We are subject to numerous federal and state regulations, including, but not limited to:
• |
federal and state laws applicable to billing and claims payment; |
• |
federal and state laboratory anti-mark-up laws; |
• |
federal and state anti-kickback laws; |
• |
physician self-referral law; |
• |
federal and state false claims laws; |
• |
federal self-referral and financial inducement prohibition laws, commonly known as the Stark Law, and the state equivalents; |
• |
federal and state laws governing laboratory licensing and testing, including CLIA; |
• |
federal and state laws governing the development, use and distribution of LDTs; |
• |
HIPAA, along with the revisions to HIPAA as a result of the HITECH Act, and analogous state laws and non-US laws, including the General Data Protection Regulation; |
47
• |
federal, state and foreign regulation of privacy, security, electronic transactions and identity theft; |
• |
federal, state and local laws governing the handling, transportation and disposal of medical and hazardous waste; |
• |
Occupational Safety and Health Administration rules and regulations; |
• |
changes to laws, regulations and rules as a result of the implementation and/or repeal of part or all of 2010 Health Care Reform Legislation; and |
• |
changes to other federal, state and local laws, regulations and rules, including tax laws. |
If we fail to comply with existing or future applicable laws and regulations, we could suffer civil or criminal penalties, including the loss of our licenses to operate our laboratories and our ability to participate in federal and state healthcare programs. Different interpretations and enforcement policies of existing statutes and regulations applicable to our business could subject our current practices to allegations of impropriety or illegality, or could require us to make significant changes to our operations. Under the FCA, whistleblower or qui tam provisions allow a private individual to bring actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In recent years, the number of suits brought by private individuals has increased dramatically and we may be subject to such suits. Violations of the FCA could result in enormous economic liability and could have a material impact on us. As a result of political, economic, and regulatory influences, the healthcare delivery industry in the U.S. is under intense scrutiny and subject to fundamental changes. We cannot predict which reform proposals will be adopted, when they may be adopted, or what impact they may have on us. The costs associated with complying with federal and state regulations could be significant and the failure to comply with any such legal requirements could have a material adverse effect on our financial condition, results of operations, and liquidity.
Tax reform may significantly affect us and our stockholders.
On December 22, 2017, President Trump signed into law the Tax Cuts and Jobs Act (the “Tax Act”) that significantly reforms the Internal Revenue Code of 1986, as amended (the “Code”). The Tax Act, among other things, includes changes to U.S. federal tax rates, including reduction of the corporate tax rate from a top marginal rate of 35% to a flat rate of 21%, limitations of the tax deduction for interest expense to 30% of adjusted earnings (except for certain small businesses), limitations of the deduction for net operating losses to 80% of current year taxable income and elimination of net operating loss carrybacks, one time taxation of offshore earnings at reduced rates regardless of whether they are repatriated, elimination of U.S. tax on foreign earnings (subject to certain important exceptions), immediate deductions for certain new investments instead of deductions for depreciation expense over time, modifying or repealing many business deductions and credits and putting into effect the migration from a “worldwide” system of taxation to a territorial system.
Failure to maintain the security of patient-related information or compliance with security requirements could damage our reputation with customers, cause us to incur substantial additional costs and become subject to litigation.
Pursuant to HIPAA, and certain similar state laws, we must comply with comprehensive privacy and security standards with respect to the use and disclosure of protected health information. If we do not comply with existing or new laws and regulations related to protecting privacy and security of personal or health information, it could be subject to monetary fines, civil penalties, or criminal sanctions. Under the HITECH amendments to HIPAA, HIPAA was expanded to require certain data breach notification, to extend certain HIPAA privacy and security standards directly to business associates, to heighten penalties for noncompliance, and enhance enforcement efforts.
We may also be required to comply with the data privacy and security laws of other countries in which it operates or from which it receives data transfers. The European Union enacted the General Data Protection Regulation (GDPR) to replace the current data protection directive, Directive 95/46/EC, which took effect May 25, 2018, and which has a broader application and enhanced penalties for noncompliance. The GDPR, which is wide-ranging in scope, governs the collection and use of personal data in the European Union and imposes operational requirements for companies that receive or process personal data of residents of the European Union that are different than those currently in place in the European Union. The GDPR will apply to our European operations and possibly to our laboratory and clinical development operations. We have implemented policies and procedures required to comply with the new EU regulations and will continue to evaluate compliance.
In March 2014, CareEvolve, BioReference’s wholly-owned connectivity subsidiary, became aware that there had been a HIPAA breach with regard to one of its servers managed at an internet service provider site called XAND, where the server was inadvertently configured so that it was accessible to the Internet for a brief period. Upon becoming aware of the matter, CareEvolve immediately took the server offline and removed all indexed files that could be located on the internet. In the meantime, an Internet data collection “robot” operated by Google, Inc. had briefly acquired data from a server and made it available to Internet searches. To the best of our knowledge, there were no known disclosures of this Patient Health
48
Information (“PHI”) to unauthorized parties. BioReference self-reported this incident to the appropriate government agency, the Office of Civil Rights (“OCR”). OCR notified BioReference that it has initiated an investigation of the breach report, and we are awaiting further discussion, investigation and action by OCR. Since March 2014, BioReference has taken meaningful steps to further improve its HIPAA and cybersecurity platform, including engaging independent and specialized IT consultants to conduct HIPAA and cybersecurity assessments, reviewing data security and internal safeguards, and continuously implementing enhanced security measures to minimize the risk of similar occurrences in the future. We have had other data and security breaches in the ordinary course and such breaches may continue to happen from time to time despite our best efforts to prevent such breaches and safeguard private information. Some of these other data and security breaches have been reported to OCR and we are awaiting discussion, investigation or action by OCR. Any action by OCR may require us to pay fines or take remedial actions that may be expensive and require the attention of management, any of which may have a material adverse effect on us and our results of operations.
We have and will continue to receive certain personal and financial information about our clients and their patients. In addition, we depend upon the secure transmission of confidential information over public networks. While we take reasonable and prudent steps to protect this protected information, a compromise in our security systems that results in client or patient personal information being obtained by unauthorized persons or our failure to comply with security requirements for financial transactions could adversely affect our reputation with our clients and result in litigation against us or the imposition of penalties, all of which may adversely impact our results of operations, financial condition and liquidity.
Failure to comply with environmental, health and safety laws and regulations, including the Federal Occupational Safety and Health Administration Act, the Needlestick Safety and Prevention Act and the Comprehensive Medical Waste Management Act, could result in fines and penalties and loss of licensure, and have a material adverse effect upon our business.
We are subject to licensing and regulation under federal, state and local laws and regulations relating to the protection of the environment and human health and safety, including laws and regulations relating to the handling, transportation and disposal of medical specimens, infectious and hazardous waste and radioactive materials, as well as regulations relating to the safety and health of laboratory employees. The Federal Occupational Safety and Health Administration has established extensive requirements relating to workplace safety for health care employers, including clinical laboratories, whose workers may be exposed to blood-borne pathogens such as HIV and the hepatitis B virus. These requirements, among other things, require work practice controls, protective clothing and equipment, training, medical follow-up, vaccinations and other measures designed to minimize exposure to, and transmission of, blood-borne pathogens. In addition, the Needlestick Safety and Prevention Act requires, among other things, that we include in our safety programs the evaluation and use of engineering controls such as safety needles if found to be effective at reducing the risk of needlestick injuries in the workplace.
Waste management is subject to federal and state regulations governing the transportation and disposal of medical waste including bodily fluids. Federal regulations require licensure of interstate transporters of medical waste. In New Jersey, we are subject to the Comprehensive Medical Waste Management Act (“CMWMA”), which requires us to register as a generator of special medical waste. All of our medical waste is disposed of by a licensed interstate hauler. The hauler provides a manifest of the disposition of the waste products as well as a certificate of incineration, which is retained by us. These records are audited by the State of New Jersey on a yearly basis. We are also subject to the Federal Hazardous Materials Transportation Law, 49 U.S.C. 5101 et seq., and the Hazardous Materials Regulations (“HMR”), 49 CFR parts 171-180. The federal government has classified hazardous medical waste as hazardous materials for the purpose of regulation. These regulations preempt state regulation, which must be “substantively the same,” meaning that “the non-federal requirement must conform “in every significant respect to the federal requirement. Editorial and other similar de minimis changes are permitted,” 49 CFR 107.202(d).
Failure to comply with such federal, state and local laws and regulations could subject us to denial of the right to conduct business, fines, criminal penalties and/or other enforcement actions, any of which could have a material adverse effect on our business. In addition, compliance with future legislation could impose additional requirements us, which may be costly.
Our failure or the failure of third-party payors or physicians to comply with ICD-10-CM Code Set, and our failure to comply with other emerging electronic transaction standards could adversely impact our business.
Compliance with the ICD-10-CM Code Set was required to be in place by October 1, 2015. We will continue our assessment of information systems, applications and processes for compliance with these requirements. Clinical laboratories are typically required to submit health care claims with diagnosis codes to third party payors. The diagnosis codes must be obtained from the ordering physician for clinical laboratory testing and from the interpreting pathologist for anatomic pathology services. Our failure or the failure of third party payors or physicians to comply with these requirements could have an adverse impact on reimbursement, days sales and cash collections.
49
Also, the failure of our IT systems to keep pace with technological advances may significantly reduce our revenues or increase our expenses. Public and private initiatives to create healthcare information technology (“HCIT”) standards and to mandate standardized clinical coding systems for the electronic exchange of clinical information, including test orders and test results, could require costly modifications to our existing HCIT systems. If we fail to adopt or delay in implementing HCIT standards, we could lose customers and business opportunities.
Failure to comply with complex federal and state laws and regulations related to submission of claims for clinical laboratory services could result in significant monetary damages and penalties and exclusion from the Medicare and Medicaid programs.
We are subject to extensive federal and state laws and regulations relating to the submission of claims for payment for clinical laboratory services, including those that relate to coverage of our services under Medicare, Medicaid and other governmental health care programs, the amounts that may be billed for our services and to whom claims for services may be submitted. These rules may also affect us in light of the practice management products that we market, to the extent that these products are considered to affect the manner in which our customers’ submit their own claims for services. Submission of our claims is particularly complex because we provide both anatomic pathology services and clinical laboratory tests, which generally are paid using different reimbursement principles. The clinical laboratory tests are often paid under a clinical laboratory fee schedule, and the anatomic pathology services are often paid under a physician fee schedule.
Our failure to comply with applicable laws and regulations could result in our inability to receive payment for our services or result in attempts by third-party payors, such as Medicare and Medicaid, to recover payments from us that have already been made. Submission of claims in violation of certain statutory or regulatory requirements can result in penalties, including substantial civil money penalties for each item or service billed to Medicare in violation of the legal requirement, and exclusion from participation in Medicare and Medicaid. Government authorities may also assert that violations of laws and regulations related to submission or causing the submission of claims violate the federal False Claims Act (“FCA”) or other laws related to fraud and abuse, including submission of claims for services that were not medically necessary. Under the FCA, whistleblower or qui tam provisions allow a private individual to bring actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In recent years, the number of suits brought by private individuals has increased dramatically and we may be subject to such suits. Violations of the FCA could result in enormous economic liability. The FCA provides that all damages are trebled, and each false claim submitted is subject to a penalty of up to $21,916. For example, we could be subject to FCA liability if it was determined that the services we provided were not medically necessary and not reimbursable, particularly if it were asserted that we contributed to the physician’s referrals of unnecessary services to us. It is also possible that the government could attempt to hold us liable under fraud and abuse laws for improper claims submitted by an entity for services that we performed if we were found to have knowingly participated in the arrangement that resulted in submission of the improper claims.
Changes in regulation and policies, including increasing downward pressure on health care reimbursement, may adversely affect reimbursement for diagnostic services and could have a material adverse impact on our business.
Reimbursement levels for health care services are subject to continuous and often unexpected changes in policies, and we face a variety of efforts by government payors to reduce utilization and reimbursement for diagnostic testing services. Changes in governmental reimbursement may result from statutory and regulatory changes, retroactive rate adjustments, administrative rulings, competitive bidding initiatives, and other policy changes.
The U.S. Congress has considered, at least yearly in conjunction with budgetary legislation, changes to one or both of the Medicare fee schedules under which we receive reimbursement, which include the physician fee schedule for anatomical pathology services, and the clinical laboratory fee schedule for our clinical laboratory services. For example, currently there is no copayment or coinsurance required for clinical laboratory services, although there is for our services that are paid under the physician fee schedule. However, Congress has periodically considered imposing a 20 percent coinsurance on laboratory services. If enacted, this would require us to attempt to collect this amount from patients, although in many cases the costs of collection would exceed the amount actually received. In April 2015, changes to the physician fee schedule were enacted under the Medicare Access and CHIP Reauthorization Act of 2015 (“MACRA”).
Our reimbursement for our pathology services is paid primarily under the physician fee schedule of Medicare and Medicaid. Historically, the physician fee schedule was governed by a complex formula, referred to as the Sustainable Growth Rate, or SGR. However, in April 2015, MACRA was passed, which permanently replaces the SGR formula with a value-based payment system. The passage of MACRA also repealed the 21.1% reduction of the physician fee schedule that was scheduled for April 1, 2015. Under MACRA, the physician fee schedule conversion factor increases of 0.5% from July 1, 2015 to December 31, 2015, and 0.5% in each of years 2016-2019, followed by 0.0% updates for 2020-2025. Subsequent years will
50
vary based on participation in alternative payment models. Beginning in 2019, rates were adjusted under the new Merit-based Incentive Payment System.
The Center for Medicare and Medicaid Services (“CMS”) pays laboratories on the basis of a fee schedule that is reviewed and re-calculated on an annual basis. CMS may change the fee schedule upward or downward on billing codes that we submit for reimbursement on a regular basis. Our revenue and business may be adversely affected if the reimbursement rates associated with such codes are reduced. Even when reimbursement rates are not reduced, policy changes add to our costs by increasing the complexity and volume of administrative requirements. Medicaid reimbursement, which varies by state, is also subject to administrative and billing requirements and budget pressures. Recently, state budget pressures have caused states to consider several policy changes that may impact our financial condition and results of operations, such as delaying payments, reducing reimbursement, restricting coverage eligibility and service coverage, and imposing taxes on our services.
CMS has changed or discussed making changes to certain types of reimbursement which could affect our rate of reimbursement. Certain cases are comprised of both a technical component (“TC”) and a professional component (“PC”). In certain specified areas of testing, primarily in the area of anatomic pathology, CMS has determined that some providers have over-utilized these testing procedures and CMS has introduced changes in reimbursement policies to discourage over-utilization. We are always subject to review by CMS and cannot be certain that CMS won’t interpret our practices differently than we do.
Third party payors are increasingly challenging established prices, and new products that are more expensive than existing treatments may have difficulty finding ready acceptance unless there is a clear therapeutic benefit. On April 1, 2014, the Protecting Access to Medicare Act of 2014 (“PAMA”) was enacted into law. Under PAMA, Medicare payment for clinical diagnostic laboratory tests is established by calculating a weighted mean of private payor rates. Effective January 1, 2018, clinical laboratory fee schedule rates will be based on weighted median private payor rates as required by PAMA. Even though the permitted annual decrease are capped through 2023, the cap does not apply to new tests or new advanced diagnostic tests. We cannot assure you that any of our products will be considered cost effective, or that reimbursement will be available or sufficient to allow us to sell them competitively and profitably.
The federal government is faced with significant economic decisions in the coming years. Some solutions being offered in the government could substantially change the way laboratory testing is reimbursed by government entities. We cannot be certain what or how any such government changes may affect our business.
Medicare legislation and future legislative or regulatory reform of the health care system may affect our ability to sell our products profitably.
In the U.S., there have been a number of legislative and regulatory initiatives, at both the federal and state government levels, to change the healthcare system in ways that, if approved, could affect our ability to sell our products and provide our laboratory services profitably. As such, we cannot assure you that reimbursement payments under governmental and private third party payor programs will remain at levels comparable to present levels or will be sufficient to cover the costs allocable to patients eligible for reimbursement under these programs. Any changes that lower reimbursement rates under Medicare, Medicaid or private payor programs could negatively affect our business.
Most significantly, on March 23, 2010, President Obama signed into law both the Affordable Care Act and the reconciliation law known as Health Care and Education Affordability Reconciliation Act (the “Reconciliation Act”) and, combined we refer to both Acts as the “2010 Health Care Reform Legislation.” The constitutionality of the 2010 Health Care Reform Legislation was confirmed on June 28, 2012 by the Supreme Court of the U. S. However, as discussed in further detail below, the current Presidential administration has attempted to repeal and replace the 2010 Health Care Reform Legislation.
Beyond coverage and reimbursement changes, the 2010 Health Care Reform Legislation subjects manufacturers of medical devices to an excise tax of 2.3% on certain U.S. sales of medical devices beginning in January 2013. However, a two-year moratorium on the tax was issued on December 18, 2015. The moratorium was extended for an additional two-year period on January 22, 2018. As such, the excise tax does not apply to sales in 2016 through 2019. The return of the tax in January 2020 will likely increase our expense in the future.
Additionally, the 2010 Health Care Reform Legislation included significant fraud and abuse measures, including (i) required disclosures under the Open Payments Program (which implements the requirements of the Physician Payments Sunshine Act), which in conjunction with its implementing regulations, requires certain manufacturers of certain drugs, biologics, and devices that are reimbursed by Medicare and Medicaid to report annually certain payments or “transfers of value” provided to physicians and teaching hospitals and to report annually ownership and investment interests held by physicians and their immediate family members during the preceding calendar year, (ii) lower thresholds for violations, and (iii) increasing potential penalties for such violations. Federal funding available for combating health care fraud and abuse
51
generally has increased. Many of the laws and regulations applicable to our business, particularly those relating to billing and reimbursement of tests and those relating to relationships with physicians, hospitals and patients, contain language that has not been interpreted by courts. We must rely on our interpretation of these laws and regulations based on the advice of our counsel and regulatory or law enforcement authorities may not agree with our interpretation of these laws and regulations and may seek to enforce legal remedies or penalties against us for violations. From time to time we may need to change our operations, particularly pricing or billing practices, in response to changing interpretations of these laws and regulations or regulatory or judicial determinations with respect to these laws and regulations. These occurrences, regardless of their outcome, could damage our reputation and harm important business relationships that we have with healthcare providers, payors and others. Furthermore, if a regulatory or judicial authority finds that we have not complied with applicable laws and regulations, we could be required to refund amounts that were billed and collected in violation of such laws and regulations. In addition, we may voluntarily refund amounts that were alleged to have been billed and collected in violation of applicable laws and regulations. In either case, we could suffer civil and criminal damages, fines and penalties, exclusion from participation in governmental healthcare programs and the loss of licenses, certificates and authorizations necessary to operate our business, as well as incur liabilities from third-party claims, all of which could harm our operating results and financial condition. Moreover, regardless of the outcome, if we or physicians or other third parties with whom we do business are investigated by a regulatory or law enforcement authority we could incur substantial costs, including legal fees, and our management may be required to divert a substantial amount of time to an investigation.
Prior to the 2016 U.S. elections (including the current Presidential administration), regulations under the 2010 Health Care Reform Legislation were expected to continue being drafted, released and finalized throughout the next several years. In 2017, the President and members of Congress sought to repeal and replace the 2010 Health Care Reform Legislation. It is uncertain whether such repeal and replacement legislation will be enacted into law, and if enacted, what the impact might be on our business. It is also uncertain whether regulatory changes to the implementation of the 2010 Health Care Reform Legislation will restrict patient access to affordable insurance and impact their access to novel, biosimilar and complex generic products. The full effects of any repeal and replacement of the 2010 Health Care Reform Legislation, or regulatory changes to its implementation, cannot be known until a new law is enacted or existing law is implemented through regulations or guidance issued by the CMS and other federal and state health care agencies. Because of the continued uncertainty about the implementation of the 2010 Health Care Reform Legislation, including the potential for further legal challenges or repeal of that legislation, we cannot quantify or predict with any certainty the likely impact of the 2010 Health Care Reform Legislation or its repeal on our business model, prospects, financial condition or results of operations. We also anticipate that Congress, state legislatures, and third-party payors may continue to review and assess alternative healthcare delivery and payment systems and may in the future propose and adopt legislation or policy changes or implementations effecting additional fundamental changes in the healthcare delivery system. In addition, litigation may prevent some or all of the legislation from taking effect. We cannot assure you as to the ultimate content, timing, or effect of changes, nor is it possible at this time to estimate the impact of any such potential legislation.
To enhance compliance with applicable health care laws, and mitigate potential liability in the event of noncompliance, regulatory authorities, such as the U. S. Health and Human Services Department Office of Inspector General (the “OIG”), have recommended the adoption and implementation of a comprehensive health care compliance program that generally contains the elements of an effective compliance and ethics program described in Section 8B2.1 of the U. S. Sentencing Commission Guidelines Manual, and for many years the OIG has made available a model compliance program targeted to the clinical laboratory industry. In addition, certain states, such as New York, require that health care providers, such as clinical laboratories, that engage in substantial business under the state Medicaid program have a compliance program that generally adheres to the standards set forth in the Model Compliance Program. Also, under the 2010 Health Care Reform Legislation, the U.S. Department of Health and Human Services, or HHS, requires suppliers, such as us, to adopt, as a condition of Medicare participation, compliance programs that meet a core set of requirements. While we have adopted U.S. healthcare compliance and ethics programs that generally incorporate the OIG’s recommendations and train our employees in such compliance, having such a program can be no assurance that we will avoid any compliance issues.
RISKS RELATED TO INTERNATIONAL OPERATIONS
Failure to obtain regulatory approval outside the U.S. will prevent us from marketing our products and product candidates abroad.
We intend to market certain of our products and product candidates in non-U.S. markets. In order to market our products and product candidates in the European Union and many other non-U.S. jurisdictions, we must obtain separate regulatory approvals. We have had limited interactions with non-U.S. regulatory authorities, the approval procedures vary among countries and can involve additional testing, and the time required to obtain approval may differ from that required to obtain FDA approval or clearance. Approval or clearance by the FDA does not ensure approval by regulatory authorities in other countries, and approval by one or more non-U.S. regulatory authority does not ensure approval by other regulatory authorities
52
in other countries or by the FDA. The non-U.S. regulatory approval process may include all of the risks associated with obtaining FDA approval or clearance. We may not obtain non-U.S. regulatory approvals on a timely basis, if at all. We may not be able to file for non-U.S. regulatory approvals and may not receive necessary approvals to commercialize our products and product candidates in any market, which would have a material adverse effect on our business, results of operations and financial condition.
Non-U.S. governments often impose strict price controls, which may adversely affect our future profitability.
We intend to seek approval to market certain of our products and product candidates in both the U.S. and in non‑U.S. jurisdictions. If we obtain approval in one or more non-U.S. jurisdictions, we will be subject to rules and regulations in those jurisdictions relating to our product. In some countries, particularly countries of the European Union, each of which has developed its own rules and regulations, pricing is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a drug or medical device candidate. To obtain reimbursement or pricing approval in some countries, we may be required to conduct a clinical trial that compares the cost-effectiveness of our product and product candidates to other available products. If reimbursement of our products and product candidates is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, we may be unable to generate revenues and achieve or sustain profitability, which would have a material adverse effect on our business, results of operations and financial condition.
Potential political, economic and military instability in the State of Israel, where we have office, laboratory and manufacturing operations, may adversely affect our results of operations.
We maintain office, laboratory and manufacturing facilities in the State of Israel. Political, economic and military conditions in Israel may directly affect our ability to conduct business. Since the State of Israel was established in 1948, a number of armed conflicts have occurred between Israel and its neighbors. Any hostilities involving Israel or the interruption or curtailment of trade between Israel and its present trading partners, or a significant downturn in the economic or financial condition of Israel, could affect adversely our operations. Ongoing and revived hostilities or other Israeli political or economic factors could harm our operations and product development and cause our revenues to decrease.
Due to the international scope of our business activities, our results of operations may be significantly affected by currency fluctuations.
We derive a significant portion of our consolidated net revenues from international sales, subjecting us to risks relating to fluctuations in currency exchange rates. Currency variations can adversely affect margins on sales of our products in countries outside of the U.S. and margins on sales of products that include components obtained from suppliers located outside of the U.S. Through our subsidiaries, we operate in a wide variety of jurisdictions. Certain countries in which we operate or may operate have experienced geopolitical instability, economic problems and other uncertainties from time to time. To the extent that world events or economic conditions negatively affect our future sales to customers in these and other regions of the world, or the collectability of receivables, our future results of operations, liquidity and financial condition may be adversely affected. We may manage exposures arising in the normal course of business related to fluctuations in foreign currency exchange rates by entering into offsetting positions through the use of foreign exchange forward contracts. Certain firmly committed transactions are hedged with foreign exchange forward contracts whereby exchange rates change, gains and losses on the exposed transactions are partially offset by gains and losses related to the hedging contracts. However, our subsidiaries receive their income and pay their expenses primarily in their local currencies. To the extent that transactions of these subsidiaries are settled in their local currencies, a devaluation of those currencies versus the U.S. dollar could reduce the contribution from these subsidiaries to our consolidated results of operations as reported in U.S. dollars. For financial reporting purposes, such depreciation will negatively affect our reported results of operations since earnings denominated in foreign currencies would be converted to U.S. dollars at a decreased value. While we have employed economic cash flow and fair value hedges to minimize the risks associated with these exchange rate fluctuations, the hedging activities may be ineffective or may not offset more than a portion of the adverse financial impact resulting from currency variations. Accordingly, we cannot assure you that fluctuations in the values of the currencies of countries in which we operate will not materially adversely affect our future results of operations.
We may be exposed to liabilities under the Foreign Corrupt Practices Act, and any determination that we violated the Foreign Corrupt Practices Act could have a material adverse effect on our business.
We are subject to the Foreign Corrupt Practice Act (“FCPA”) and other laws that prohibit U.S. companies or their agents and employees from providing anything of value to a foreign official or political party for the purposes of influencing any act or decision of these individuals in their official capacity to help obtain or retain business, direct business to any person or corporate entity or obtain any unfair advantage. We have operations and agreements with third parties and we generate sales internationally. Our international activities create the risk of unauthorized and illegal payments or offers of payments by our
53
employees, consultants, sales agents or distributors, even though they may not always be subject to our control. We discourage these practices by our employees and agents. However, our existing safeguards and any future improvements may prove to be less than effective, and our employees, consultants, sales agents or distributors may engage in conduct for which we might be held responsible. Any failure by us to adopt appropriate compliance procedures and ensure that our employees and agents comply with the FCPA and applicable laws and regulations in foreign jurisdictions could result in substantial penalties or restrictions on our ability to conduct business in certain foreign jurisdictions.
Violations of the FCPA may result in severe criminal or civil sanctions, and we may be subject to other liabilities, which could negatively affect our business, operating results and financial condition. In addition, the U.S. government may seek to hold our Company liable for successor liability FCPA violations committed by companies in which we invest or that we acquire.
We are subject to risks associated with doing business globally.
Our operations, both within and outside the U.S., are subject to risks inherent in conducting business globally and under the laws, regulations and customs of various jurisdictions and geographies. These risks differ in some respects from those associated with our U.S. business and our exposure to such risks may increase if our international business continues to grow. These risks include fluctuations in currency exchange rates, changes in exchange controls, loss of business in government tenders that are held annually in many cases, nationalization, increasingly complex labor environments, expropriation and other governmental actions, changes in taxation, including legislative changes in U.S. and international taxation of income earned outside of the U.S., importation limitations, export control restrictions, violations of U.S. or local laws, including the FCPA, dependence on a few government entities as customers, pricing restrictions, economic destabilization, political and economic instability and disruption or destruction in a significant geographic region - due to the location of manufacturing facilities, distribution facilities or customers - regardless of cause, including war, terrorism, riot, civil insurrection or social unrest, or natural or man-made disasters, including famine, flood, fire, earthquake, storm or disease.
Our international business is subject to both U.S. and foreign laws and regulations, including, without limitation, regulations relating to import-export controls, technology transfer restrictions, repatriation of earnings, data privacy and protection, investment, exchange rates and controls, the FCPA and other anti-corruption laws, the anti-boycott provisions of the U.S. Export Administration Act, labor and employment, works councils and other labor groups, taxes, environment, security restrictions, intellectual property, changes in taxation, including legislative changes in U.S. and international taxation of income earned outside of the U.S., handling of regulated substances, and other commercial activities. Failure by us, our employees, affiliates, partners or others with whom we work to comply with these laws and regulations could result in administrative, civil or criminal liabilities. New regulations and requirements, or changes to existing ones in the various countries in which we operate can significantly increase our costs and risks of doing business internationally. Failure to comply with the laws and regulations that affect our global operations, could have an adverse effect on our business, financial condition or results of operations.
Changes in regulations, political leadership and environment, or security risks may dramatically affect our ability to conduct or continue to conduct business in international markets. Our international business may also be impacted by changes in foreign national policies and priorities, which may be influenced by changes in the environment, geopolitical uncertainties, government budgets, and economic and political factors more generally, any of which could impact funding for programs or delay purchasing decisions or customer payments. We also could be affected by the legal, regulatory and economic impacts of Britain’s exit from the European Union, the impact of which is not known at this time. The occurrence and impact of these factors is difficult to predict, but one or more of them could have a material adverse effect on our financial position, results of operations and/or cash flows.
RISKS RELATED TO ACQUISITIONS AND INVESTMENTS
Acquisitions, investments and strategic alliances that we have made or may make in the future may use significant resources, result in disruptions to our business or distractions of our management, may not proceed as planned, and could expose us to unforeseen liabilities. We intend to continue to expand our business through the acquisition of, investments in and strategic alliances with companies, technologies, products and services. Acquisitions, investments and strategic alliances involve a number of special problems and risks, including, but not limited to:
• |
difficulty integrating acquired technologies, products, services, operations, and personnel with the existing businesses; |
• |
diversion of management’s attention in connection with both negotiating the acquisitions and integrating the businesses; |
• |
strain on managerial and operational resources as management tries to oversee larger operations and investments; |
54
• |
difficulty implementing and maintaining effective internal control over financial reporting at businesses that we acquire or invest in, particularly if they are not located near our existing operations; |
• |
exposure to unforeseen liabilities of acquired companies or companies in which we invest; |
• |
potential costly and time-consuming litigation, including stockholder lawsuits; |
• |
potential issuance of securities to equity holders of the company being acquired with rights that are superior to the rights of holders of our Common Stock, or which may have a dilutive effect on our stockholders; |
• |
the need to incur additional debt or use cash; and |
• |
the requirement to record potentially significant additional future operating costs for the amortization of intangible assets. |
As a result of these or other problems and risks, businesses we acquire or invest in may not produce the revenues, earnings, or business synergies that we anticipated, and acquired products, services, or technologies might not perform as we expected. As a result, we may incur higher costs and realize lower revenues than we had anticipated. We may not be able to successfully address these problems and we cannot assure you that the acquisitions or investments will be successfully identified and completed or that, if completed, the acquired businesses, investments, products, services, or technologies will generate sufficient revenue to offset the associated costs or other negative effects on our business.
Any of these risks can be greater if an acquisition or investment is large relative to our size. Failure to manage effectively our growth through acquisitions could adversely affect our growth prospects, business, results of operations, financial condition and cash flows.
We may fail to realize the anticipated benefits of the mergers with BioReference, Transition Therapeutics, and other acquisitions.
The success of the mergers will depend on, among other things, our ability to combine our business with that of BioReference and Transition in a manner that facilitates growth opportunities and realizes synergies and cost savings. We believe that the mergers will provide an opportunity for revenue growth. However, we must successfully combine our business with that of BioReference and Transition in a manner that permits these benefits to be realized. In addition, we must achieve the anticipated growth and cost savings without adversely affecting current revenues and investments in future growth. If we are not able to successfully achieve these objectives, the anticipated benefits of the mergers may not be realized fully, or at all, or may take longer to realize than expected.
The failure to integrate successfully the business and operations of BioReference in the expected time frame may adversely affect our future results.
Historically, we and BioReference have operated as independent companies. There can be no assurances that our and BioReference’s businesses can be integrated successfully. It is possible that the integration process could result in the loss of our or BioReference’s key employees, the loss of customers, the disruption of either company’s or both companies’ ongoing businesses or in unexpected integration issues, higher than expected integration costs and an overall post-completion integration process that takes longer than originally anticipated. Specifically, the following issues, among others, must be addressed in integrating our operations with BioReference’s operations in order to realize the anticipated benefits of the merger so we perform as expected:
• |
combining the companies’ operations and corporate functions, as well as obtaining anticipated synergies; |
• |
combining our business with BioReference’s business and meeting the capital requirements of the combined company, in a manner that permits us to achieve the cost savings or revenue synergies anticipated to result from the merger, the failure of which would result in the anticipated benefits of the merger not being realized in the time frame currently anticipated or at all; |
• |
integrating the companies’ technologies; |
• |
integrating and unifying the offerings and services available to customers; |
• |
identifying and eliminating redundant and underperforming functions and assets; |
• |
harmonizing and/or addressing differences in the companies’ operating practices, employee development and compensation programs, internal controls and other policies, procedures and processes; |
• |
maintaining existing agreements with customers, distributors, providers and vendors and avoiding delays in entering into new agreements with prospective customers, distributors, providers and vendors; |
55
• |
addressing possible differences in business backgrounds, corporate cultures and management philosophies; |
• |
consolidating the companies’ administrative and information technology infrastructure; |
• |
coordinating distribution and marketing efforts; |
• |
managing the movement of certain positions to different locations; |
• |
coordinating geographically dispersed organizations; and |
• |
effecting actions that may be required in connection with obtaining regulatory approvals. |
In addition, at times the attention of our management and resources may be focused on the integration of the businesses of the two companies and diverted from day-to-day business operations, which may disrupt our ongoing business.
Funding may not be available for us to continue to make acquisitions, investments and strategic alliances in order to grow our business.
We have made and anticipate that we may continue to make acquisitions, investments and strategic alliances with complementary businesses, technologies, products and services to expand our business. Our growth plans rely, in part, on the successful completion of future acquisitions. At any particular time, we may need to raise substantial additional capital or to issue additional equity to finance such acquisitions, investments, and strategic alliances. There is no assurance that we will be able to secure additional funding on acceptable terms, or at all, or obtain the stockholder approvals necessary to issue additional equity to finance such acquisitions, investments, and strategic alliances. If we are unsuccessful in obtaining the financing, our business would be adversely impacted.
We have a large amount of goodwill and other intangible assets on our balance sheet that are subject to periodic impairment evaluations. As a result of the annual impairment test for the year ended December 31, 2018, we wrote down goodwill and intangible assets and we may have similar charges in the future, which would have a material adverse impact on our financial condition and results of operations.
We have a large amount of goodwill and other intangible assets and we are required to perform an annual, or in certain situations a more frequent, assessment for possible impairment for accounting purposes. At December 31, 2018, we have goodwill and other intangible assets of $2.0 billion, or approximately 79% of our total assets. Goodwill is tested at least annually for impairment or when events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, by assessing qualitative factors or performing a quantitative analysis in determining whether it is more likely than not that its fair value exceeds the carrying value. Examples of qualitative factors include our share price, our financial performance compared to budgets, long-term financial plans, macroeconomic, industry and market conditions as well as the substantial excess of fair value over the carrying value of net assets from the annual impairment test previously performed. The estimated fair value of a reporting unit is highly sensitive to changes in projections and assumptions; therefore, in some instances, changes in these assumptions could potentially lead to impairment. We perform sensitivity analyses around our assumptions in order to assess the reasonableness of the assumptions and the results of our testing. Ultimately, future potential changes in these assumptions may impact the estimated fair value of a reporting unit and cause the fair value of the reporting unit to be below its carrying value. Intangible assets are tested for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, although in-process research and development (“IPR&D”) is required to be tested at least annually until the project is completed or abandoned. Upon obtaining regulatory approval, the IPR&D asset is then accounted for as a finite-lived intangible asset and amortized on a straight-line basis over its estimated useful life. If the project is abandoned, the IPR&D asset is charged to expense. IPR&D is tested for impairment by assessing qualitative factors or performing a quantitative analysis in determining whether it is more likely than not that its fair value exceeds the carrying amount. If the carrying amount of the IPR&D exceeds its fair value, an impairment loss will be recognized in an amount equal to that excess.
For the year ended December 31, 2018, we recorded asset impairment charges of $21.8 million, which were related to an impairment charge of $10.1 million to write our IPR&D assets for Alpharen and OPK88004 down to their estimated fair value and a goodwill impairment charge of $11.7 million to write the carrying amount of the FineTech reporting unit down to its estimated fair value. We also took a charge of $2.9 million to write down our investment in InCellDX, Inc. to its fair value as of December 31, 2018.
There can be no assurance that future reviews of our goodwill and other intangible assets will not result in additional impairment charges. A significant write down of goodwill and/or other intangible assets would have a material adverse effect on our reported results of operations and net worth and the trading price of our securities.
RISKS RELATED TO OWNERSHIP OF OUR COMMON STOCK
56
The trading prices of our securities may fluctuate significantly.
The trading prices of our securities may fluctuate significantly in response to numerous factors, some of which are beyond our control, such as:
• |
the announcement of new products or product enhancements by us or our competitors; |
• |
results of our clinical trials and other development efforts; |
• |
developments concerning intellectual property rights and regulatory approvals; |
• |
variations in our and our competitors’ results of operations; |
• |
changes in earnings estimates or recommendations by securities analysts, if our common stock is covered by analysts; |
• |
developments in the biotechnology, pharmaceutical, diagnostic and medical device industry; |
• |
the announcement and/or commencement and/or settlement of lawsuits or similar claims against us or any of our officers, directors and affiliates; |
• |
the results of product liability or intellectual property lawsuits; |
• |
future issuances of our common stock or other securities, including debt; |
• |
purchases and sales of our common stock by our officers, directors or affiliates; |
• |
the addition or departure of key personnel; |
• |
announcements by us or our competitors of acquisitions, investments or strategic alliances; and |
• |
general market conditions and other factors, including factors unrelated to our operating performance. |
Further, the securities market in general, and the market for biotechnology, pharmaceutical, diagnostic and medical device companies in particular, has experienced extreme price and volume fluctuations in recent years. Continued market fluctuations could result in extreme volatility in the trading prices of our securities, which could cause a decline in the value of our securities.
Directors, executive officers, principal stockholders and affiliated entities own a substantial amount of our capital stock, and they may make decisions that you do not consider to be in the best interests of our stockholders.
As of January 28, 2019, our directors, executive officers, principal stockholders and affiliated entities beneficially owned, in the aggregate, approximately 44.2% of our outstanding voting securities. Phillip Frost, M.D., our Chairman and CEO, is deemed to beneficially own, in the aggregate, approximately 36.9% of our common stock as of January 28, 2019. As a result, Dr. Frost, acting with other members of management, would have the ability to significantly impact the election of our Board of Directors, the adoption or amendment of provisions in our Certificate of Incorporation, the approval of mergers and other significant corporate transactions and the outcome of issues requiring approval by our stockholders. This concentration of ownership may also have the effect of delaying or preventing a change in control of our company that may be favored by other stockholders. This could prevent transactions in which holders of our securities might otherwise recover a premium for their securities over current market prices.
A significant short position in our stock could have a substantial impact on the trading price of our stock.
Historically, there has been a significant “short” position in our common stock. As of January 31, 2019, investors held a short position of approximately 59,146,878 million shares of our common stock which represented approximately 9.6% of our outstanding common stock. The anticipated downward pressure on our stock price due to actual or anticipated sales of our stock by some institutions or individuals who engage in short sales of our common stock could cause our stock price to decline. Such stock price decrease could encourage further short-sales that could place additional downward pressure on our stock price. This could lead to further increases in the already large short position in our common stock and cause volatility in our stock price.
The volatility of our stock may cause the value of a stockholder’s investment to decline rapidly. Additionally, if our stock price declines, it may be more difficult for us to raise capital and may have other adverse effects on our business.
Failure to maintain effective internal controls in accordance with Section 404 of the Sarbanes-Oxley Act, including with respect to companies we acquire, could have a material adverse effect on our business and operating results. In addition, current and potential stockholders could lose confidence in our financial reporting, which could have a material adverse
57
effect on the price of our Common Stock.
Section 404 of the Sarbanes-Oxley Act of 2002 requires annual management assessments of the effectiveness of our internal control over financial reporting and a report by our independent registered public accounting firm on the effectiveness of internal control over financial reporting as of year end. We are required to report, among other things, control deficiencies that constitute material weaknesses or changes in internal control that, or that are reasonably likely to, materially affect internal control over financial reporting. A “material weakness” is a significant deficiency or combination of significant deficiencies that results in more than a remote likelihood that a material misstatement of the annual or interim financial statements will not be prevented or detected.
We have identified and remediated control deficiencies in the past, and we cannot assure you that we will at all times in the future be able to report that our internal controls are effective. In addition, material weaknesses in the design and operation of the internal control over financial reporting of companies that we acquire could have a material adverse effect on our business and operating results. Our acquisition of BioReference and Transition Therapeutics and possible future acquisitions may increase this risk by expanding the scope and nature of operations over which we must develop and maintain internal control over financial reporting. If we cannot provide reliable financial reports or prevent fraud, our results of operation could be harmed. Our failure to maintain the effective internal control over financial reporting could cause the cost related to remediation to increase and could cause our stock price to decline. In addition, we may not be able to accurately report our financial results, may be subject to regulatory sanction, and investors may lose confidence in our financial statements.
Compliance with changing regulations concerning corporate governance and public disclosure may result in additional expenses.
There have been changing laws, regulations, and standards relating to corporate governance and public disclosure, including the Sarbanes-Oxley Act of 2002, the Dodd-Frank Act, regulations promulgated by the Securities and Exchange Commission and rules promulgated by the Nasdaq Global Select Market and the other national securities exchanges. These new or changed laws, regulations, and standards are subject to varying interpretations in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies, which could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. As a result, our efforts to comply with evolving laws, regulations, and standards are likely to continue to result in increased general and administrative expenses and a diversion of management time and attention from revenue-generating activities to compliance activities. Our board members, Chief Executive Officer, Chief Financial Officer, and Principal Accounting Officer could face an increased risk of personal liability in connection with the performance of their duties. As a result, we may have difficulty attracting and retaining qualified board members and executive officers, which could harm our business. If our efforts to comply with new or changed laws, regulations, and standards differ from the activities intended by regulatory or governing bodies, we could be subject to liability under applicable laws or our reputation may be harmed, which could materially adversely affect our business, results of operations and financial condition.
ITEM 1B. UNRESOLVED STAFF COMMENTS.
None.
ITEM 2. PROPERTIES.
Our principal corporate office is located at 4400 Biscayne Blvd, Miami, Florida. We lease this space from Frost Real Estate Holdings, LLC (“Frost Real Estate”), an entity which is controlled by Dr. Phillip Frost, our Chairman of the Board and Chief Executive Officer. Pursuant to the lease agreement with Frost Real Estate Holdings, we lease approximately 29,500 square feet, which encompasses space for our corporate offices and administrative services.
The table below summarizes certain information as to our significant physical properties as of December 31, 2018:
58
Location |
Segment and Purpose |
Type of Occupancy |
||
Miami, FL |
Diagnostics & Pharmaceutical: Corporate Headquarters |
Leased |
||
Elmwood Park, NJ |
Diagnostics: Main Laboratory |
Leased |
||
Gaithersburg, MD |
Diagnostics: Genetics Laboratory |
Leased |
||
Kiryat Gat, Israel |
Pharmaceutical: Research and Development, CTP |
Leased |
||
Woburn, MA |
Diagnostics |
Leased |
||
Nesher, Israel |
Pharmaceuticals: API Manufacturing |
Leased |
||
Guadalajara, Mexico |
Pharmaceuticals: Pharmaceutical Manufacturing |
Owned |
||
Banyoles, Spain |
Pharmaceuticals: Pharmaceutical Manufacturing |
Owned |
||
Palol de Revardit, Spain |
Warehouse |
Leased |
||
Barcelona, Spain |
Pharmaceuticals: Research and Development |
Leased |
||
Waterford, Ireland |
Pharmaceuticals: Pharmaceutical Manufacturing |
Leased |
||
Santiago, Chile |
Pharmaceuticals: Office; Warehouse |
Leased |
||
ITEM 3. LEGAL PROCEEDINGS.
We are involved from time to time in various claims and legal actions arising in the ordinary course of business.
As previously reported, in April 2017, the Civil Division of the U. S. Attorney’s Office for the Southern District of New York (the “SDNY”) informed BioReference that it believes that, from 2006 to the present, BioReference had, in violation of the False Claims Act, improperly billed Medicare and TRICARE (both are federal government health care programs) for clinical laboratory services provided to hospital inpatient beneficiaries at certain hospitals. BioReference is reviewing and assessing the allegations made by the SDNY, and, at this point, BioReference has not determined whether there is any merit to the SDNY’s claims nor can it determine the extent of any potential liability. While management cannot predict the outcome of these matters at this time, the ultimate outcome could be material to our business, financial condition, results of operations, and cash flows.
As previously disclosed, on September 7, 2018, the Securities and Exchange Commission (“SEC”) filed a lawsuit in the Southern District of New York (the “SEC Complaint”) against a number of individuals and entities (the “Defendants”), including the Company and its CEO and Chairman, Phillip Frost (“Dr. Frost”). The SEC alleged, among other things, that the Company (i) aided and abetted an illegal “pump and dump” scheme perpetrated by a number of the Defendants, and (ii) failed to file required Schedules 13D or 13G with the SEC. On December 27, 2018, the Company announced that the Company and Dr. Frost entered into settlement agreements with the SEC, resolving the SEC Complaint. Pursuant to the settlement, and without admitting or denying any of the allegations of the Complaint, the Company is enjoined from violating Section 13(d) of the Exchange Act and paid a $100,000 penalty. Liability under Section 13(d) can be established without any showing of wrongful intent or negligence. The settlement was approved by the Court in January 2019.
The Company also agreed to, within certain stipulated time periods: (i) establish a Management Investment Committee (“MIC”) that will make recommendations to an Independent Investment Committee (“IIC”) of the Board of Directors in connection with existing and future strategic minority investments; and (ii) retain an Independent Compliance Consultant (“ICC”) to (a) advise the Company on whether filings pursuant to Section 13(d) for previous strategic investments made at the suggestion of or in tandem with Dr. Frost should be amended or made to reflect group membership with Dr. Frost and his related entities; (b) review the Company’s existing policies and procedures relating to compliance with Section 13(d) of the Exchange Act; and (c) review the independence of the MIC and IIC of the Board of Directors solely for purposes of the handling of strategic minority investments. The ICC will report its findings to the SEC within fifteen (15) days of completion of its work, and the Company will certify its compliance with these undertakings in writing.
Under the terms of the settlement between the SEC and Dr. Frost, and without admitting or denying any of the allegations in the Complaint, Dr. Frost agreed to injunctions from violations of Sections 5(a) and (c) and 17(a)(2) of the Securities Act, claims which may be satisfied by strict liability and negligence, respectively, and Section 13(d) of the Exchange Act, also a strict liability claim; to pay approximately $5.5 million in penalty, disgorgement and pre-judgment interest, which has been paid; and to be prohibited, with certain exceptions, from trading in penny stocks.
59
Following the SEC’s announcement of the SEC Complaint, a number of class actions and derivative suits were filed concerning the allegations in the SEC Complaint and related matters.
On or about September 12, 2018, Jason Kerznowski (“Kerznowski”), a purported stockholder, filed a putative class action lawsuit in the United States District Court for the District of New Jersey against the Company and certain of its current and former executive officers (the “Kerznowski Lawsuit”). This lawsuit was brought by Kerznowski both individually and on behalf of a putative class of the Company’s stockholders, claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Kerznowski Lawsuit seeks to declare the action to be a class action and certify Kerznowski as the class representative, monetary damages, including prejudgment and post judgment interest, an award of reasonable attorneys’ fees, expert fees, and other costs, and such other relief as the Court may deem just and proper. On February 4, 2019, the United States District Court for the District of New Jersey appointed the Amitim Funds as lead plaintiffs and transferred the action to the United States District Court for the Southern District of Florida.
On or about September 13, 2018, Idan Sharon filed an Application for Approval of a Class Action in the Tel Aviv Israel District Court against the Company and certain of its current and former executive officers, and certain members of its Board of Directors (the “Sharon Claim”). This application was filed by a purported stockholder, both individually and on behalf of a putative class of the Company’s stockholders, claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Sharon Claim seeks to declare the action to be a class action and monetary damages.
On or about September 14, 2018, Charles Steinberg (“Steinberg”), a purported stockholder, filed a putative class action lawsuit in the United States District Court for the Southern District of Florida against the Company and certain of its current and former executive officers (the “Steinberg Lawsuit”). This lawsuit was brought by Steinberg both individually and on behalf of a putative class of the Company’s stockholders claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Steinberg Lawsuit seeks to declare the action to be a class action, monetary damages, including prejudgment and post judgment interest, an award of reasonable attorneys’ fees and expert fees and other costs, and such additional or different relief as the interests of law or equity may require.
On or about September 16, 2018, Dalia Avraham filed an Application for Approval of a Class Action in the Tel Aviv Israel District Court against the Company and Dr. Frost. This application was filed by a purported stockholder, both individually and on behalf of a putative class of the Company’s stockholders (the “Avraham Claim”). The Avraham Claim alleges a negligent and/or deliberate act related to the trade of the Company’s shares on the Tel Aviv Stock Exchange (“TASE”) which was intended to or which in fact caused damage to the Company’s investors based on the Company’s decision to delist from TASE in April 2018 and its subsequent decision to continue to be listed on TASE. The Avraham Claim seeks to declare the action to be a class action and an estimated NIS 20 million in damages.
On or about September 17, 2018, Adsport, Inc. (“Adsport”), a purported stockholder, filed a putative class action lawsuit in the United States District Court for the Southern District of New York against the Company and Dr. Frost (the “Adsport Lawsuit”). This lawsuit was brought by Adsport individually and on behalf of a putative class of the Company’s stockholders, claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Adsport Lawsuit seeks to declare the action to be a proper class action, monetary damages, including interest, an award of reasonable costs, and such equitable/injunctive relief as the Court may deem proper. On December 26, 2018, the Adsport Lawsuit was transferred to the United States District Court for the Southern District of Florida, which stayed and administratively closed the action on January 4, 2019.
On or about September 21, 2018, Michael Brennan (“Brennan”), a purported stockholder, filed a putative class action lawsuit in the United States District Court for the Southern District of Florida against the Company and certain of its current and former executive officers (the “Brennan Lawsuit”). This lawsuit was brought by Brennan individually and on behalf of a putative class of the Company’s stockholders, claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Brennan Lawsuit seeks to declare the action to be a class action and certify Brennan as the class representative, monetary damages, including prejudgment and post judgment interest, an award of reasonable costs, including attorneys’ fees, expert fees and other costs, and such other relief as the Court may deem proper.
60
On or about September 27, 2018, Frank Lipsius (“Lipsius”), a purported stockholder, filed a shareholder derivative complaint in the Circuit Court of the Eleventh Judicial Circuit of Florida serving Miami-Dade County against the Company as a nominal defendant, certain of the Company’s current and former executive officers, and members of its Board of Directors (the “Lipsius Lawsuit”). This lawsuit was brought by Lipsius and alleges breach of fiduciary duty against the officers and directors named therein, based on the allegations raised by the SEC in the SEC Lawsuit that the Company made misleading statements, and failed to maintain proper internal controls. The Lipsius Lawsuit seeks to declare that Lipsius maintain the action on behalf of the Company and that Lipsius is a proper and adequate representative of the Company, to direct the Company to improve its corporate governance and internal procedures, monetary damages, restitution, an award of reasonable attorneys’ fees and expert fees and other costs, and such additional or different relief as the Court may deem just and proper. On November 21, 2018, Lipsius and Louis Alexander filed a motion to consolidate the Lipsius Lawsuit and Alexander Lawsuit (defined below). On December 14, 2018, the Company and the other defendants named in the Lipsius Lawsuit and the Alexander Lawsuit filed a motion to dismiss the Lipsius and Alexander Lawsuits arguing that the case should be adjudicated in Delaware, or, in the alternative, stayed pending resolution of the various pending actions in federal and Delaware state court.
On or around September 27, 2018, the Company received a demand for the inspection of the books and records of the Company from counsel representing Jamie Gewirtz and Emily Gewirtz Stiebel (“Gewirtz/Stiebel”), each a purported stockholder of the Company, based on the allegations raised by the SEC in the SEC Complaint. On or around December 4, 2018, Gewirtz/Stiebel filed a shareholder derivative complaint in the Court of Chancery of the State of Delaware against the Company, as a nominal defendant, certain of the Company’s current executives and officers, and certain of the Company’s current and former members of its Board of Directors (the “Gewirtz/Stiebel Lawsuit”). The Gewirtz/Stiebel Lawsuit alleges breach of fiduciary duty against the officers and directors named therein based on the allegations raised by the SEC in the SEC Complaint. The Gewirtz/Stiebel Lawsuit seeks to declare that Gewirtz/Stiebel may maintain the action on behalf of the Company and that Gewirtz/Stiebel are proper and adequate representatives of the Company, a finding that the defendants breached their fiduciary duties, to have the defendants disgorge certain remuneration received from the Company, to direct the defendants to account for all damages sustained or to be sustained by the Company and all profits obtained by the alleged wrongdoing, have the Company improve its internal controls, equitable and injunctive relief, an award of monetary damages, including pre- and post-judgment interest, an award of reasonable attorneys’, expert witness fees and other costs, and such other and further relief as the Court deems just and proper.
On or about October 2, 2018 Andy Yu (“Yu”), a purported stockholder, filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida against the Company as a nominal defendant, certain of the Company’s current and former executive officers, certain current and former members of its Board of Directors, and Frost Gamma Investments Trust (the “Yu Lawsuit”). The Yu Lawsuit alleges breach of fiduciary duty against the officers and directors named therein based on the allegations raised by the SEC in the SEC Complaint, unjust enrichment, and that the Company made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading and failed to maintain effective internal controls. The Yu Lawsuit seeks to declare that Yu maintain the action on behalf of the Company and that Yu is a proper and adequate representative of the Company, to direct the Company to improve its corporate governance and internal procedures, monetary damages, restitution, an award of reasonable attorneys’ fees and expert fees and other costs, and such additional or different relief as the Court may deem just and proper. On December 14, 2018, the Company and the other defendants named in the Yu Lawsuit filed a motion to stay proceedings pending resolution of the SEC Complaint, the federal securities class actions and the various stockholder derivative actions in Delaware, and also requested an enlargement of time to respond to the Yu Lawsuit pending resolution of such motion to stay. On January 9, 2019, the United States District Court for the Southern District of Florida granted the defendants’ motion for an enlargement of time to respond to the Yu Lawusit pending resolution of the motion to stay until May 4, 2019.
On or about October 8, 2018 Paul Camhi (“Camhi”), a purported stockholder, filed a putative class action lawsuit in the United States District Court for the Southern District of Florida against the Company and Dr. Frost (the “Camhi Lawsuit”). The Camhi Lawsuit was brought by Camhi individually and on behalf of a putative class of the Company’s stockholders, claiming that in connection with the facts and circumstances underlying the allegations in the SEC Complaint, the Company engaged in fraudulent conduct and made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading. The Camhi Lawsuit seeks to declare the action to be a class action,to certify Camhi as the class representative, and to award monetary damages, including prejudgment and post judgment interest, an award of reasonable attorneys’ fees, and such other relief as the Court may deem just and proper. On November 28, 2018, Camhi voluntarily dismissed the Camhi Lawsuit.
On or around October 10, 2018, counsel for plaintiffs filed a Consolidated Complaint for Violations or the Federal Securities Laws against MabVax Therapeutics, Inc. in the United States District Court of the Southern District of California, in which the Company and its CEO, Phillip Frost, M.D, were also named as a defendants. This lawsuit was brought by a purported holder of MabVax’s securities, both individually and on behalf of a putative class of the MabVax’s stockholders, based on the allegations raised by the SEC in its lawsuit filed against the Company and others on September 7, 2018. The
61
lawsuit seeks to declare the action to be a class action, monetary damages in an amount to be proven at trial, including interest thereon, an award of reasonable costs and expenses, including counsel fees and expert fees, and such other and further relief as the Court may deem just and proper. On November 13, 2018, the case was voluntarily dismissed without prejudice.
On or about October 15, 2018 Richard Tunick (“Tunick”), a purported stockholder, filed a shareholder derivative complaint in the Court of Chancery of the State of Delaware against the Company as a nominal defendant, Dr. Frost and the Company’s Board of Directors (the “Tunick Lawsuit”). The Tunick Lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint. The lawsuit seeks to declare the action a proper derivative action, monetary damages, equitable and injunctive relief, to direct the Company to improve its internal controls and Board oversight , an award of reasonable attorneys’ fees and expert fees, and such other and further relief as the Court deems just and proper. On February 11, 2019, a Verified Consolidated Derivative Complaint was filed in the Court of Chancery of the State of Delaware (the “Consolidated Delaware Action”) consolidating this matter with the Gewirtz/Stiebel Lawsuit, Lutzker Lawsuit(as defined below), Davis Lawsuit (as defined below), and the Pawlenko/Breuninger Lawsuit (as defined below). Gewirtz/Stiebel, Tunick, Lutzker, and Davis were named as Co-Lead Plaintiffs in the filing. On February 12, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed a motion to stay proceedings pending resolution of the federal securities class actions. On February 19, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed their opening brief in support of their motion to stay.
On November 1, 2018, Lisette Demetriades (“Demetriades”), a purported stockholder, filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida against the Company as a nominal defendant, certain of the Company’s current and former executive officers, certain current and former members of its Board of Directors, and Frost Gamma Investment Trust (the “Demetriades Lawsuit”). The Demetriades Lawsuit alleges breach of fiduciary duty against the officers and directors named therein, based on the allegations raised by the SEC in the SEC Lawsuit, unjust enrichment, violations of the federal securities laws, that the individual defendants and Frost Gamma Investment Trust caused the Company made false and misleading statements of material fact or omitted to state material facts necessary to make the statements made not misleading, and the Company failed to maintain effective internal controls. The lawsuit seeks to declare the action a proper derivative action, monetary damages, to direct the Company to improve its internal controls and Board oversight concerning investments and self-dealing, restitution and disgorgement of profits, an award of reasonable attorneys’ fees and experts’ fees, and such other and further relief as the Court deems just and proper. On December 14, 2018, the Company and the other defendants named in the Demetriades Lawsuit filed a motion to stay proceedings pending resolution of the SEC Complaint, the federal securities class actions and the various stockholder derivative actions in Delaware, and subsequently, on January 10, 2019, requested an enlargement of time to respond to the Demetriades Lawsuit pending resolution of such motion to stay. On January 16, 2019, the United States District Court for the Southern District of Florida granted the defendants’ motion for an enlargement of time to respond to the Demetriades complaint pending resolution of the motion to stay.
On or about November 2, 2018, Louis T. Alexander (“Alexander”), a purported stockholder, filed a shareholder derivative complaint in the Circuit Court of the Eleventh Judicial Circuit of Florida serving Miami-Dade County against the Company, as a nominal defendant, Dr. Frost, certain current and former members of the Company’s Board of Directors and executive officers (the “Alexander Lawsuit”). The Alexander Lawsuit alleges breach of fiduciary duty against the officers and directors named therein, based on the allegations raised by the SEC in the SEC Lawsuit. The lawsuit seeks to declare that the Alexander maintain the action on behalf of the Company and that Alexander is a proper and adequate representative of the Company, monetary damages, to have the Company improve its corporate governance and internal procedures, a restitution award to the Company and disgorgement of profits, benefits and other compensation, an award of reasonable attorneys’ fees, accountants’ and experts’ fees, costs and expenses, and such other relief as the Court deems just and proper. On November 21, 2018, Lipsius and Alexander filed a motion to consolidate the Lipsius and Alexander Lawsuits. On December 14, 2018, the Company and the other defendants named in the Lipsius Lawsuit and the Alexander Lawsuit filed a motion to dismiss the Lipsius and Alexander Lawsuits arguing that the cases should be adjudicated in Delaware, or, in the alternative, stayed pending resolution of the various pending actions in federal and Delaware state court.
On or about November 7, 2018, Esther Susan Lutzker (“Lutzker”), a purported stockholder, filed a shareholder derivative complaint in the Court of Chancery of the State of Delaware against the Company as a nominal defendant and certain members of the Company’s Board of Directors (the “Lutzker Lawsuit”). This lawsuit was brought by Lutzker and alleges breach of fiduciary duty against the directors named therein, based on the allegations raised by the SEC in the SEC Complaint, and that the Company made false and misleading statements and failed to maintain effective internal controls. The Lutzker Lawsuit seeks to declare that Lutzker maintain the action on behalf of the Company, that Lutzker is a proper and adequate representative of the Company, monetary damages, appropriate equitable relief, an award of costs and disbursements, including reasonable attorneys’, accountants’, and experts’ fees costs and expenses, and such other and further relief as the Court may deem just and proper. On February 11, 2019, a Verified Consolidated Derivative Complaint was filed in the Court of Chancery of the State of Delaware consolidating this matter with the Gewirtz/Stiebel Lawsuit, Tunick Lawsuit, Davis Lawsuit, and Pawlenko/
62
Breuninger Lawsuit. Gewirtz/Stiebel, Tunick, Lutzker, and Davis were named as Co-Lead Plaintiffs in the filing. On February 12, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed a motion to stay proceedings pending resolution of the federal securities class actions. On February 19, 2019, the Company and the other defendants named in the Consolidated Delaware Action filed their opening brief in support of their motion to stay.
On or about November 14, 2018, Sammy Lee (“Lee”), a purported stockholder, filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida against the Company as a nominal defendant, Dr. Frost, Frost Gamma Investments Trust, an entity controlled by Dr. Frost, and certain of the Company’s current and former executive officers, and current and former members of its Board of Directors (the “Lee Lawsuit”). The Lee Lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint, unjust enrichment, violations of the federal securities laws, and that the Company made false and misleading statements and failed to maintain effective internal controls. The lawsuit seeks to declare that Lee maintain the action on behalf of the Company and that Lee is a proper and adequate representative of the Company, to declare that the defendants breached or aided and abetted the breach of their fiduciary duties to the Company, monetary damages to the Company together with pre-judgment and post-judgment interest thereon, to have the Company improve its corporate governance and internal procedures, a restitution award to the Company, an award of reasonable attorneys’ fees, experts’ fees, costs and expenses, and such other and further relief as the Court may deem just and proper. The action was dismissed sua sponte by the Judge for failure to state a claim. The lawsuit was amended and refiled on November 30, 2018.
On or around December 4, 2018, the Company received a demand for the inspection of the books and records of the Company from counsel representing Ivan Pawlenko (“Pawlenko”), a stockholder of the Company, based on the allegations raised by the SEC in the SEC Complaint. On or around December 5, 2018, the Company received a demand for the inspection of the books and records of the Company from counsel representing Martin Breuninger (“Breuninger”), a stockholder of the Company, based on the allegations raised by the SEC in the SEC Complaint. Pawlenko and Breuninger are represented by the same counsel. The Company is providing the requested documents to counsel. On or about January 29, 2019, Pawlenko and Breuninger jointly filed a shareholder derivative complaint in the Court of Chancery of the State of Delaware against the Company as a nominal defendant,Dr. Frost, and certain former and current members of the Company’s Board of Directors (the “Pawlenko/Breuninger Lawsuit”). This lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint. The lawsuit seeks to declare that the action is a proper derivative action and that plaintiffs are proper and adequate representatives of the Company’s interests, a finding that the defendants breached their fiduciary duties, to have the defendants disgorge all gains received and repayment of same to the Company, award monetary damages to the Company, including pre- and post-judgment interest, equitable relief sufficient to remedy alleged harms, an award of the costs, expenses, and disbursements of the action, including any attorneys’ and experts’ fees, including pre- and post-judgment interest, and such other relief as the Court deems just, equitable, and proper. On February 11, 2019, a Verified Consolidated Derivative Complaint was filed in the Court of Chancery of the State of Delaware consolidating this matter with the Gewirtz/Stiebel Lawsuit, Tunick Lawsuit, Lutzker Lawsuit, and Davis Lawsuit. Gewirtz/Stiebel, Tunick, Lutzker, and Davis were named as Co-Lead Plaintiffs in the filing. On February 12, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed a motion to stay proceedings pending resolution of the federal securities class actions. On February 19, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed their opening brief in support of their motion to stay.
On or about December 17, 2018, Thaddeus R. Sobieski and Donnis W. King, purported stockholders of the Company, filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida against the Company as a nominal defendant, Dr. Frost, Frost Gamma Investments Trust, and certain of the Company’s current and former executive officers, and current and former members of its Board of Directors (the “Sobieski King Lawsuit”). The Sobieski King Lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint, violations of the federal securities laws, unjust enrichment, waste of corporate assets, and that the Company made false and misleading statements and failed to maintain effective internal controls. The lawsuit seeks to declare that the action is a proper derivative action, breach of fiduciary duties, an award monetary damages to the Company, requires the current and former directors named as defendants to remit to the Company all salaries and other compensation received during the period of alleged breach of fiduciary duties, to have the Company improve its corporate governance and internal procedures, an award of pre-judgement and post- judgement interest, reasonable attorneys’ fees, experts’ fees, costs and expenses, and such other and further relief as the Court may deem just and proper. On February 19, 2019, the Company and other defendants named in the Sobieski Lawsuit filed an unopposed motion to stay proceedings pending resolution of the federal securities class actions and for an enlargement of time to respond to the Sobieski complaint.
On December 31, 2018, Connie Wendt (“Wendt”), a purported stockholder, filed a shareholder derivative complaint in the United States District Court for the Southern District of Florida against the Company as a nominal defendant, certain of its current and former executive officers, certain current and former members of its Board of Directors, Frost Gamma Investments Trust, and certain other individuals and an entity named in the SEC Complaint (the “Wendt Lawsuit”). Wendt Lawsuit alleges breach of fiduciary duties, unjust enrichment, violations of the federal securities laws, and breach of fiduciary duties for insider selling and
63
misappropriation of information. The lawsuit seeks to declare that Wendt maintain the action on behalf of the Company and that Wendt is a proper and adequate representative of the Company, declare that each of the Company’s named officers and directors breached their fiduciary duties to the Company, monetary damages, disgorgement of alleged profits, an award of costs and disbursements including reasonable attorneys’ fees, accountants’ and experts’ fees and costs and expenses, and such other and further relief as the Court deems just and proper.
On or around November 7, 2018, the Company received a demand for the inspection of the books and records of the Company from counsel representing Vivian Davis (“Davis”), a stockholder of the Company, based on the allegations raised by the SEC in the SEC Complaint. The Company provided the requested documents to Davis’ counsel. On or about January 18, 2019, Davis, a purported stockholder, filed a shareholder derivative complaint in the Court of Chancery of the State of Delaware against the Company as a nominal defendant, Dr. Frost, and members of its Board of Directors (the “Davis Lawsuit”). The Davis Lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint. The Davis Lawsuit seeks a finding of breach of fiduciary duties, extraordinary equitable and injunctive relief, disgorgement, an award of reasonable attorneys’, accountants’, and experts’ fees, costs and expenses, and such other further relief as the Court deems just and proper. On February 11, 2019, a Verified Consolidated Derivative Complaint was filed in the Court of Chancery of the State of Delaware consolidating this matter with the Gewirtz/Stiebel Lawsuit, Tunick Lawsuit, Lutzker Lawsuit, and the Pawlenko/Breuninger Lawsuit. Gewirtz/Stiebel, Tunick, Lutzker, and Davis were named as Co-Lead Plaintiffs in the filing. On February 12, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed a motion to stay proceedings pending resolution of the federal securities class actions. On February 19, 2019, the Company and the other defendants named in the Consolidated Delaware Action, filed their opening brief in support of their motion to stay.
On or around January 28, 2019, Robert Davydov (“Davydov”), a purported stockholder, filed a shareholder derivative complaint in the Circuit Court of the Eleventh Judicial Circuit of Florida serving Miami-Dade County against the Company as a nominal defendant and the certain members of its Board of Directors (the “Davydov Lawsuit”). The Davydov Lawsuit alleges breach of fiduciary duty based on the allegations raised by the SEC in the SEC Complaint, corporate waste and unjust enrichment. The Davydov Lawsuit seeks to declare that Davydov may maintain the action on behalf of the Company and that Davydov is a proper and adequate representative of the Company, to declare that the defendants breached and/or aided and abetted the breach of their fiduciary duties, an award of monetary damages, restitution, an award of reasonable attorneys’ fees and expert witness fees and other costs, and such other and further equitable relief as the Court deems just and proper.
The Company intends to vigorously defend itself against the class action and derivative claims. Based on the early stages of these legal proceedings, at this time, the Company is not able to reasonably estimate a possible range of loss, if any, that may result from these allegations.
ITEM 4. MINE SAFETY DISCLOSURES.
Not applicable.
64
PART II
ITEM 5. |
MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES. |
Our Common Stock is traded publicly on the NASDAQ Stock Market (“NASDAQ”) and the Tel Aviv Stock Exchange under the symbol “OPK”.
The following table sets forth for the periods indicated the high and low sales prices per share of our Common Stock during each of the quarters set forth below as reported on the NASDAQ :
High |
Low |
||||||
2018 |
|||||||
First Quarter |
$ |
5.14 |
$ |
2.66 |
|||
Second Quarter |
5.02 |
2.91 |
|||||
Third Quarter |
6.40 |
3.21 |
|||||
Fourth Quarter |
3.92 |
2.34 |
|||||
2017 |
|||||||
First Quarter |
$ |
9.55 |
$ |
7.13 |
|||
Second Quarter |
8.04 |
5.99 |
|||||
Third Quarter |
7.24 |
5.85 |
|||||
Fourth Quarter |
7.08 |
4.50 |
|||||
As of February 15, 2019, there were approximately 480 holders of record of our Common Stock.
We have not declared or paid any cash dividends on our Common Stock. No cash dividends have been previously paid on our Common Stock and none are anticipated in fiscal 2019.
Stock Performance Graph
The following graph compares the five-year cumulative total return of our Common Stock with the S&P 500 Index and the NASDAQ Biotechnology Index. The graph assumes $100 invested on December 31, 2013 in our Common Stock and in each of the foregoing indices. The stock price performance reflected in the graph below is not necessarily indicative of future price performance.
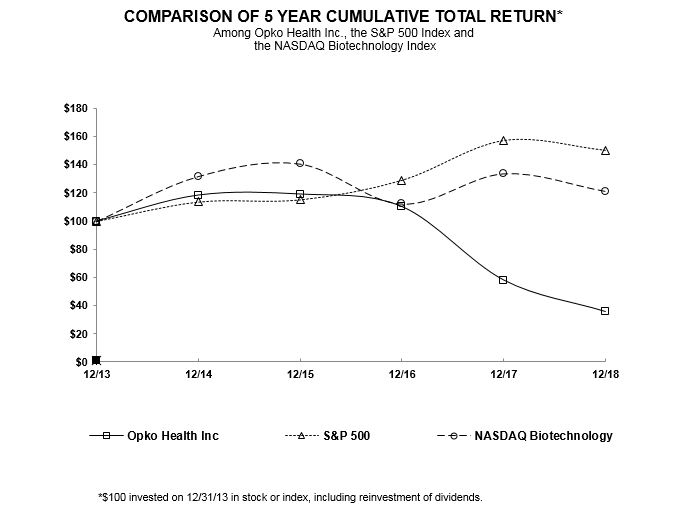
12/31/2013 |
12/31/2014 |
12/31/2015 |
12/31/2016 |
12/31/2017 |
12/31/2018 |
||||||||||||||||||
OPKO Health, Inc. |
$ |
100.00 |
$ |
118.36 |
$ |
119.08 |
$ |
110.19 |
$ |
58.06 |
$ |
35.66 |
|||||||||||
S&P 500 |
100.00 |
113.69 |
115.26 |
129.05 |
157.22 |
150.33 |
|||||||||||||||||
NASDAQ Biotechnology |
100.00 |
131.71 |
140.56 |
112.25 |
133.67 |
121.24 |
|||||||||||||||||
Recent Sales of Unregistered Securities
All recent sales of unregistered securities were previously disclosed in a Current Report on Form 8-K or Quarterly Report on Form 10-Q.
66
ITEM 6. SELECTED FINANCIAL DATA.
The following selected historical consolidated statement of operations data for the years ended December 31, 2018, 2017, 2016, 2015 and 2014 and the consolidated balance sheet data as of December 31, 2018, 2017, 2016, 2015 and 2014, below are derived from our audited consolidated financial statements and related notes thereto. This data should be read in conjunction with our “Management’s Discussion and Analysis of Financial Condition and Results of Operation” and our consolidated financial statements and the related notes thereto.
Effective January 1, 2018, we adopted Accounting Standards Codification Topic 606, Revenue from Contracts with Customers, using the full retrospective transition method. The information contained in the table below for the years ended December 31, 2017, 2016 and 2015 has been adjusted to to reflect our retrospective adoption of Topic 606. For further discussion on the impact of adopting Topic 606, refer to Note 2 to the Consolidated Financial Statements, “Summary of Significant Accounting Policies.” The information for the year ended December 31, 2014 has not been adjusted to reflect the impact of the adoption of ASC 606.
For the years ended December 31, |
||||||||||||||||||||
(In thousands, except share and per share information) |
2018 |
2017 |
2016 |
2015 |
2014 |
|||||||||||||||
Statement of operations data: |
||||||||||||||||||||
Revenues |
$ |
990,266 |
$ |
966,006 |
$ |
1,117,494 |
$ |
447,517 |
$ |
91,125 |
||||||||||
Costs and expenses: |
||||||||||||||||||||
Cost of revenue |
604,636 |
620,130 |
611,482 |
235,239 |
48,009 |
|||||||||||||||
Operating expenses |
556,827 |
622,318 |
602,563 |
332,858 |
188,931 |
|||||||||||||||
Total costs and expenses |
1,161,463 |
1,242,448 |
1,214,045 |
568,097 |
236,940 |
|||||||||||||||
Operating loss |
(171,197 |
) |
(276,442 |
) |
(96,551 |
) |
(120,580 |
) |
(145,815 |
) |
||||||||||
Other income and (expense), net |
(6,072 |
) |
4,518 |
(271 |
) |
(39,517 |
) |
(25,212 |
) |
|||||||||||
Income tax benefit (provision) |
38,726 |
(18,855 |
) |
56,115 |
113,675 |
(24 |
) |
|||||||||||||
Net loss |
(153,040 |
) |
(305,250 |
) |
(48,359 |
) |
(53,527 |
) |
(174,638 |
) |
||||||||||
Net loss attributable to common shareholders |
$ |
(153,040 |
) |
$ |
(305,250 |
) |
$ |
(48,359 |
) |
$ |
(52,127 |
) |
$ |
(171,666 |
) |
|||||
Loss per share: |
||||||||||||||||||||
loss per share, basic |
$ |
(0.27 |
) |
$ |
(0.55 |
) |
$ |
(0.09 |
) |
$ |
(0.11 |
) |
$ |
(0.41 |
) |
|||||
loss per share, diluted |
$ |
(0.27 |
) |
$ |
(0.55 |
) |
$ |
(0.10 |
) |
$ |
(0.11 |
) |
$ |
(0.41 |
) |
|||||
Weighted average number of common shares outstanding basic: |
563,143,663 |
559,160,565 |
550,846,553 |
488,065,908 |
422,014,039 |
|||||||||||||||
Weighted average number of common shares outstanding diluted: |
563,143,663 |
559,160,565 |
555,605,448 |
488,065,908 |
422,014,039 |
|||||||||||||||
Balance sheet data: |
||||||||||||||||||||
Total assets |
$ |
2,451,072 |
$ |
2,589,956 |
$ |
2,766,619 |
$ |
2,799,188 |
$ |
1,267,664 |
||||||||||
Long-term liabilities |
$ |
371,460 |
$ |
434,304 |
$ |
480,166 |
$ |
614,423 |
$ |
348,812 |
||||||||||
Total shareholders’ equity |
$ |
1,791,291 |
$ |
1,843,623 |
$ |
2,046,433 |
$ |
1,957,695 |
$ |
835,741 |
||||||||||
67
ITEM 7. |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS. |
This Annual Report on Form 10-K contains certain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 (“PSLRA”), Section 27A of the Securities Act of 1933, as amended, (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended, (the “Exchange Act”), about our expectations, beliefs, or intentions regarding our product development efforts, business, financial condition, results of operations, strategies, or prospects. You can identify forward-looking statements by the fact that these statements do not relate strictly to historical or current matters. Rather, forward-looking statements relate to anticipated or expected events, activities, trends, or results as of the date they are made. Because forward-looking statements relate to matters that have not yet occurred, these statements are inherently subject to risks and uncertainties that could cause our actual results to differ materially from any future results expressed or implied by the forward-looking statements. Many factors could cause our actual activities or results to differ materially from the activities and results anticipated in forward-looking statements. These factors include those contained in “Item 1A — Risk Factors” of this Annual Report on Form 10-K. We do not undertake any obligation to update forward-looking statements. We intend that all forward-looking statements be subject to the safe harbor provisions of PSLRA. These forward-looking statements are only predictions and reflect our views as of the date they are made with respect to future events and financial performance.
OVERVIEW
We are a diversified healthcare company that seeks to establish industry-leading positions in large and rapidly growing medical markets. Our diagnostics business includes BioReference Laboratories (“BioReference”), the nation’s third-largest clinical laboratory with a core genetic testing business and an almost 300-person sales and marketing team to drive growth and leverage new products, including the 4Kscore prostate cancer test. Our pharmaceutical business features Rayaldee, an FDA-approved treatment for secondary hyperparathyroidism (“SHPT”) in adults with stage 3 or 4 chronic kidney disease (“CKD”) and vitamin D insufficiency (launched in November 2016), OPK88004, a selective androgen receptor modulator which we have studied for benign prostatic hyperplasia, but for which we are exploring other indications, and OPK88003, a once or twice weekly oxyntomodulin for type 2 diabetes and obesity which is a clinically advanced drug candidate among the new class of GLP-1 glucagon receptor dual agonists (phase 2b). Our pharmaceutical business also features hGH-CTP, a once-weekly human growth hormone injection (in phase 3 and partnered with Pfizer).
We operate established pharmaceutical platforms in Spain, Ireland, Chile and Mexico, which are generating revenue and from which we expect to generate positive cash flow and facilitate future market entry for our products currently in development. We have a development and commercial supply pharmaceutical company, as well as a global supply chain operation and holding company in Ireland, which we expect will play an important role in the development, manufacturing, distribution and approval of a wide variety of drugs with an emphasis on high potency products. We also own a specialty active pharmaceutical ingredients (“APIs”) manufacturer in Israel, which we expect will facilitate the development of our pipeline of molecules and compounds for our proprietary molecular diagnostic and therapeutic products.
RECENT DEVELOPMENTS
In February 2019, we issued $200.0 million aggregate principal amount of Convertible Senior Notes due 2025 (the “2025 Convertible Notes”) in an underwritten public offering. The 2025 Convertible Notes will bear interest at a rate of 4.50% per year, payable semiannually in arrears on February 15 and August 15 of each year, beginning on August 15, 2019. The notes mature on February 15, 2025, unless earlier repurchased, redeemed or converted.
On February 1, 2019, we announced that the FDA has approved our point-of-care Sangia Total PSA Test using the Claros 1 Analyzer. The product is indicated to quantitatively measure total PSA in whole blood from a fingerstick of blood collected by a healthcare professional and is used in conjunction with a digital rectal exam as an aid in the detection of prostate cancer in men aged 50 years and older. We are evaluating commercialization strategies for the PSA test on the Claros 1 Analyzer, including the expansion of the test menu, prior to commercialization.
On January 31, 2019, we announced that Novitas Solutions, Inc. has issued a notice of a future non-coverage determination for the 4Kscore test to be effective March 20, 2019. The notice released by Novitas does not appear to be different from the draft local coverage determination released by Novitas on May 18, 2018. We are evaluating options to appeal the decision and undertake other steps with the U.S. Centers for Medicare & Medicaid Services (CMS) in an effort to have this determination rescinded or reversed. We are also developing a strategy to obtain FDA approval for the 4Kscore test, among other efforts, to assist in securing broad reimbursement coverage.
On December 27, 2018, we announced that both the Company and Dr. Frost, entered into settlement agreements with the Securities and Exchange Commission (the “Commission”), subject to court approval, resolving the complaint filed by the
68
Commission against the Company and Dr. Frost in the U.S. District Court for the Southern District of New York on September 7, 2018. Pursuant to the settlement, and without admitting or denying any of the allegations of the Complaint, the Company is enjoined from violating Section 13(d) of the Exchange Act and paid a $100,000 penalty. Liability under Section 13(d) can be established without any showing of wrongful intent or negligence. The settlement was approved by the Court in January 2019.
On November 8, 2018, we entered into stock purchase agreements with certain investors pursuant to which we agreed to sell to such investors in private placements an aggregate of approximately 26.5 million shares of our common stock at a purchase price of $3.49 per share, which was the closing bid price of our Common Stock on the NASDAQ on such date, for an aggregate purchase price of $92.5 million.
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. The credit agreement was terminated on or around February 20, 2019 and amounts borrowed during 2019 were repaid from the proceeds of the 2025 Convertible Notes offering. Borrowings under the line of credit bore interest at a rate of 10% per annum and could be repaid and reborrowed at any time. The credit agreement included various customary remedies for the lender following an event of default, including the acceleration of repayment of outstanding amounts under line of credit. The line of credit would have matured on November 8, 2023.
RESULTS OF OPERATIONS
For The Years Ended December 31, 2018 and December 31, 2017
Effective January 1, 2018, we adopted Accounting Standards Codification Topic 606, Revenue from Contracts with Customers, using the full retrospective transition method. Under this method, we have revised our Consolidated Financial Statements for the years ended December 31, 2017 and 2016, as if Topic 606 had been effective for those periods. For further discussion on the impact of adopting Topic 606, refer to Note 2 to the Consolidated Financial Statements, “Summary of Significant Accounting Policies.”
Revenues |
For the years ended December 31, |
||||||||||
(In thousands) |
2018 |
2017 |
Change |
||||||||
Revenue from services |
$ |
813,248 |
$ |
782,710 |
$ |
30,538 |
|||||
Revenue from products |
107,112 |
107,759 |
(647 |
) |
|||||||
Revenue from transfer of intellectual property and other |
69,906 |
75,537 |
(5,631 |
) |
|||||||
Total revenues |
$ |
990,266 |
$ |
966,006 |
$ |
24,260 |
|||||
Revenue from services for the year ended December 31, 2018 increased approximately $30.5 million compared to the year ended December 31, 2017. The increase in revenue from services is attributable to reduced adjustments to estimated collection amounts from third-party payors as discussed in the paragraph below. Revenue from services for the year ended December 31, 2017 was also negatively affected by claims of overpayment as a result of payor error of approximately $30.0 million. In addition, Revenue from services for the year ended December 31, 2018 increased by $12.9 million from improved collections for our clinical testing resulting from improvements in our billing cycle and $4.7 million from higher volume in our genomics testing. Partially offsetting these increases, Revenue from services for the year ended December 31, 2018 was negatively affected by $24.5 million as a result of changes in clinical test volumes as a result of increased competition, reduced clinical reimbursement of $15.6 million due to PAMA which came into effect in January 2018, and reduced genomics reimbursement of $11.6 million as a result of an increase in denial rates and changes to medical and procedural requirements.
Estimated collection amounts are subject to the complexities and ambiguities of billing, reimbursement regulations and claims processing, as well as issues unique to Medicare and Medicaid programs, and require us to consider the potential for retroactive adjustments when estimating variable consideration in the recognition of revenue in the period the related services are rendered. For the year ended December 31, 2018, adjustments to estimated collection amounts from third-party payors decreased revenue by $22.8 million compared to $66.0 million in 2017. For the year ended December 31, 2017, approximately $35.1 million of adjustments related to our genomics testing and approximately $30.9 million related to our clinical testing. The adjustments for our genomics testing in 2017 primarily relate to changes in payor medical and procedural requirements for our genomics testing and the adjustments for our clinical testing in 2017 and 2018 primarily relate to delays in the billing cycle resulting from our implementation of a new clinical testing billing system in late 2016 as well as reduced clinical reimbursement as discussed above.
69
We may have an obligation to reimburse Medicare, Medicaid, and third-party payors for overpayments regardless of fault. We have periodically identified and reported overpayments, reimbursed payors for overpayments and taken what we believe to be appropriate corrective action. Settlements with third-party payors for retroactive adjustments due to audits, reviews or investigations are considered variable consideration and are included in the determination of the estimated transaction price for providing services. These settlements are estimated based on the terms of the payment agreement with the payor, correspondence from the payor and our historical settlement activity, including an assessment of the probability a significant reversal of cumulative revenue recognized will occur when the uncertainty is subsequently resolved. Estimated settlements are adjusted in future periods as adjustments become known (that is, new information becomes available), or as years are settled or are no longer subject to such audits, reviews, and investigations. During the year ended December 31, 2017, a payor informed us it had overpaid BioReference due to an error on its part over a period of several years, including multiple years prior to the acquisition of BioReference by OPKO in August 2015. For the years ended December 31, 2018 and 2017, Revenue from services was reduced by approximately $8.1 million and $30.0 million, respectively, related to claims of overpayment.
The composition of Revenue from services by payor for the years ended December 31, 2018 and 2017 is as follows:
For the years ended December 31, |
|||||||
(In thousands) |
2018 |
2017 |
|||||
Healthcare insurers |
$ |
370,096 |
$ |
368,628 |
|||
Government payors |
271,590 |
264,493 |
|||||
Client payors |
150,259 |
128,867 |
|||||
Patients |
21,303 |
20,722 |
|||||
Total |
$ |
813,248 |
$ |
782,710 |
|||
Overall, Revenue from products for the year ended December 31, 2018 was consistent with the comparative period in 2017 as an increase in sales of Rayaldee of $20.3 million in the year ended December 31, 2018 compared to $9.1 million in the year ended December 31, 2017, was partially offset by a decrease in revenue at FineTech and in Chile. Revenue from transfer of intellectual property for the years ended December 31, 2018 and 2017 principally reflected $60.0 million and $61.2 million, respectively, of revenue related to the Pfizer Transaction. Revenue from transfer of intellectual property for the years ended December 31, 2018 and 2017, also reflects $2.0 million and $10.0 million, respectively, of revenue from milestone payments from our licensees, VFMCRP and TESARO.
Costs of revenue. Costs of revenue for the year ended December 31, 2018 decreased $15.5 million compared to 2017. Cost of service revenue decreased in 2018 due to a decrease in volume and employee related costs for clinical testing at BioReference. The decrease in cost of product revenue is attributable to $5.4 million of inventory obsolescence expense recognized in 2017 related primarily to the launch of Rayaldee and to changes in the product mix of items sold during the period. Cost of revenue for the years ended December 31, 2018 and 2017 were as follows:
Cost of Revenue |
For the years ended December 31, |
||||||||||
(In thousands) |
2018 |
2017 |
Change |
||||||||
Cost of service revenue |
$ |
546,654 |
$ |
558,953 |
$ |
(12,299 |
) |
||||
Cost of product revenue |
57,982 |
61,177 |
(3,195 |
) |
|||||||
Total cost of revenue |
$ |
604,636 |
$ |
620,130 |
$ |
(15,494 |
) |
||||
Selling, general and administrative expenses. Selling, general and administrative expenses for the years ended December 31, 2018 and 2017 were $358.3 million and $414.6 million, respectively. The decrease in selling, general and administrative expenses was primarily due to decreased expenses at BioReference due to planned cost reduction initiatives and to a decrease in corporate expenses, which was partially offset by $9.6 million of expenses related to the defense and investigation of actions brought by the U.S. Securities and Exchange Commission, which were settled in December 2018. Selling, general and administrative expenses for the year ended December 31, 2017 also reflected higher professional fees related to the implementation of a new billing system at BioReference. Selling, general and administrative expenses for the year ended December 31, 2017 included $8.8 million of expense to write-off certain other current assets. Selling, general and administrative expenses during the years ended December 31, 2018 and 2017 included equity-based compensation expense of $14.7 million and $21.2 million, respectively.
70
Research and development expenses. Research and development expenses for the years ended December 31, 2018 and 2017 were $125.6 million and $126.4 million, respectively. Research and development costs include external and internal expenses, partially offset by third-party grants and funding arising from collaboration agreements. External expenses include clinical and non-clinical activities performed by contract research organizations, lab services, purchases of drug and diagnostic product materials and manufacturing development costs. We track external research and development expenses by individual program for phase 3 clinical trials for drug approval and PMAs for diagnostics tests, if any. Internal expenses include employee-related expenses such as salaries, benefits and equity-based compensation expense. Other internal research and development expenses are incurred to support overall research and development activities and include expenses related to general overhead and facilities.
The following table summarizes the components of our research and development expenses:
For the years ended December 31, |
|||||||
2018 |
2017 |
||||||
External expenses: |
|||||||
Phase 3 clinical trials |
$ |
16,215 |
$ |
15,339 |
|||
Manufacturing expense for biological products |
39,974 |
47,737 |
|||||
PMA studies |
59 |
1,089 |
|||||
Earlier-stage programs |
15,591 |
7,620 |
|||||
Research and development employee-related expenses |
27,318 |
29,970 |
|||||
Other internal research and development expenses |
27,247 |
24,680 |
|||||
Third-party grants and funding from collaboration agreements |
(818 |
) |
— |
||||
Total research and development expenses |
$ |
125,586 |
$ |
126,435 |
|||
Overall research and development expenses for the year ended December 31, 2018 were consistent with the comparative period in 2017 as an increase in research and development expenses related to a once or twice weekly oxyntomodulin for type 2 diabetes and to a selective androgen receptor modulator for benign prostatic hyperplasia were offset by research and development tax credits recognized in 2018. Research and development expenses for the years ended December 31, 2018 and 2017 include equity-based compensation expenses of $4.2 million and $5.1 million, respectively. We expect our research and development expenses to increase as we continue to expand our research and development of potential future products.
Contingent consideration. Contingent consideration for the years ended December 31, 2018 and 2017, were $16.8 million and $3.4 million of income, respectively. The change in contingent consideration was primarily attributable to changes in assumptions regarding the timing of achievement of future milestones for OPKO Renal. The contingent consideration liabilities of $24.6 million at December 31, 2018 related to potential amounts payable to former stockholders of CURNA, OPKO Diagnostics and OPKO Renal pursuant to our acquisition agreements in January 2011, October 2011 and March 2013, respectively.
Amortization of intangible assets. Amortization of intangible assets was $67.9 million and $71.5 million, respectively, for the years ended December 31, 2018 and 2017. Amortization expense reflects the amortization of acquired intangible assets with defined useful lives. Our indefinite lived IPR&D assets will not be amortized until the underlying development programs are completed. Upon obtaining regulatory approval by the U.S. FDA, the IPR&D assets will be accounted for as a finite-lived intangible asset and amortized on a straight-line basis over its estimated useful life.
Asset impairment charges. Asset impairment charges was $21.8 million and $13.2 million, respectively, for the years ended December 31, 2018 and 2017. Asset impairment charges for the year ended December 31, 2018 is related to an impairment charge of $10.1 million to write our IPR&D assets for Alpharen and OPK88004 down to their estimated fair value and a goodwill impairment charge of $11.7 million to write the carrying amount of the FineTech reporting unit down to its estimated fair value due to the loss of a significant customer in 2018. Asset impairment charges for the year ended December 31, 2017 is related to an impairment charge of $13.2 million to write our intangible asset for VARUBI™ down to its estimated fair value.
Interest income. Interest income for the years ended December 31, 2018 and 2017, was not significant as our cash investment strategy emphasizes the security of the principal invested and fulfillment of liquidity needs.
Interest expense. Interest expense for the years ended December 31, 2018 and 2017, was $11.9 million and $6.6 million, respectively. Interest expense is principally related to interest incurred on the 2033 Senior Notes, on BioReference’s outstanding debt under its credit facility and on the 2023 Convertible Notes issued in February 2018. The increase in interest
71
expense for the year ended December 31, 2018 is primarily due to interest incurred on the 2023 Convertible Notes and to higher outstanding debt and interest rates under BioReference’s credit facility in 2018 compared to 2017.
Fair value changes of derivative instruments, net. Fair value changes of derivative instruments, net for the years ended December 31, 2018 and 2017, were $3.0 million and $0.1 million of income, respectively. Derivative income for the year ended December 31, 2018 principally related to the change in fair value of warrants to purchase additional shares of Neovasc. Fair value changes of derivative instruments, net for the year ended December 31, 2017 is primarily related $3.2 million of income due to the change in the fair value of the embedded derivatives in the 2033 Senior Notes, which was partially offset by $2.9 million of expense related to the change in the fair value of warrants and options to purchase additional shares of Neovasc, Inc. (“Neovasc”) and Xenetic Biosciences, Inc. (“Xenetic”).
Other income and (expense), net. Other income and (expense), net for the years ended December 31, 2018 and 2017, were $1.5 million and $10.5 million of income, respectively. Other income for the year ended December 31, 2018 primarily consists of net unrealized gains recognized during the period on equity securities. Other income for the year ended December 31, 2017 primarily consists of a $3.0 million gain on the sale of non-strategic assets at a wholly-owned BioReference subsidiary, a $1.5 million gain on the sale of certain available for sale investments, a $2.5 million gain in connection with the acquisition transaction between Eloxx Pharmaceuticals, Inc. and Sevion Therapeutics, Inc., and a $1.9 million gain in connection with the dilution of our equity method investment in VBI Vaccines Inc. (“VBI”).
Income tax benefit (provision). Our income tax benefit (provision) for the years ended December 31, 2018 and 2017 was $38.7 million, and $(18.9) million, respectively. The change in income tax benefit is primarily a result of our analysis of the realization of deferred tax assets and corresponding release of the valuation allowance associated with U.S. and non-U.S. deferred tax assets. As of December 31, 2017, the Company determined that it was more likely than not that certain U.S. and non-U.S. deferred tax assets would not be realized and recorded a valuation allowance of $28.7 million. On December 22, 2017, the Tax Act was enacted into law and the new legislation reduced the corporate income tax rate from 35% to 21% which required us to remeasure our U.S. deferred tax assets and liabilities and recognize the effect in the period of enactment, resulting in $31.8 million of expense, with an equal offset to valuation allowance.
Loss from investments in investees. We have made investments in other early stage companies that we perceive to have valuable proprietary technology and significant potential to create value for us as a shareholder or member. We account for these investments under the equity method of accounting, resulting in the recording of our proportionate share of their losses until our share of their loss exceeds our investment. Until the investees’ technologies are commercialized, if ever, we anticipate they will report a net loss. Loss from investments in investees was $14.5 million and $14.5 million for the years ended December 31, 2018 and 2017, respectively. Included in Loss from investments in investees for the year ended December 31, 2018 is a charge of $2.9 million to write our investment in InCellDx, Inc. down to its fair value as of December 31, 2018.
For The Years Ended December 31, 2017 and December 31, 2016
Revenues |
For the years ended December 31, |
||||||||||
(In thousands) |
2017 |
2016 |
Change |
||||||||
Revenue from services |
$ |
782,710 |
$ |
928,572 |
$ |
(145,862 |
) |
||||
Revenue from products |
107,759 |
83,467 |
24,292 |
||||||||
Revenue from transfer of intellectual property and other |
75,537 |
105,455 |
(29,918 |
) |
|||||||
Total revenues |
$ |
966,006 |
$ |
1,117,494 |
$ |
(151,488 |
) |
||||
Revenue from services for the year ended December 31, 2017 decreased approximately $145.9 million compared to 2016. The decrease in revenue from services is attributable to approximately $15.5 million of reduced reimbursement within our genomics testing as a result of an increase in denial rates and changes to payor medical and procedural requirements, approximately $35.1 million of adjustments to the estimated collection amounts from third-party payors for our genomics testing, and decreased volume in genomics testing of approximately $1.5 million. Revenue from services also declined by approximately $21.9 million in clinical test volumes as a result of increased competition, approximately $30.9 million related to changes in the estimated collection amounts from third-party payors for our clinical testing and approximately $11.0 million related to lower collections on patient billings for our clinical testing offset in part by improvements in our billing cycle. Revenue from services for the year ended December 31, 2017 was also affected by claims of overpayment as a result of payor error of approximately $30.0 million.
Estimated collection amounts are subject to the complexities and ambiguities of billing, reimbursement regulations and claims processing, as well as issues unique to Medicare and Medicaid programs, and require us to consider the potential for
72
retroactive adjustments when estimating variable consideration in the recognition of revenue in the period the related services are rendered. Actual amounts are adjusted in the period those adjustments become known based on actual collection experience. For the year ended December 31, 2017, changes to estimated collection amounts from third-party payors negatively affected revenue by approximately $35.1 million for our genomics testing and approximately $30.9 million for our clinical testing. The adjustments for our genomics testing primarily relate to changes in payor medical and procedural requirements for our genomics testing. The adjustments for our clinical testing primarily relate to delays in the billing cycle resulting from our implementation of a new clinical testing billing system in late 2016.
We may have an obligation to reimburse Medicare, Medicaid, and third-party payors for overpayments regardless of fault. We have periodically identified and reported overpayments, reimbursed payors for overpayments and taken what we believe to be appropriate corrective action. Settlements with third-party payors for retroactive adjustments due to audits, reviews or investigations are considered variable consideration and are included in the determination of the estimated transaction price for providing services. These settlements are estimated based on the terms of the payment agreement with the payor, correspondence from the payor and our historical settlement activity, including an assessment of the probability a significant reversal of cumulative revenue recognized will occur when the uncertainty is subsequently resolved. Estimated settlements are adjusted in future periods as adjustments become known (that is, new information becomes available), or as years are settled or are no longer subject to such audits, reviews, and investigations. During the year ended December 31, 2017, a payor informed us it had overpaid BioReference due to an error on its part over a period of several years, including multiple years prior to the acquisition of BioReference by OPKO in August 2015. For the year ended December 31, 2017, Revenue from services was reduced by approximately $30.0 million related to claims of overpayment as a result of payor error.
The increase in Revenue from products principally reflects an increase in revenue from OPKO Chile, Spain and EirGen. Revenue from products in 2017 also reflects $9.1 million of revenue from sales of Rayaldee, which was launched in the U.S. in November 2016. Revenue from transfer of intellectual property decreased as a result of $50.0 million of revenue from the initial payment in the VFMCRP Agreement for the year ended December 31, 2016, which was partially offset by $10.0 million of revenue from a milestone payment from our licensee, TESARO, for the year ended December 31, 2017. Revenue from transfer of intellectual property for the years ended December 31, 2017 and 2016 also reflects $66.5 million and $51.0 million, respectively, of revenue related to the Pfizer Transaction.
Costs of revenue. Costs of revenue for the year ended December 31, 2017 increased $8.6 million compared to the prior year. The decrease in cost of service revenue is attributable to decreased revenue at BioReference. The increase in cost of product revenue is attributable to an increase in revenue at OPKO Chile, Spain and EirGen and to cost of revenue related to sales of Rayaldee, which was launched in the U.S. in November 2016. Also included in cost of product revenue for the year ended December 31, 2017 is $5.4 million of inventory obsolescence expense related primarily to the launch of Rayaldee. Cost of revenue for the years ended December 31, 2017 and 2016 were as follows:
Cost of Revenue |
For the years ended December 31, |
||||||||||
(In thousands) |
2017 |
2016 |
Change |
||||||||
Cost of service revenue |
$ |
558,953 |
$ |
564,103 |
$ |
(5,150 |
) |
||||
Cost of product revenue |
61,177 |
47,379 |
13,798 |
||||||||
Total cost of revenue |
$ |
620,130 |
$ |
611,482 |
$ |
8,648 |
|||||
Selling, general and administrative expenses. Selling, general and administrative expenses for the years ended December 31, 2017 and 2016 were $414.6 million and $407.3 million, respectively. The increase in selling, general and administrative expenses was primarily due to costs related to the launch of Rayaldee and increased selling, general and administrative expenses at BioReference, which was partially offset by a decrease in severance costs. Included in selling, general and administrative expenses for the years ended December 31, 2017 and 2016 are $5.8 million and $17.9 million, respectively, of net severance costs for certain BioReference executives. These severance costs include $2.8 million and $8.9 million of expense related to the acceleration of stock option vesting for certain BioReference executives in 2017 and 2016, respectively. Selling, general and administrative expenses for the year ended December 31, 2017 also include $8.8 million of expense to write-off certain other current assets.
Selling, general and administrative expenses during the years ended December 31, 2017 and 2016, include equity-based compensation expense of $21.2 million and $33.4 million, respectively, including the expense related to the acceleration of stock option vesting for certain BioReference executives.
Research and development expenses. Research and development expenses for the years ended December 31, 2017 and 2016 were $126.4 million and $113.9 million, respectively. Research and development costs include external and internal expenses, partially offset by third-party grants and funding arising from collaboration agreements. External expenses include
73
clinical and non-clinical activities performed by contract research organizations, lab services, purchases of drug and diagnostic product materials and manufacturing development costs. We track external research and development expenses by individual program for phase 3 clinical trials for drug approval and PMAs for diagnostics tests, if any. Internal expenses include employee-related expenses including salaries, benefits and equity-based compensation expense. Other internal research and development expenses are incurred to support overall research and development activities and include expenses related to general overhead and facilities.
The following table summarizes the components of our research and development expenses:
For the years ended December 31, |
|||||||
2017 |
2016 |
||||||
External expenses: |
|||||||
Phase 3 clinical trials |
$ |
15,339 |
$ |
12,161 |
|||
Manufacturing expense for biological products |
47,737 |
35,985 |
|||||
PMA studies |
1,089 |
— |
|||||
Earlier-stage programs |
7,620 |
6,297 |
|||||
Research and development employee-related expenses |
29,970 |
28,676 |
|||||
Other internal research and development expenses |
24,680 |
30,752 |
|||||
Total research and development expenses |
$ |
126,435 |
$ |
113,871 |
|||
The increase in research and development expenses is primarily due to an increase in research and development expenses related to hGH-CTP, a long acting human growth hormone which was outlicensed to Pfizer in 2015, and to the acquisition of Transition Therapeutics in August 2016. Research and development expenses for the years ended December 31, 2017 and 2016 include equity-based compensation expenses of $5.1 million and $7.5 million, respectively. We expect our research and development expenses to increase as we continue to expand our research and development of potential future products.
Contingent consideration. Contingent consideration income (expense) for the years ended December 31, 2017 and 2016, were $3.4 million of income and $17.0 million of expense, respectively. The change in contingent consideration income (expense) was attributable to contingent consideration for OPKO Renal during the year ended December 31, 2017 due to changes in assumptions regarding the timing of achievement of future milestones of Rayaldee. The contingent consideration liabilities of $41.4 million at December 31, 2017 relate to potential amounts payable to former stockholders of CURNA, OPKO Diagnostics and OPKO Renal pursuant to our acquisition agreements in January 2011, October 2011 and March 2013, respectively.
Amortization of intangible assets. Amortization of intangible assets was $84.7 million and $64.4 million, respectively, for the years ended December 31, 2017 and 2016. Amortization expense reflects the amortization of acquired intangible assets with defined useful lives. Amortization of intangible assets for the years ended December 31, 2017 and 2016 includes $16.0 million and $8.0 million, respectively, of amortization expense related to intangible assets for Rayaldee. Upon the FDA’s approval of Rayaldee in June 2016, we reclassified $187.6 million of IPR&D related to Rayaldee from In-process research and development to Intangible assets, net in our Consolidated Balance Sheets and began to amortize that asset. Our indefinite lived IPR&D assets will not be amortized until the underlying development programs are completed. Upon obtaining regulatory approval by the U.S. FDA, the IPR&D assets will be accounted for as a finite-lived intangible asset and amortized on a straight-line basis over its estimated useful life.
Asset impairment charges. During the year ended December 31, 2017, we recognized an impairment charge of $13.2 million to write our intangible asset for VARUBI™ down to its estimated fair value.
Interest income. Interest income for the years ended December 31, 2017 and 2016, was not significant as our cash investment strategy emphasizes the security of the principal invested and fulfillment of liquidity needs.
Interest expense. Interest expense for the years ended December 31, 2017 and 2016, was $6.6 million and $7.4 million, respectively. Interest expense is principally related to interest incurred on the 2033 Senior Notes including amortization of related deferred financing costs and to the interest incurred on BioReference’s outstanding debt under its credit facility.
Fair value changes of derivative instruments, net. Fair value changes of derivative instruments, net for the years ended December 31, 2017 and 2016, were $0.1 million and $2.8 million of income, respectively. Fair value changes of derivative instruments, net reflects non-cash income related to the changes in the fair value of the embedded derivatives in the 2033 Senior Notes of $3.2 million and $7.0 million for the years ended December 31, 2017 and 2016, respectively. For the year ended December 31, 2017, we observed a decrease in the market price of our Common Stock which resulted in the decrease in
74
the estimated fair value of our embedded derivatives in the 2033 Senior Notes through the last valuation on February 1, 2017. Fair value changes of derivative instruments, net for the year ended December 31, 2017 also reflects $2.9 million of expense related to the change in the fair value of warrants and options to purchase additional shares of Neovasc, Inc. (“Neovasc”) and Xenetic Biosciences, Inc. (“Xenetic”). Fair value changes of derivative instruments, net for the year ended December 31, 2016 also reflects $4.2 million of expense related to the change in the fair value of warrants and options to purchase additional shares of Neovasc , Cocrystal Pharma, Inc. (“Cocrystal”), ARNO Therapeutics, Inc. (“ARNO”) and MabVax Therapeutics Holdings, Inc. (“MabVax”).
Other income and (expense), net. Other income and (expense), net for the years ended December 31, 2017 and 2016, were $10.5 million and $3.9 million of income, respectively. Other income for the year ended December 31, 2017 primarily consists of a $3.0 million gain on the sale of non-strategic assets at a wholly-owned BioReference subsidiary, a $1.5 million gain on the sale of certain available for sale investments, a $2.5 million gain in connection with the acquisition transaction between Eloxx Pharmaceuticals, Inc. and Sevion Therapeutics, Inc., and a $1.9 million gain in connection with the dilution of our equity method investment in VBI Vaccines Inc. (“VBI”). Other income (expense), net for the year ended December 31, 2016 primarily consists of a $2.5 million gain recognized in connection with the merger of SciVac Therapeutics Inc. (“STI”) and VBI, a $5.0 million gain recognized in connection with the settlement of a legal matter and foreign currency transaction gains recognized during the period, which was partially offset by a $4.8 million other-than-temporary impairment charge to write our investments in Xenetic, ARNO and RXi Pharmaceuticals Corporation (“RXi”) down to their respective fair values.
Income tax benefit (provision). Our income tax benefit (provision) for the years ended December 31, 2017 and 2016 was $(18.9) million, and $56.1 million, respectively. The change in income tax provision is primarily due to the establishment of valuation allowance against certain U.S. and non-U.S. deferred tax assets. As of December 31, 2017, the Company determined that it is more likely than not that certain U.S. and non-U.S. deferred tax assets will not be realized and recorded a valuation allowance of $28.7 million. On December 22, 2017, the Tax Act was enacted into law and the new legislation reduced the corporate income tax rate from 35% to 21% which required us to remeasure our U.S. deferred tax assets and liabilities and recognize the effect in the period of enactment, resulting in $31.8 million of expense, with an equal offset to valuation allowance.
Loss from investments in investees. We have made investments in other early stage companies that we perceive to have valuable proprietary technology and significant potential to create value for us as a shareholder or member. We account for these investments under the equity method of accounting, resulting in the recording of our proportionate share of their losses until our share of their loss exceeds our investment. Until the investees’ technologies are commercialized, if ever, we anticipate they will report a net loss. Loss from investments in investees was $14.5 million and $7.7 million for the years ended December 31, 2017 and 2016, respectively. The increase in Loss from investments in investees is attributable to losses recognized on our investment in Pharmsynthez in 2017.
75
LIQUIDITY AND CAPITAL RESOURCES
At December 31, 2018, we had cash and cash equivalents of approximately $96.5 million. Cash used in operations of $109.1 million during 2018 principally reflected expenses related to general and administrative activities of our corporate operations, research and development activities and commercialization activities related to Rayaldee. Cash used in investing activities primarily reflects capital expenditures of $27.9 million. Cash provided by financing activities primarily reflects the issuance of $55.0 million of 2023 Convertible Notes in February 2018 and the sale of approximately 26.5 million shares of our common stock for $92.5 million in November 2018. We have not generated sustained positive cash flow sufficient to offset our operating and other expenses and our primary source of cash has been from the public and private placement of stock, the issuance of the 2033 Senior Notes and 2023 Convertible Notes and credit facilities available to us.
In February 2019, we issued $200.0 million aggregate principal amount of the 2025 Convertible Notes in an underwritten public offering. The 2025 Convertible Notes will bear interest at a rate of 4.50% per year, payable semiannually in arrears on February 15 and August 15 of each year, beginning on August 15, 2019. The notes mature on February 15, 2025, unless earlier repurchased, redeemed or converted.
Holders may convert their 2025 Convertible Notes at their option at any time prior to the close of business on the business day immediately preceding November 15, 2024 only under the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2019 (and only during such calendar quarter), if the last reported sale price of our common stock for at least 20 trading days (whether or not consecutive) during a period of 30 consecutive trading days ending on the last trading day of the immediately preceding calendar quarter is greater than or equal to 130% of the conversion price on each applicable trading day; (2) during the five business day period after any five consecutive trading day period (the “measurement period”) in which the trading price per $1,000 principal amount of 2025 Convertible Notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price of our common stock and the conversion rate on each such trading day; (3) if we call any or all of the 2025 Convertible Notes for redemption, at any time prior to the close of business on the scheduled trading day immediately preceding the redemption date; or (4) upon the occurrence of specified corporate events. On or after November 15, 2024, until the close of business on the business day immediately preceding the maturity date, holders of the 2025 Convertible Notes may convert their notes at any time, regardless of the foregoing circumstances. Upon conversion, we will pay or deliver, as the case may be, cash, shares of our common stock, or a combination of cash and shares of our common stock, at our election.
The conversion rate for the notes will initially be 236.7424 shares of common stock per $1,000 principal amount of 2025 Convertible Notes (equivalent to an initial conversion price of approximately $4.22 per share of common stock). The conversion rate for the 2025 Convertible Notes will be subject to adjustment in some events but will not be adjusted for any accrued and unpaid interest.
On November 8, 2018, we entered into stock purchase agreements with certain investors pursuant to which we agreed to sell to such investors in private placements an aggregate of approximately 26.5 million shares of our common stock at a purchase price of $3.49 per share, which was the closing bid price of our common stock on the NASDAQ on such date, for an aggregate purchase price of $92.5 million. The investors in the private placements include an affiliate of Dr. Phillip Frost, our Chairman and Chief Executive Officer ($70 million), and Dr. Jane Hsiao, our Vice Chairman and Chief Technical Officer ($2 million). We intend to use the proceeds from the private placements for general corporate purposes.
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. The credit agreement was terminated on or around February 20, 2019 and amounts borrowed during 2019 were repaid from the proceeds of the 2025 Notes offering. Borrowings under the line of credit bore interest at a rate of 10% per annum and could be repaid and reborrowed at any time. The credit agreement included various customary remedies for the lender following an event of default, including the acceleration of repayment of outstanding amounts under line of credit. The line of credit would have matured on November 8, 2023. As of as of December 31, 2018, no funds were borrowed under the line of credit.
On February 1, 2019, approximately $28.8 million aggregate principal amount of 2033 Senior Notes were tendered by holders pursuant to such holders’ option to require us to repurchase the 2033 Senior Notes. At December 31, 2018, $31.9 million principal amount of 2033 Senior Notes was outstanding. Holders of the remaining $3.0 million principal amount of the 2033 Senior Notes may require us to repurchase the 2033 Senior Notes for 100% of their principal amount, plus accrued and unpaid interest, again on February 1, 2023 and February 1, 2028, or following the occurrence of a fundamental change as defined in the indenture governing the 2033 Senior Notes.
In August 2017, we entered into a Commitment Letter (the “Commitment Letter”) with Veterans Accountable Care Group, LLC (“VACG”) in connection with the submission of a bid by its affiliate, the Veterans Accountable Care Organization, LLC (“VACO”) in response to a request for proposal (“RFP”) from the Veterans Health Administration (“VA”) regarding its
76
Community Care Network. We were notified in January 2019 that the bid was awarded to a third party. If VACO were to have been successful in its bid, we would have acquired a fifteen percent (15%) membership interest in VACO. In addition, BioReference, our wholly-owned subsidiary, would have provided laboratory services for the Community Care Network, a region which currently includes approximately 2,133,000 veterans in the states of Massachusetts, Maine, New Hampshire, Vermont, New York, Pennsylvania, New Jersey, Rhode Island, Connecticut, Maryland, Virginia, West Virginia, and North Carolina.
Pursuant to the Commitment Letter and had VACO been successful in its bid, we committed to provide, or to arrange from a third-party lender, a line of credit for VACG in the amount of $50.0 million (the “Facility”). Funds drawn under the Facility would be contributed by VACG to VACO in order to satisfy the financial stability requirement of VACO in connection with its submission of the RFP. VACG would not be permitted to draw down on the Facility unless and until the VHA awards a contract to VACO. The Facility would have a maturity of 5 years. Interest on the Facility would be payable at a rate equal to 6.5% per annum, payable quarterly in arrears. The Facility would be subject to the negotiation of definitive documentation conditions customary for transactions of such type and otherwise acceptable to VACG and the lender under the Facility.
As of December 31, 2018, the total availability under our Credit Agreement with JPMorgan Chase Bank, N.A. (“CB”) and our lines of credit with financial institutions in Chile and Spain was $129.4 million, of which $108.7 million was used and outstanding as of December 31, 2018. The weighted average interest rate on these lines of credit is approximately 4.7%. These lines of credit are short-term and are used primarily as a source of working capital. The highest balance at any time during the year ended December 31, 2018, was $114.9 million. We intend to continue to enter into these lines of credit as needed. There is no assurance that these lines of credit or other funding sources will be available to us on acceptable terms, or at all, in the future.
In February 2018, we issued the 2023 Convertible Notes in the aggregate principal amount of $55.0 million. The 2023 Convertible Notes mature 5 years from the date of issuance. Each holder of a 2023 Convertible Note has the option, from time to time, to convert all or any portion of the outstanding principal balance of such 2023 Convertible Note, together with accrued and unpaid interest thereon, into shares of our Common Stock, par value $0.01 per share, at a conversion price of $5.00 per share of common stock. We may redeem all or any part of the then issued and outstanding 2023 Convertible Notes, together with accrued and unpaid interest thereon, pro ratably among the holders, upon no fewer than 30 days, and no more than 60 days, notice to the holders. The 2023 Convertible Notes contain customary events of default and representations and warranties of OPKO.
The issuance of the 2023 Convertible Notes and the issuance of the Shares, if any, upon conversion thereof was not, and will not be, respectively, registered under the Securities Act, pursuant to the exemption provided by Section 4(a)(2) thereof, and we have not agreed to register the shares of common stock issuable upon conversion if or when such shares are issued. Purchasers of the 2023 Convertible Notes include Dr. Hsiao and an affiliate of Dr. Frost.
On October 12, 2017, EirGen, our wholly-owned subsidiary, and Japan Tobacco Inc. (“JT”) entered into a Development and License Agreement (the “JT Agreement”) granting JT the exclusive rights for the development and commercialization of Rayaldee in Japan (the “JT Territory”). The license grant to JT covers the therapeutic and preventative use of Rayaldee for (i) SHPT in non-dialysis and dialysis patients with CKD, (ii) rickets, and (iii) osteomalacia, as well as such additional indications as may be added to the scope of the license subject to the terms of the JT Agreement. In connection with the transaction, OPKO received an initial upfront payment of $6 million, and OPKO received another $6 million upon the initiation of OPKO’s phase 2 study for Rayaldee in dialysis patients in the U.S. in September 2018. OPKO is also eligible to receive up to an additional aggregate amount of $31 million upon the achievement of certain regulatory and development milestones by JT for Rayaldee in the JT Territory, and $75 million upon the achievement of certain sales based milestones by JT in the JT Territory. OPKO will also receive tiered, double digit royalty payments at rates ranging from low double digits to mid-teens on sales of Rayaldee within the JT Territory. JT will, at its sole cost and expense, be responsible for performing all development activities necessary to obtain all regulatory approvals for Rayaldee in Japan and for all commercial activities pertaining to Rayaldee in Japan.
In May 2016, EirGen, our wholly-owned subsidiary, partnered with VFMCRP through a Development and License Agreement for the development and commercialization of Rayaldee in Europe, Canada, Mexico, Australia, South Korea and certain other international markets. The license to VFMCRP potentially covers all therapeutic and prophylactic uses of the product in human patients, provided that initially the license is for the use of the product for the treatment or prevention of SHPT related to patients with stage 3 or 4 CKD and vitamin D insufficiency/deficiency (“VFMCRP Initial Indication”). We have received non-refundable and non-creditable payments of $52 million and are eligible to receive up to an additional $230 million upon the achievement of certain regulatory and sales-based milestones. In addition, we are eligible to receive tiered royalties on sales of the product at percentage rates that range from the mid-teens to the mid-twenties or a minimum royalty, whichever is greater, upon commencement of sales of the product.
77
As part of the arrangement, the companies will share responsibility for the conduct of trials specified within an agreed-upon development plan, with each company leading certain activities within the plan. For the initial development plan, the companies have agreed to certain cost sharing arrangements. VFMCRP will be responsible for all other development costs that VFMCRP considers necessary to develop the product for the VFMCRP Initial Indication in the VFMCRP Territory except as otherwise provided in the VFMCRP Agreement. EirGen also granted to VFMCRP an option to acquire an exclusive license to use, import, offer for sale, sell, distribute and commercialize the product in the U.S. for treatment of SHPT in dialysis patients with stage 5 CKD and vitamin D insufficiency (the “Dialysis Indication”). Upon exercise of the Option, VFMCRP will reimburse EirGen for all of the development costs incurred by EirGen with respect to the product for the Dialysis Indication in the U.S. VFMCRP would also pay EirGen up to an additional aggregate amount of $555 million upon the achievement of certain milestones and would be obligated to pay royalties on sales of the product at percentage rates that range from the mid-teens to the mid-twenties or a minimum royalty, whichever is greater, upon commencement of sales of the product.
In January 2015, we partnered with Pfizer through a worldwide agreement for the development and commercialization of our long-acting hGH-CTP for the treatment of GHD in adults and children, as well as for the treatment of growth failure in children born SGA. Under the terms of the agreements with Pfizer, we received non-refundable and non-creditable upfront payments of $295 million in 2015 and are eligible to receive up to an additional $275 million upon the achievement of certain regulatory milestones. Pfizer received the exclusive license to commercialize hGH-CTP worldwide. In addition, we are eligible to receive initial tiered royalty payments associated with the commercialization of hGH-CTP for Adult GHD with percentage rates ranging from the high teens to mid-twenties. Upon the launch of hGH-CTP for Pediatric GHD in certain major markets, the royalties will transition to regional, tiered gross profit sharing for both hGH-CTP and Pfizer’s Genotropin®.
Under the agreement, we agreed to lead the clinical activities and will be responsible for funding the development programs for the key indications, which includes Adult and Pediatric GHD and Pediatric SGA. Pfizer agreed to be responsible for all development costs for additional indications as well as all post-marketing studies. In addition, Pfizer agreed to fund the commercialization activities for all indications and lead the manufacturing activities covered by the global development plan. The agreement obligated us to fund development up to an agreed cap
In December 2016, we announced preliminary topline data from our phase 3, double blind, placebo controlled study of hGH-CTP in adults with GHD. Although there was no statistically significant difference between hGH-CTP and placebo on the primary endpoint of change in trunk fat mass from baseline to 26 weeks, after unblinding the study, we identified an exceptional value of trunk fat mass reduction in the placebo group that may have affected the primary outcome. We have completed post-hoc sensitivity analyses to evaluate the influence of outliers on the primary endpoint results using multiple statistical approaches. Analyses that excluded outliers showed a statistically significant difference between hGH-CTP and placebo on the change in trunk fat mass. Additional analyses that did not exclude outliers showed mixed results. Following completion of the analyses, OPKO and Pfizer agreed that OPKO may proceed to discuss a possible BLA submission with the FDA. We believe there is a path for submission in which the FDA may assess the totality of the data, including all relevant efficacy and safety data in adult and pediatric patients. We will continue to assess the regulatory strategy for the adult indication going forward, including the timing of a possible submission.
In August 2018, we announced that we had completed enrollment in a global phase 3 study of hGH-CTP in growth hormone deficient children. The development project for hGH-CTP has exceeded our original estimates and will result in additional expenses beyond our estimates and the agreed development cap. If we are unable to reach an agreement with Pfizer regarding cost sharing for overruns, as well as other obligations, including development obligations, it could have a material adverse impact on the expected benefits of the Pfizer transaction and our overall financial condition. See “Risk Factors - Our exclusive worldwide agreement with Pfizer Inc. is important to our business”. If we do not successfully develop hGH- CTP and/or Pfizer Inc. were to terminate the agreement or not successfully commercialize hGH-CTP for any reason, our business would be adversely affected.
We are constructing a research, development and manufacturing center in Waterford, Ireland, for which we will incur between $40 million and $50 million for the construction and validation of the facility. Construction of the facility began in the fourth quarter of 2016 with expected completion in 2019. Currently, we plan to fund the project from cash on hand or from third party funding sources that may be available to us. Through December 31, 2018, the cumulative expenditures we incurred to date on the construction of the facility was approximately $39.1 million.
In connection with our acquisitions of CURNA, OPKO Diagnostics and OPKO Renal, we agreed to pay future consideration to the sellers upon the achievement of certain events, including up to an additional $19.1 million in shares of our Common Stock to the former stockholders of OPKO Diagnostics upon and subject to the achievement of certain milestones; and up to an additional $125.0 million in either shares of our Common Stock or cash, at our option subject to the achievement of certain milestones, to the former shareholders of OPKO Renal.
78
In November 2015, BioReference and certain of its subsidiaries entered into a credit agreement with JPMorgan Chase Bank, N.A. (“CB”), as lender and administrative agent, as amended (the “Credit Agreement”). The Credit Agreement provides for a $175.0 million secured revolving credit facility and includes a $20.0 million sub-facility for swingline loans and a $20.0 million sub-facility for the issuance of letters of credit. BioReference may increase the credit facility to up to $275.0 million on a secured basis, subject to the satisfaction of specified conditions. The Credit Agreement matures on November 5, 2020 and is guaranteed by all of BioReference’s domestic subsidiaries. The Credit Agreement is also secured by substantially all assets of BioReference and its domestic subsidiaries, as well as a non-recourse pledge by us of our equity interest in BioReference. Availability under the Credit Agreement is based on a borrowing base comprised of eligible accounts receivables of BioReference and certain of its subsidiaries, as specified therein.
We expect to continue to incur substantial research and development expenses, including expenses related to the hiring of personnel and additional clinical trials. We expect that selling, general and administrative expenses will also increase as we expand our sales, marketing and administrative staff and add infrastructure.
We believe that the cash and cash equivalents on hand at December 31, 2018, the amounts available to be borrowed under our lines of credit and the proceeds from the issuance of the 2025 Convertible Notes in February 2019 are sufficient to meet our anticipated cash requirements for operations and debt service beyond the next 12 months. We based this estimate on assumptions that may prove to be wrong or are subject to change, and we may be required to use our available cash resources sooner than we currently expect. If we acquire additional assets or companies, accelerate our product development programs or initiate additional clinical trials, we will need additional funds. Our future cash requirements will depend on a number of factors, including our relationship with Pfizer, the commercial success of Rayaldee, BioReference’s financial performance, possible acquisitions, the continued progress of research and development of our product candidates, the timing and outcome of clinical trials and regulatory approvals, the costs involved in preparing, filing, prosecuting, maintaining, defending, and enforcing patent claims and other intellectual property rights, the status of competitive products, the availability of financing, our success in developing markets for our product candidates and results of government investigations, payor claims, and legal proceedings that may arise, including, without limitation class action and derivative litigation to which we are subject, and our ability to obtain insurance coverage for such claims. If we are not able to secure additional funding when needed, we may have to delay, reduce the scope of, or eliminate one or more of our clinical trials or research and development programs or possible acquisitions or reduce our marketing or sales efforts or cease operations.
The following table provides information as of December 31, 2018, with respect to the amounts and timing of our known contractual obligation payments due by period.
|
Contractual obligations
(In thousands)
|
2019 |
2020 |
2021 |
2022 |
2023 |
Thereafter |
Total |
|||||||||||||||||||||
Open purchase orders |
$ |
98,409 |
$ |
87 |
$ |
— |
$ |
— |
$ |
— |
$ |
— |
$ |
98,496 |
||||||||||||||
Operating leases |
18,387 |
11,535 |
7,385 |
4,073 |
2,446 |
1,650 |
45,476 |
|||||||||||||||||||||
Capital leases |
3,037 |
2,635 |
1,924 |
829 |
473 |
— |
8,898 |
|||||||||||||||||||||
2033 Senior Notes |
31,562 |
— |
— |
— |
— |
55,000 |
86,562 |
|||||||||||||||||||||
Deferred payments |
5,000 |
— |
— |
— |
— |
— |
5,000 |
|||||||||||||||||||||
Mortgages and other debts payable |
1,872 |
1,176 |
514 |
414 |
4,277 |
— |
8,253 |
|||||||||||||||||||||
Lines of credit |
3,501 |
— |
105,198 |
— |
— |
— |
108,699 |
|||||||||||||||||||||
Interest commitments |
214 |
1,213 |
122 |
85 |
63 |
13,990 |
15,687 |
|||||||||||||||||||||
Total |
$ |
161,982 |
$ |
16,646 |
$ |
115,143 |
$ |
5,401 |
$ |
7,259 |
$ |
70,640 |
$ |
377,071 |
||||||||||||||
The preceding table does not include information where the amounts of the obligations are not currently determinable, including the following:
- Contractual obligations in connection with clinical trials, which span over two years, and that depend on patient enrollment. The total amount of expenditures is dependent on the actual number of patients enrolled and as such, the contracts do not specify the maximum amount we may owe.
- Product license agreements effective during the lesser of 15 years or patent expiration whereby payments and amounts are determined by applying a royalty rate on uncapped future sales.
79
- Contingent consideration that includes payments upon achievement of certain milestones including meeting development milestones such as the completion of successful clinical trials, NDA approvals by the FDA and revenue milestones upon the achievement of certain revenue targets all of which are anticipated to be paid within the next seven years and are payable in either shares of our Common Stock or cash, at our option, and that may aggregate up to $159.1 million.
80
CRITICAL ACCOUNTING POLICIES AND ESTIMATES
Accounting estimates. The preparation of financial statements in conformity with accounting principles generally accepted in the U.S. requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ significantly from these estimates.
Goodwill and intangible assets. Goodwill and other intangible assets, including IPR&D, acquired in business combinations, licensing and other transactions at both December 31, 2018 and 2017 was $2.0 billion, representing approximately 80% and 79% of total assets, respectively.
Assets acquired and liabilities assumed in business combinations, licensing and other transactions are generally recognized at the date of acquisition at their respective fair values. Any excess of the purchase price over the estimated fair values of the net assets acquired is recognized as goodwill. We determined the fair value of intangible assets, including IPR&D, using the “income method.” This method starts with a forecast of net cash flows, risk adjusted for estimated probabilities of technical and regulatory success (for IPR&D) and adjusted to present value using an appropriate discount rate that reflects the risk associated with the cash flow streams. All assets are valued from a market participant view which might be different than our specific views. The valuation process is very complex and requires significant input and judgment using internal and external sources. Although a valuation is required to be finalized within a one-year period, it must consider all and only those facts and evidence which existed at the acquisition date. The most complex and judgmental matters applicable to the valuation process are summarized below:
• |
Unit of account – Most intangible assets are valued as single global assets rather than multiple assets for each jurisdiction or indication after considering the development stage, expected levels of incremental costs to obtain additional approvals, risks associated with further development, amount and timing of benefits expected to be derived in the future, expected patent lives in various jurisdictions and the intention to promote the asset as a global brand. |
• |
Estimated useful life – The asset life expected to contribute meaningful cash flows is determined after considering all pertinent matters associated with the asset, including expected regulatory approval dates (if unapproved), exclusivity periods and other legal, regulatory or contractual provisions as well as the effects of any obsolescence, demand, competition, and other economic factors, including barriers to entry. |
• |
Probability of Technical and Regulatory Success (“PTRS”) Rate – PTRS rates are determined based upon industry averages considering the respective program’s development stage and disease indication and adjusted for specific information or data known at the acquisition date. Subsequent clinical results or other internal or external data obtained could alter the PTRS rate and materially impact the estimated fair value of the intangible asset in subsequent periods leading to impairment charges. |
• |
Projections – Future revenues are estimated after considering many factors such as initial market opportunity, pricing, sales trajectories to peak sales levels, competitive environment and product evolution. Future costs and expenses are estimated after considering historical market trends, market participant synergies and the timing and level of additional development costs to obtain the initial or additional regulatory approvals, maintain or further enhance the product. We generally assume initial positive cash flows to commence shortly after the receipt of expected regulatory approvals which typically may not occur for a number of years. Actual cash flows attributed to the project are likely to be different than those assumed since projections are subjected to multiple factors including trial results and regulatory matters which could materially change the ultimate commercial success of the asset as well as significantly alter the costs to develop the respective asset into commercially viable products. |
• |
Tax rates – The expected future income is tax effected using a market participant tax rate. In determining the tax rate, we consider the jurisdiction in which the intellectual property is held and location of research and manufacturing infrastructure. We also consider that any repatriation of earnings would likely have U.S. tax consequences. |
• |
Discount rate – Discount rates are selected after considering the risks inherent in the future cash flows; the assessment of the asset’s life cycle and the competitive trends impacting the asset, including consideration of any technical, legal, regulatory, or economic barriers to entry, as well as expected changes in standards of practice for indications addressed by the asset. |
Goodwill was $700.2 million and $717.1 million, respectively, at December 31, 2018 and 2017. Goodwill is tested at least annually for impairment or when events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, by assessing qualitative factors or performing a quantitative analysis in determining whether it is more
81
likely than not that its fair value exceeds the carrying value. Examples of qualitative factors include our share price, our financial performance compared to budgets, long-term financial plans, macroeconomic, industry and market conditions as well as the excess of fair value over the carrying value of net assets from the annual impairment test previously performed. Due to the loss of a significant customer in 2018, we recorded a goodwill impairment charge of $11.7 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2018 to write the carrying amount of the FineTech reporting unit down to its estimated fair value. No goodwill impairment was recorded for the years ended December 31, 2017 and 2016 as a result of our testing.
The estimated fair value of a reporting unit is highly sensitive to changes in projections and assumptions; therefore, in some instances, changes in these assumptions could potentially lead to impairment. We perform sensitivity analyses around our assumptions in order to assess the reasonableness of the assumptions and the results of our testing. Ultimately, future potential changes in these assumptions may impact the estimated fair value of a reporting unit and cause the fair value of the reporting unit to be below its carrying value.
We believe that our estimates are consistent with assumptions that marketplace participants would use in their estimates of fair value. However, if actual results are not consistent with our estimates and assumptions, we may be exposed to an impairment charge that could be material. If we are unable to generate profits and cash flows from BioReference, or we are unable successfully commercialize or obtain reimbursement for our 4Kscore test and our other diagnostic products under development, or other changes in projections and assumptions negatively impact our forecast of net cash flows, we may be exposed to a material impairment charge related to the goodwill at BioReference.
Intangible assets, net were $1.3 billion and $1.3 billion, including IPR&D of $635.6 million and $647.3 million, respectively, at December 31, 2018 and 2017. Intangible assets are tested for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, although IPR&D is required to be tested at least annually until the project is completed or abandoned. Upon obtaining regulatory approval, the IPR&D asset is then accounted for as a finite-lived intangible asset and amortized on a straight-line basis over its estimated useful life. If the project is abandoned, the IPR&D asset is charged to expense.
IPR&D is tested for impairment by assessing qualitative factors or performing a quantitative analysis in determining whether it is more likely than not that its fair value exceeds the carrying amount. If the carrying amount of the IPR&D exceeds its fair value, an impairment loss shall be recognized in an amount equal to that excess. Intangible assets with defined lives are tested for impairment by a comparison of the carrying amount of the asset to its estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future cash flows, then an impairment charge is recognized for the amount by which the carrying amount of the asset exceeds the fair value of the asset. We recorded an impairment charge of $10.1 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2018 to write our IPR&D assets for Alpharen and OPK88004 down to their estimated fair value as a result of our testing. We recorded an impairment charge of $13.2 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2017 to write our intangible asset for VARUBI™ down to its estimated fair value as a result of our testing.
Intangible assets are highly vulnerable to impairment charges, particularly newly acquired assets for recently launched products and IPR&D. These assets are initially measured at fair value and therefore any reduction in expectations used in the valuations could potentially lead to impairment. Some of the more common potential risks leading to impairment include competition, earlier than expected loss of exclusivity, pricing pressures, adverse regulatory changes or clinical trial results, delay or failure to obtain regulatory approval and additional development costs, inability to achieve expected synergies, higher operating costs, changes in tax laws and other macro-economic changes.
Considering the high risk nature of research and development and the industry’s success rate of bringing developmental compounds to market, IPR&D impairment charges are likely to occur in future periods. IPR&D is closely monitored and assessed each period for impairment indicators.
In August 2018, we announced that we had completed enrollment in a global phase 3 study of hGH-CTP in growth hormone deficient children. The development project for hGH-CTP has exceeded our original estimates and will result in additional expenses beyond our estimates and the agreed development cap. If we are unable to reach an agreement with Pfizer regarding cost sharing for overruns, as well as other obligations, including development obligations, it could have a material adverse impact on the expected benefits of the Pfizer transaction. If we are unable to successfully develop hGH-CTP, or changes in projections and assumptions negatively impact our forecast of net cash flows, we may be exposed to a material impairment charge related to the IPR&D for hGH-CTP.
We amortize intangible assets with definite lives on a straight-line basis over their estimated useful lives, ranging from 3 to 20 years. We use the straight-line method of amortization as there is no reliably determinable pattern in which the economic
82
benefits of our intangible assets are consumed or otherwise used up. Amortization expense was $67.9 million and $71.5 million for the years ended December 31, 2018 and 2017, respectively.
Revenue recognition. Effective January 1, 2018, we adopted Accounting Standards Codification Topic 606, Revenue from Contracts with Customers. We generate revenues from services, products and intellectual property as follows:
Revenue from services. Revenue for laboratory services is recognized at the time test results are reported, which approximates when services are provided and the performance obligations are satisfied. Services are provided to patients covered by various third-party payor programs including various managed care organizations, as well as the Medicare and Medicaid programs. Billings for services are included in revenue net of allowances for contractual discounts, allowances for differences between the amounts billed and estimated program payment amounts, and implicit price concessions provided to uninsured patients which are all elements of variable consideration.
The following are descriptions of our payors for laboratory services:
Healthcare Insurers. Reimbursements from healthcare insurers are based on negotiated fee-for-service schedules. Revenues consist of amounts billed, net of contractual allowances for differences between amounts billed and the estimated consideration we expect to receive from such payors, which considers historical denial and collection experience and the terms of our contractual arrangements. Adjustments to the allowances, based on actual receipts from the third-party payors, are recorded upon settlement.
Government Payors. Reimbursements from government payors are based on fee-for-service schedules set by governmental authorities, including traditional Medicare and Medicaid. Revenues consist of amounts billed, net of contractual allowances for differences between amounts billed and the estimated consideration we expect to receive from such payors, which considers historical denial and collection experience and the terms of our contractual arrangements. Adjustments to the allowances, based on actual receipts from the government payors, are recorded upon settlement.
Client Payors. Client payors include physicians, hospitals, employers, and other institutions for which services are performed on a wholesale basis, and are billed and recognized as revenue based on negotiated fee schedules.
Patients. Uninsured patients are billed based on established patient fee schedules or fees negotiated with physicians on behalf of their patients. Insured patients (including amounts for coinsurance and deductible responsibilities) are billed based on fees negotiated with healthcare insurers. Collection of billings from patients is subject to credit risk and ability of the patients to pay. Revenues consist of amounts billed net of discounts provided to uninsured patients in accordance with our policies and implicit price concessions. Implicit price concessions represent differences between amounts billed and the estimated consideration that we expect to receive from patients, which considers historical collection experience and other factors including current market conditions. Adjustments to the estimated allowances, based on actual receipts from the patients, are recorded upon settlement.
The complexities and ambiguities of billing, reimbursement regulations and claims processing, as well as issues unique to Medicare and Medicaid programs, require us to estimate the potential for retroactive adjustments as an element of variable consideration in the recognition of revenue in the period the related services are rendered. Actual amounts are adjusted in the period those adjustments become known. For the years ended December 31, 2018 and 2017, revenue reductions due to changes in estimates of implicit price concessions for performance obligations satisfied in prior periods of $22.8 million and $66.0 million, respectively, were recognized.
Third-party payors, including government programs, may decide to deny payment or recoup payments for testing they contend were improperly billed or not medically necessary, against their coverage determinations, or for which they believe they have otherwise overpaid (including as a result of their own error), and we may be required to refund payments already received. Our revenues may be subject to retroactive adjustment as a result of these factors among others, including without limitation, differing interpretations of billing and coding guidance and changes by government agencies and payors in interpretations, requirements, and “conditions of participation” in various programs. We have processed requests for recoupment from third-party payors in the ordinary course of our business, and it is likely that we will continue to do so in the future. If a third-party payer denies payment for testing or recoups money from us in a later period, reimbursement revenue for our testing could decline.
As an integral part of our billing compliance program, we periodically assess our billing and coding practices, respond to payor audits on a routine basis, and investigate reported failures or suspected failures to comply with federal and state healthcare reimbursement requirements, as well as overpayment claims which may arise from time to time without fault on the part of the Company. We may have an obligation to reimburse Medicare, Medicaid, and third-party payors for overpayments
83
regardless of fault. We have periodically identified and reported overpayments, reimbursed payors for overpayments and taken appropriate corrective action.
Settlements with third-party payors for retroactive adjustments due to audits, reviews or investigations are also considered variable consideration and are included in the determination of the estimated transaction price for providing services. These settlements are estimated based on the terms of the payment agreement with the payor, correspondence from the payor and our historical settlement activity, including an assessment of the probability a significant reversal of cumulative revenue recognized will occur when the uncertainty is subsequently resolved. Estimated settlements are adjusted in future periods as adjustments become known (that is, new information becomes available), or as years are settled or are no longer subject to such audits, reviews, and investigations.
Revenue from products. We recognize revenue from product sales when a customer obtains control of promised goods or services. The amount of revenue that is recorded reflects the consideration that we expect to receive in exchange for those goods or services. Our estimates for sales returns and allowances are based upon the historical patterns of product returns and allowances taken, matched against the sales from which they originated, and our evaluation of specific factors that may increase or decrease the risk of product returns. Product revenues are recorded net of estimated rebates, chargebacks, discounts, co-pay assistance and other deductions (collectively, “Sales Deductions”) as well as estimated product returns which are all elements of variable consideration. Allowances are recorded as a reduction of revenue at the time product revenues are recognized. The actual amounts of consideration ultimately received may differ from our estimates. If actual results in the future vary from our estimates, we will adjust these estimates, which would affect Revenue from products in the period such variances become known.
We launched Rayaldee in the U.S. through our dedicated renal sales force in November 2016. Rayaldee is distributed in the U.S. principally through the retail pharmacy channel, which initiates with the largest wholesalers in the U.S. (collectively, “Rayaldee Customers”). In addition to distribution agreements with Rayaldee Customers, we have entered into arrangements with many healthcare providers and payors that provide for government-mandated and/or privately-negotiated rebates, chargebacks and discounts with respect to the purchase of Rayaldee.
We recognize revenue for shipments of Rayaldee at the time of delivery to customers after estimating Sales Deductions and product returns as elements of variable consideration utilizing historical information and market research projections. For the years ended December 31, 2018, and 2017, we recognized $20.3 million and $9.1 million in net product revenue from sales of Rayaldee.
The following table presents an analysis of product sales allowances and accruals as contract liabilities for the year ended December 31, 2018:
(In thousands) |
Chargebacks, discounts, rebates and fees |
Governmental |
Returns |
Total |
||||||||||||
Balance at December 31, 2017 |
$ |
233 |
$ |
348 |
$ |
437 |
$ |
1,018 |
||||||||
Provision related to current period sales |
5,704 |
10,061 |
680 |
16,445 |
||||||||||||
Credits or payments made |
(4,621 |
) |
(8,319 |
) |
(480 |
) |
(13,420 |
) |
||||||||
Balance at December 31, 2018 |
$ |
1,316 |
$ |
2,090 |
$ |
637 |
$ |
4,043 |
||||||||
Total gross Rayaldee sales
|
$ |
36,715 |
||||||||||||||
Provision for Rayaldee sales allowances and accruals as a percentage of gross Rayaldee sales
|
45 |
% |
||||||||||||||
Taxes collected from customers related to revenues from services and revenues from products are excluded from revenues.
Revenue from intellectual property. We recognize revenues from the transfer of intellectual property generated through license, development, collaboration and/or commercialization agreements. The terms of these agreements typically include payment to us for one or more of the following: non-refundable, up-front license fees; development and commercialization milestone payments; funding of research and/or development activities; and royalties on sales of licensed products. Revenue is recognized upon satisfaction of a performance obligation by transferring control of a good or service to the customer.
For research, development and/or commercialization agreements that result in revenues, we identify all material performance obligations, which may include a license to intellectual property and know-how, and research and development activities. In order to determine the transaction price, in addition to any upfront payment, we estimate the amount of variable
84
consideration at the outset of the contract either utilizing the expected value or most likely amount method, depending on the facts and circumstances relative to the contract. We constrain (reduce) our estimates of variable consideration such that it is probable that a significant reversal of previously recognized revenue will not occur throughout the life of the contract. When determining if variable consideration should be constrained, we consider whether there are factors outside of our control that could result in a significant reversal of revenue. In making these assessments, we consider the likelihood and magnitude of a potential reversal of revenue. These estimates are re-assessed each reporting period as required.
Upfront License Fees: If a license to our intellectual property is determined to be functional intellectual property distinct from the other performance obligations identified in the arrangement, we recognize revenue from nonrefundable, upfront license fees based on the relative value prescribed to the license compared to the total value of the arrangement. The revenue is recognized when the license is transferred to the customer and the customer is able to use and benefit from the license. For licenses that are not distinct from other obligations identified in the arrangement, we utilize judgment to assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time. If the combined performance obligation is satisfied over time, we apply an appropriate method of measuring progress for purposes of recognizing revenue from nonrefundable, upfront license fees. We evaluate the measure of progress each reporting period and, if necessary, adjust the measure of performance and related revenue recognition.
Development and Regulatory Milestone Payments: Depending on facts and circumstances, we may conclude that it is appropriate to include the milestone in the estimated transaction price or that it is appropriate to fully constrain the milestone. A milestone payment is included in the transaction price in the reporting period that we conclude that it is probable that recording revenue in the period will not result in a significant reversal in amounts recognized in future periods. We may record revenues from certain milestones in a reporting period before the milestone is achieved if we conclude that achievement of the milestone is probable and that recognition of revenue related to the milestone will not result in a significant reversal in amounts recognized in future periods. We record a corresponding contract asset when this conclusion is reached. Milestone payments that have been fully constrained are not included in the transaction price to date. These milestones remain fully constrained until we conclude that achievement of the milestone is probable and that recognition of revenue related to the milestone will not result in a significant reversal in amounts recognized in future periods. We re-evaluate the probability of achievement of such development milestones and any related constraint each reporting period. We adjust our estimate of the overall transaction price, including the amount of revenue recorded, if necessary.
Research and Development Activities: If we are entitled to reimbursement from our customers for specified research and development expenses, we account for them as separate performance obligations if distinct. We also determine whether the research and development funding would result in revenues or an offset to research and development expenses in accordance with provisions of gross or net revenue presentation. The corresponding revenues or offset to research and development expenses are recognized as the related performance obligations are satisfied.
Sales-based Milestone and Royalty Payments: Our customers may be required to pay us sales-based milestone payments or royalties on future sales of commercial products. We recognize revenues related to sales-based milestone and royalty payments upon the later to occur of (i) achievement of the customer’s underlying sales or (ii) satisfaction of any performance obligation(s) related to these sales, in each case assuming the license to our intellectual property is deemed to be the predominant item to which the sales-based milestones and/or royalties relate.
Other Potential Products and Services: Arrangements may include an option for license rights, future supply of drug substance or drug product for either clinical development or commercial supply at the licensee’s election. We assess if these options provide a material right to the licensee and if so, they are accounted for as separate performance obligations at the inception of the contract and revenue is recognized only if the option is exercised and products or services are subsequently delivered or when the rights expire. If the promise is based on market terms and not considered a material right, the option is accounted for if and when exercised. If we are entitled to additional payments when the licensee exercises these options, any additional payments are generally recorded in license or other revenues when the licensee obtains control of the goods, which is upon delivery.
For the years ended December 31, 2018, 2017 and 2016 we recorded $69.9 million, $75.5 million and $105.5 million of revenue from the transfer of intellectual property, respectively. For the year ended December 31, 2018, revenue from the transfer of intellectual property included $60.0 million related to the Pfizer Transaction and $2.0 million related to a milestone payment from our licensee, Vifor Fresenius Medical Care Renal Pharma Ltd (“VFMCRP”). For the year ended December 31, 2017, revenue from the transfer of intellectual property included $61.2 million related to the Pfizer Transaction and $10.0 million related to a milestone payment from our licensee, TESARO, Inc. (“TESARO”). For the year ended December 31, 2016, revenue from the transfer of intellectual property included $50.0 million related to the our agreement with VFMCRP and $47.3 million related to the Pfizer Transaction. Refer to Note 15. Total contract liabilities included in Accrued expenses and
85
Other long-term liabilities was $91.1 million and $152.3 million at December 31, 2018 and December 31, 2017, respectively. The contract liability balance at December 31, 2018 and 2017 relates primarily to the Pfizer Transaction.
Concentration of credit risk and allowance for doubtful accounts. Financial instruments that potentially subject us to concentrations of credit risk consist primarily of accounts receivable. Substantially all of our accounts receivable are with either companies in the health care industry or patients. However, credit risk is limited due to the number of our clients as well as their dispersion across many different geographic regions.
While we have receivables due from federal and state governmental agencies, we do not believe that such receivables represent a credit risk since the related healthcare programs are funded by federal and state governments, and payment is primarily dependent upon submitting appropriate documentation. At December 31, 2018 and 2017, receivable balances (net of contractual adjustments) from Medicare and Medicaid in total were 17% and 16%, respectively, of our consolidated Accounts receivable, net.
The portion of our accounts receivable due from individual patients comprises the largest portion of credit risk. At December 31, 2018 and 2017, receivables due from patients represent approximately 3.1% and 3.2%, respectively, of our consolidated Accounts receivable, net.
We assess the collectability of accounts receivable balances by considering factors such as historical collection experience, customer credit worthiness, the age of accounts receivable balances, regulatory changes and current economic conditions and trends that may affect a customer’s ability to pay. Actual results could differ from those estimates. Our reported net income (loss) is directly affected by our estimate of the collectability of accounts receivable. The allowance for doubtful accounts was $1.8 million and $1.4 million at December 31, 2018 and 2017, respectively.
Income taxes. Income taxes are accounted for under the asset-and-liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and the respective tax bases and for operating loss and tax credit carryforwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in operations in the period that includes the enactment date. We periodically evaluate the realizability of our net deferred tax assets. Our tax accruals are analyzed periodically and adjustments are made as events occur to warrant such adjustment. Valuation allowances on certain U.S. deferred tax assets and non-U.S. deferred tax assets are established, when realization of these tax benefits does not meet the more-likely-than-not threshold.
On December 22, 2017, the 2017 Tax Cuts and Jobs Act (the “Tax Act”) was enacted into law and the new legislation contained several key tax provisions, including a reduction of the corporate income tax rate from 35% to 21% effective January 1, 2018 and a one-time mandatory transition tax on accumulated foreign earnings, among others. We were required to recognize the effect of the tax law changes in the period of enactment, such as remeasuring our U.S. deferred tax assets and liabilities, as well as reassessing the net realizability of our deferred tax assets and liabilities. In December 2017, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act (SAB 118), which allowed us to record provisional amounts during a measurement period not to extend beyond one year of the enactment date. As of December 22, 2018 we completed our analysis in accordance with SAB 118 and recorded immaterial adjustments.
Effective January 1, 2018, the Tax Act provides for a new global intangible low-taxed income (GILTI) provision. Under the GILTI provision, certain foreign subsidiary earnings in excess of an allowable return on the foreign subsidiary’s tangible assets are included in U.S. taxable income. The Company currently estimates GILTI will be immaterial for the year ended December 31, 2018, although interpretive guidance continues to be issued and future guidance may impact this analysis. The Company has not recorded any deferred taxes for future GILTI inclusions as any future inclusions are expected to be offset by net operating loss carryforwards in the U.S.
Equity-based compensation. We measure the cost of employee services received in exchange for an award of equity instruments based on the grant-date fair value of the award. That cost is recognized in the Consolidated Statement of Operations over the period during which an employee is required to provide service in exchange for the award. We record excess tax benefits, realized from the exercise of stock options, as cash flows from operations. Equity-based compensation arrangements to non-employees are recorded at their fair value on the measurement date. The measurement of equity-based compensation to non-employees is subject to periodic adjustment as the underlying equity instruments vest. We estimate the grant-date fair value of our stock option grants using a valuation model known as the Black-Scholes-Merton formula or the “Black-Scholes Model.” The Black-Scholes Model requires the use of several variables to estimate the grant-date fair value of stock options including expected term, expected volatility, expected dividends and risk-free interest rate. We perform analyses to calculate and select the appropriate variable assumptions used in the Black-Scholes Model and to estimate forfeitures of
86
equity-based awards. We are required to adjust our forfeiture estimates on at least an annual basis based on the number of share-based awards that ultimately vest. The selection of assumptions and estimated forfeiture rates is subject to significant judgment and future changes to our assumptions and estimates which may have a material impact on our Consolidated Financial Statements.
Inventories. Inventories are valued at the lower of cost and net realizable value. Cost is determined by the first-in, first-out method. We consider such factors as the amount of inventory on hand, estimated time required to sell such inventories, remaining shelf-life, and current market conditions to determine whether inventories are stated at the lower of cost and net realizable value. Inventories at our diagnostics segment consist primarily of purchased laboratory supplies, which is used in our testing laboratories. Inventory obsolescence for the years ended December 31, 2018 and 2017 was $1.9 million and $5.4 million, respectively.
Pre-launch inventories. We may accumulate commercial quantities of certain product candidates prior to the date we anticipate that such products will receive final U.S. FDA approval. The accumulation of such pre-launch inventories involves the risk that such products may not be approved for marketing by the FDA on a timely basis, or ever. This risk notwithstanding, we may accumulate pre-launch inventories of certain products when such action is appropriate in relation to the commercial value of the product launch opportunity. In accordance with our policy, this pre-launch inventory is expensed.
Contingent consideration. Each period we revalue the contingent consideration obligations associated with certain prior acquisitions to their fair value and record increases in the fair value as contingent consideration expense and decreases in the fair value as a reduction in contingent consideration expense. Changes in contingent consideration result from changes in the assumptions regarding probabilities of successful achievement of related milestones, the estimated timing in which the milestones are achieved and the discount rate used to estimate the fair value of the liability. Contingent consideration may change significantly as our development programs progress, revenue estimates evolve and additional data is obtained, impacting our assumptions. The assumptions used in estimating fair value require significant judgment. The use of different assumptions and judgments could result in a materially different estimate of fair value which may have a material impact on our results from operations and financial position.
87
RECENT ACCOUNTING PRONOUNCEMENTS
Recently adopted accounting pronouncements.
In May 2014, the Financial Accounting Standards Board (the “FASB”) issued Accounting Standards Update (“ASU”) No. 2014-09, “Revenue from Contracts with Customers.” ASU 2014-09, as amended and codified into Topic 606, clarifies the principles for recognizing revenue and develops a common revenue standard for GAAP that removes inconsistencies and weaknesses in revenue requirements, provides a more robust framework for addressing revenue issues, improves comparability of revenue recognition practices across entities, industries, jurisdictions, and capital markets, provides more useful information to users of financial statements through improved disclosure requirements and simplifies the preparation of financial statements by reducing the number of requirements to which an entity must refer. As required, we adopted Topic 606 on January 1, 2018 using the full retrospective approach, and have elected to use the following practical expedients that are permitted under the rules of the adoption, which have been applied consistently to all contracts within all reporting periods presented:
• |
For all reporting periods presented before January 1, 2018, we have not restated revenue from contracts that begin and are completed within the same annual reporting period. |
• |
For all reporting periods presented before January 1, 2018, we have not disclosed the amount of the transaction price allocated to the remaining performance obligations or an explanation of when we expect to recognize that amount as revenue. |
• |
We have applied the practical expedient provided for by Topic 606 by not adjusting the transaction price for significant financing components for periods less than one year. |
As a result of adopting Topic 606 on January 1, 2018 using the full retrospective approach, we revised our comparative financial statements for the prior years as if Topic 606 had been effective for those periods. As a result, the following financial statement line items for 2017 and 2016 were affected:
Consolidated Statement of Operations
|
For the year ended December 31, 2017
(in thousands)
|
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Revenue from services |
$ |
782,710 |
$ |
889,076 |
$ |
(106,366 |
) |
||||
Revenue from transfer of intellectual property and other |
75,537 |
70,668 |
4,869 |
||||||||
Selling, general and administrative |
414,628 |
520,994 |
(106,366 |
) |
|||||||
Research and development |
126,435 |
125,186 |
1,249 |
||||||||
For the year ended December 31, 2016 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Revenue from services |
$ |
928,572 |
$ |
1,012,129 |
$ |
(83,557 |
) |
||||
Revenue from transfer of intellectual property and other |
105,455 |
126,065 |
(20,610 |
) |
|||||||
Selling, general and administrative |
407,331 |
490,888 |
(83,557 |
) |
|||||||
Research and development |
113,871 |
111,205 |
2,666 |
||||||||
88
Consolidated Balance Sheet
|
December 31, 2017
(in thousands)
|
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Other current assets and prepaid expenses |
$ |
42,513 |
$ |
37,113 |
$ |
5,400 |
|||||
Accrued expenses |
225,796 |
215,102 |
10,694 |
||||||||
Other long-term liabilities, principally contract liabilities, contingent consideration and line of credit |
256,415 |
219,954 |
36,461 |
||||||||
Accumulated deficit |
(1,048,914 |
) |
(1,007,159 |
) |
(41,755 |
) |
|||||
Consolidated Statement of Cash Flows
For the year ended December 31, 2017 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Net loss |
$ |
(305,250 |
) |
$ |
(308,870 |
) |
$ |
3,620 |
|||
Other current assets and prepaid expenses |
4,771 |
10,171 |
(5,400 |
) |
|||||||
Contract liabilities |
(58,876 |
) |
(60,656 |
) |
1,780 |
||||||
For the year ended December 31, 2016 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Net loss |
$ |
(48,359 |
) |
$ |
(25,083 |
) |
$ |
(23,276 |
) |
||
Contract liabilities |
(50,893 |
) |
(74,169 |
) |
23,276 |
||||||
The most significant change above relates to amounts in our clinical laboratory operations that were historically classified as provision for bad debts, primarily related to patient responsibility, which are now considered an element of variable consideration as an implicit price concession in determining revenues under Topic 606. Accordingly, we report uncollectible balances associated with individual patients as a reduction of the transaction price and therefore as a reduction in Revenue from services when historically these amounts were classified as provision for bad debts within Selling, general and administrative expenses.
In addition, under Topic 606, the upfront consideration received for a license and contract services combined performance obligation is recognized as revenue to the extent of costs incurred based on the length of the expected performance period and the subjectivity in estimating progress towards satisfaction of the performance obligation. Under previous accounting, we recognized revenue over the expected performance period. The adoption of Topic 606 resulted in a cumulative revenue reduction of $41.8 million and an increase of our accumulated deficit balance as of December 31, 2017; with a corresponding increase in our contract liabilities. For the year ended December 31, 2017, Revenue from the transfer of intellectual property and other was increased by $3.4 million for the change in accounting. For a further discussion of the adoption of Topic 606, refer to Note 14.
In January 2016, the FASB issued ASU No. 2016-01, “Financial Instruments - Overall (Subtopic 825-10),” which addresses certain aspects of recognition, measurement, presentation, and disclosure of financial instruments. The ASU requires equity investments (except those accounted for under the equity method of accounting or those that result in consolidation of the investee) to be measured at fair value with changes in fair value recognized in net income. As a result of the required adoption of ASU 2016-01 on January 1, 2018, we recorded a cumulative-effect adjustment to reclassify our net unrealized gains on our equity securities of $4.9 million as of January 1, 2018 from Accumulated other comprehensive loss to Accumulated deficit in our Consolidated Balance Sheet. Changes in the fair value of certain of our equity securities subsequent to the adoption of ASU 2016-01 on January 1, 2018 will be predominately recognized in net income.
89
In August 2016, the FASB issued ASU No. 2016-15, “Statement of Cash Flows (Topic 230),” which addresses the classification of eight specific cash flow issues with the objective of reducing the existing diversity in practice. The required adoption of ASU 2016-15 in the first quarter of 2018 did not have a significant impact on our Consolidated Financial Statements.
In January 2017, the FASB issued ASU No. 2017-01, “Business Combinations (Topic 805),” which clarifies the definition of a business to assist entities in evaluating whether transactions should be accounted for acquisitions (or disposals) of assets or businesses. The required adoption of ASU 2017-01 in the first quarter of 2018 did not have a significant impact on our Consolidated Financial Statements.
In January 2017, the FASB issued ASU No. 2017-04, “Intangibles - Goodwill and Other (Topic 350),” which simplifies how an entity is required to test for goodwill impairment. Under ASU 2017-04, an impairment loss will reflect the amount by which the carrying amount of a reporting unit exceeds its fair value, not to exceed the carrying amount of goodwill. We early adopted the provisions of ASU 2017-04 prospectively in the fourth quarter of 2018. For a further discussion of the adoption of ASU 2017-04, refer to Note 2, Goodwill and intangible assets.
Pending accounting pronouncements.
In February 2016, the FASB issued ASU No. 2016-02, “Leases (Topic 842),” which will require organizations that lease assets with lease terms of more than 12 months to recognize assets and liabilities for the rights and obligations created by those leases on their balance sheets. ASU 2016-02, as amended, requires new qualitative and quantitative disclosures to help investors and other financial statement users better understand the amount, timing, and uncertainty of cash flows arising from leases. ASU 2016-02 will be effective for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years, with early adoption permitted. We have begun a process to identify a complete population of our leases. Such process includes reviewing various contracts to identify whether such arrangements convey the right to control the use of an identified asset. The determination of the impact of this new guidance is ongoing and, as such, we are not able to reasonably estimate the effect the adoption of this new standard will have on our financial statements. Based on our preliminary assessment of this ASU, we believe the new standard will have a significant impact on our Consolidated Balance Sheet, which has not yet been quantified. In July 2018, the FASB issued an ASU to provide an additional transition method to adopt the guidance by allowing entities to initially apply the new leases standard at the adoption date and recognize a cumulative effect to the opening balance of retained earnings. We are currently evaluating the choice of transition options.
In June 2018, the FASB issued ASU No. 2018-07, “Compensation - Stock Compensation (Topic 718),” which expands the scope of Topic 718 to include share-based payment transactions for acquiring goods and services from nonemployees. ASU 2018-07 will be effective for public business entities for fiscal years beginning after December 15, 2018, including interim periods within that fiscal year, with early adoption permitted. We are currently evaluating the impact of this new guidance on our Consolidated Financial Statements.
90
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
In the normal course of doing business, we are exposed to the risks associated with foreign currency exchange rates and changes in interest rates.
Foreign Currency Exchange Rate Risk – We operate globally and, as such, we are subject to foreign exchange risk in our commercial operations as portions of our revenues are exposed to changes in foreign currency exchange rates, primarily the Chilean Peso, the Mexican Peso, the Euro and the New Israeli Shekel.
Although we do not speculate in the foreign exchange market, we may from time to time manage exposures that arise in the normal course of business related to fluctuations in foreign currency exchange rates by entering into offsetting positions through the use of foreign exchange forward contracts. Certain firmly committed transactions may be hedged with foreign exchange forward contracts. As exchange rates change, gains and losses on the exposed transactions are partially offset by gains and losses related to the hedging contracts. Both the exposed transactions and the hedging contracts are translated and fair valued, respectively, at current spot rates, with gains and losses included in earnings.
Our derivative activities, which consist of foreign exchange forward contracts, are initiated to economically hedge forecasted cash flows that are exposed to foreign currency risk. The foreign exchange forward contracts generally require us to exchange local currencies for foreign currencies based on pre-established exchange rates at the contracts’ maturity dates. As exchange rates change, gains and losses on these contracts are generated based on the change in the exchange rates that are recognized in the Consolidated Statements of Operations and offset the impact of the change in exchange rates on the foreign currency cash flows that are hedged. If the counterparties to the exchange contracts do not fulfill their obligations to deliver the contracted currencies, we could be at risk for currency related fluctuations. Our foreign exchange forward contracts primarily hedge exchange rates on the Chilean Peso to the U.S. dollar. If Chilean Pesos were to strengthen or weaken in relation to the U.S. dollar, our loss or gain on hedged foreign currency cash-flows would be offset by the derivative contracts, with a net effect of zero.
We do not engage in trading market risk sensitive instruments or purchasing hedging instruments or “other than trading” instruments that are likely to expose us to significant market risk, whether interest rate, foreign currency exchange, commodity price, or equity price risk.
Interest Rate Risk – Our exposure to interest rate risk relates to our cash and investments and to our borrowings. We generally maintain an investment portfolio of money market funds and marketable securities. The securities in our investment portfolio are not leveraged, and are, due to their very short-term nature, subject to minimal interest rate risk. We currently do not hedge interest rate exposure. Because of the short-term maturities of our investments, we do not believe that a change in market interest rates would have a significant negative impact on the value of our investment portfolio except for reduced income in a low interest rate environment.
At December 31, 2018, we had cash and cash equivalents of $96.5 million. The weighted average interest rate related to our cash and cash equivalents for the year ended December 31, 2018 was less than 1%. As of December 31, 2018, the principal outstanding balance under our Credit Agreement with JPMorgan Chase Bank, N.A. and our Chilean and Spanish credit lines was $108.7 million in the aggregate at a weighted average interest rate of approximately 4.7%.
Our $31.9 million aggregate principal amount of our 2033 Senior Notes has a fixed interest rate of 3.0% and our $55.0 million aggregate principal amount of our 2023 Convertible Notes has a fixed interest rate of 5%, and therefore are not subject to fluctuations in market interest rates.
The primary objective of our investment activities is to preserve principal while at the same time maximizing yields without significantly increasing risk. To achieve this objective, we may invest our excess cash in debt instruments of the U.S. Government and its agencies, bank obligations, repurchase agreements and high-quality corporate issuers, and money market funds that invest in such debt instruments, and, by policy, restrict our exposure to any single corporate issuer by imposing concentration limits. To minimize the exposure due to adverse shifts in interest rates, we maintain investments at an average maturity of generally less than three months.
91
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
Page |
|
92
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of OPKO Health, Inc. and subsidiaries
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of OPKO Health, Inc. and subsidiaries (the Company) as of December 31, 2018 and 2017, the related consolidated statements of operations, comprehensive loss, equity and cash flows for each of the three years in the period ended December 31, 2018, and the related notes and financial statements schedule included at Item 15(a)(1) (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2018 and 2017, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2018, in conformity with U.S generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2018, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 Framework) and our report dated March 1, 2019 expressed an unqualified opinion thereon.
Adoption of ASU No. 2014-09
As discussed in Note 2 to the consolidated financial statements, the Company changed its method for recognizing revenue in the accompanying consolidated financial statements for each of the three years in the period ended December 31, 2018 due to the adoption of Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (Topic 606), as amended.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test bases, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2007.
Miami, Florida
March 1, 2019
93
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of OPKO Health, Inc. and subsidiaries
Opinion on Internal Control over Financial Reporting
We have audited OPKO Health, Inc. and subsidiaries’ internal control over financial reporting as of December 31, 2018, based on criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, OPKO Health, Inc. and subsidiaries (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2018, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the 2018 consolidated financial statements of the Company and our report dated March 1, 2019 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Annual Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
Miami, Florida
March 1, 2019
94
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share data)
December 31, |
|||||||
2018 |
2017 |
||||||
ASSETS |
|||||||
Current assets: |
|||||||
Cash and cash equivalents |
$ |
$ |
|||||
Accounts receivable, net |
|||||||
Inventory, net |
|||||||
Other current assets and prepaid expenses |
|||||||
Total current assets |
|||||||
Property, plant and equipment, net |
|||||||
Intangible assets, net |
|||||||
In-process research and development |
|||||||
Goodwill |
|||||||
Investments |
|||||||
Other assets |
|||||||
Total assets |
$ |
$ |
|||||
LIABILITIES AND EQUITY |
|||||||
Current liabilities: |
|||||||
Accounts payable |
$ |
$ |
|||||
Accrued expenses |
|||||||
Current portion of 2033 Senior Notes |
|||||||
Current portion of lines of credit and notes payable |
|||||||
Total current liabilities |
|||||||
2023 Convertible Notes and 2033 Senior Notes |
|||||||
Deferred tax liabilities, net |
|||||||
Other long-term liabilities, principally contract liabilities, contingent consideration and lines of credit |
|||||||
Total long-term liabilities |
|||||||
Total liabilities |
|||||||
Equity: |
|||||||
Common Stock - $0.01 par value, 750,000,000 shares authorized; 586,881,720 and 560,023,745 shares issued at December 31, 2018 and 2017, respectively |
|||||||
Treasury Stock, at cost - 549,907 shares at December 31, 2018 and 2017, respectively |
( |
) |
( |
) |
|||
Additional paid-in capital |
|||||||
Accumulated other comprehensive income (loss) |
( |
) |
( |
) |
|||
Accumulated deficit |
( |
) |
( |
) |
|||
Total shareholders’ equity |
|||||||
Total liabilities and equity |
$ |
$ |
|||||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
95
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF OPERATIONS
(In thousands, except share and per share data)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Revenues: |
|||||||||||
Revenue from services |
$ |
$ |
$ |
||||||||
Revenue from products |
|||||||||||
Revenue from transfer of intellectual property and other |
|||||||||||
Total revenues |
|||||||||||
Costs and expenses: |
|||||||||||
Cost of service revenue |
|||||||||||
Cost of product revenue |
|||||||||||
Selling, general and administrative |
|||||||||||
Research and development |
|||||||||||
Contingent consideration |
( |
) |
( |
) |
|||||||
Amortization of intangible assets |
|||||||||||
Asset impairment charges |
|||||||||||
Total costs and expenses |
|||||||||||
Operating loss |
( |
) |
( |
) |
( |
) |
|||||
Other income and (expense), net: |
|||||||||||
Interest income |
|||||||||||
Interest expense |
( |
) |
( |
) |
( |
) |
|||||
Fair value changes of derivative instruments, net |
|||||||||||
Other income (expense), net |
|||||||||||
Other income and (expense), net |
( |
) |
( |
) |
|||||||
Loss before income taxes and investment losses |
( |
) |
( |
) |
( |
) |
|||||
Income tax benefit (provision) |
( |
) |
|||||||||
Net loss before investment losses |
( |
) |
( |
) |
( |
) |
|||||
Loss from investments in investees |
( |
) |
( |
) |
( |
) |
|||||
Net loss |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Loss per share: |
|||||||||||
Loss per share, basic |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Loss per share, diluted |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
|
Weighted average number of common shares
outstanding, basic
|
|||||||||||
Weighted average number of common shares outstanding, diluted |
|||||||||||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
96
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(In thousands)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Net loss |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Other comprehensive income (loss), net of tax: |
|||||||||||
Change in foreign currency translation and other comprehensive income (loss) |
( |
) |
( |
) |
|||||||
Investments: |
|||||||||||
Change in unrealized gain (loss), net of tax |
( |
) |
|||||||||
Reclassification adjustments due to adoption of ASU 2016-01 |
( |
) |
|||||||||
Reclassification adjustments for losses included in net loss, net of tax |
( |
) |
|||||||||
Comprehensive loss |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
97
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF EQUITY
(In thousands, except share and per share data)
For the years ended December 31, 2018, 2017, 2016 (continued)
Common Stock |
Treasury |
Additional
Paid-In
Capital
|
Accumulated Other
Comprehensive
Loss
|
Accumulated
Deficit
|
Total |
||||||||||||||||||||||||
Shares |
Dollars |
Shares |
Dollars |
||||||||||||||||||||||||||
Balance at December 31, 2015 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
Equity-based compensation expense |
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Exercise of Common Stock options and warrants |
— |
— |
— |
— |
|||||||||||||||||||||||||
Issuance of Common Stock upon exchange of 2033 Senior Notes |
— |
— |
— |
— |
|||||||||||||||||||||||||
Issuance of Treasury Stock in connection with OPKO Health Europe’s Contingent Consideration |
— |
— |
— |
— |
|||||||||||||||||||||||||
Issuance of Treasury Stock for investment in Xenetic |
— |
— |
— |
— |
|||||||||||||||||||||||||
Issuance of Common Stock for OPKO Renal earnout |
— |
— |
— |
— |
|||||||||||||||||||||||||
Issuance of Common Stock for Transition Therapeutics purchase |
— |
— |
— |
— |
|||||||||||||||||||||||||
Net loss |
— |
— |
— |
— |
— |
— |
( |
) |
( |
) |
|||||||||||||||||||
Other comprehensive loss |
— |
— |
— |
— |
— |
( |
) |
— |
( |
) |
|||||||||||||||||||
Balance at December 31, 2016 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
98
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF EQUITY
(In thousands, except share and per share data)
For the years ended December 31, 2018, 2017, 2016 (continued)
Common Stock |
Treasury |
Additional
Paid-In
Capital
|
Accumulated Other
Comprehensive
Loss
|
Accumulated
Deficit
|
Total |
||||||||||||||||||||||||
Shares |
Dollars |
Shares |
Dollars |
||||||||||||||||||||||||||
Balance at December 31, 2016 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
Equity-based compensation expense |
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Exercise of Common Stock options and warrants |
— |
— |
— |
— |
|||||||||||||||||||||||||
|
Reclassification of embedded
derivatives to equity
|
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Issuance of Treasury Stock in connection with OPKO Health Europe’s Contingent Consideration |
— |
— |
— |
— |
|||||||||||||||||||||||||
Adoption of ASU 2016-09 |
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Net loss |
— |
— |
— |
— |
— |
— |
( |
) |
( |
) |
|||||||||||||||||||
Other comprehensive loss |
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Balance at December 31, 2017 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
OPKO Health, Inc. and Subsidiaries
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
99
CONSOLIDATED STATEMENTS OF EQUITY
(In thousands, except share and per share data)
For the years ended December 31, 2018, 2017, 2016 (continued)
Common Stock |
Treasury |
Additional Paid-In Capital |
Accumulated Other Comprehensive Loss |
Accumulated Deficit |
Total |
||||||||||||||||||||||||
Shares |
Dollars |
Shares |
Dollars |
||||||||||||||||||||||||||
Balance at December 31, 2017 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
Equity-based compensation expense |
— |
— |
— |
— |
— |
— |
|||||||||||||||||||||||
Exercise of Common Stock options and warrants |
— |
— |
— |
— |
|||||||||||||||||||||||||
Adoption of ASU 2016-01 |
— |
— |
— |
— |
( |
) |
|||||||||||||||||||||||
Private placement |
— |
— |
— |
— |
|||||||||||||||||||||||||
Net loss |
— |
— |
— |
— |
— |
— |
( |
) |
( |
) |
|||||||||||||||||||
Other comprehensive loss |
— |
— |
— |
— |
— |
( |
) |
— |
( |
) |
|||||||||||||||||||
Balance at December 31, 2018 |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||||||||||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
100
OPKO Health, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Cash flows from operating activities: |
|||||||||||
Net loss |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Adjustments to reconcile net loss to net cash (used in) provided by operating activities: |
|||||||||||
Depreciation and amortization |
|||||||||||
Non-cash interest |
|||||||||||
Amortization of deferred financing costs |
|||||||||||
Losses from investments in investees |
|||||||||||
Equity-based compensation – employees and non-employees |
|||||||||||
Impairment of assets |
|||||||||||
Realized loss (gain) on disposal of fixed assets and sales of equity securities |
( |
) |
|||||||||
Loss (gain) on conversion of 3.00% convertible senior notes |
|||||||||||
Change in fair value of equity securities and derivative instruments |
( |
) |
( |
) |
( |
) |
|||||
Change in fair value of contingent consideration |
( |
) |
( |
) |
|||||||
Deferred income tax provision (benefit) |
( |
) |
( |
) |
|||||||
Changes in assets and liabilities, net of the effects of acquisitions: |
|||||||||||
Accounts receivable, net |
( |
) |
|||||||||
Inventory, net |
( |
) |
( |
) |
|||||||
Other current assets and prepaid expenses |
|||||||||||
Other assets |
( |
) |
( |
) |
( |
) |
|||||
Accounts payable |
( |
) |
( |
) |
|||||||
Foreign currency measurement |
( |
) |
( |
) |
|||||||
Contract liabilities |
( |
) |
( |
) |
( |
) |
|||||
Accrued expenses and other liabilities |
|||||||||||
Net cash (used in) provided by operating activities |
( |
) |
( |
) |
|||||||
Cash flows from investing activities: |
|||||||||||
Investments in investees |
( |
) |
( |
) |
( |
) |
|||||
Proceeds from sale of equity securities |
|||||||||||
Acquisition of businesses, net of cash acquired |
|||||||||||
Acquisition of intangible assets |
( |
) |
|||||||||
Purchase of marketable securities |
( |
) |
|||||||||
Maturities of short-term marketable securities |
|||||||||||
Proceeds from the sale of property, plant and equipment |
|||||||||||
Capital expenditures |
( |
) |
( |
) |
( |
) |
|||||
Net cash used in investing activities |
( |
) |
( |
) |
( |
) |
|||||
Cash flows from financing activities: |
|||||||||||
Issuance of common stock |
|||||||||||
Issuance of 2023 Convertible Notes, including to related parties |
|||||||||||
Proceeds from the exercise of Common Stock options and warrants |
|||||||||||
Borrowings on lines of credit |
|||||||||||
Repayments of lines of credit |
( |
) |
( |
) |
( |
) |
|||||
Net cash provided by (used in) financing activities |
( |
) |
|||||||||
Effect of exchange rate changes on cash and cash equivalents |
( |
) |
( |
) |
|||||||
Net increase (decrease) in cash and cash equivalents |
( |
) |
( |
) |
|||||||
Cash and cash equivalents at beginning of period |
|||||||||||
Cash and cash equivalents at end of period |
$ |
$ |
$ |
||||||||
SUPPLEMENTAL INFORMATION: |
|||||||||||
Interest paid |
$ |
$ |
$ |
||||||||
Income taxes paid, net of refunds |
$ |
( |
) |
$ |
$ |
( |
) |
||||
Non-cash financing: |
|||||||||||
Shares issued upon the conversion of: |
|||||||||||
2033 Senior Notes |
$ |
$ |
$ |
||||||||
Common Stock options and warrants, surrendered in net exercise |
$ |
$ |
$ |
||||||||
Issuance of capital stock to acquire or contingent consideration settlement: |
|||||||||||
Transition Therapeutics, Inc. |
$ |
$ |
$ |
||||||||
OPKO Renal |
$ |
$ |
$ |
||||||||
OPKO Health Europe |
$ |
$ |
$ |
||||||||
Issuance of stock for investment in Xenetic |
$ |
$ |
$ |
||||||||
The accompanying Notes to Consolidated Financial Statements are an integral part of these statements.
101
OPKO Health, Inc. and Subsidiaries
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1 Business and Organization
We are a diversified healthcare company that seeks to establish industry-leading positions in large and rapidly growing medical markets. Our diagnostics business includes BioReference Laboratories, Inc. (“BioReference”), the nation’s third-largest clinical laboratory with a core genetic testing business and an almost 300 -person sales and marketing team to drive growth and leverage new products, including the 4Kscore prostate cancer test. Our pharmaceutical business features Rayaldee, an FDA-approved treatment for secondary hyperparathyroidism (“SHPT”) in adults with stage 3 or 4 chronic kidney disease (“CKD”) and vitamin D insufficiency (launched in November 2016), OPK88004, a selective androgen receptor modulator which has been studied for benign prostatic hyperplasia, but for which we are exploring other indications, and OPK88003, a once or twice weekly oxyntomodulin for type 2 diabetes and obesity which is a clinically advanced drug candidate among the new class of GLP-1 glucagon receptor dual agonists (phase 2b). Our pharmaceutical business also features hGH-CTP, a once-weekly human growth hormone injection (in phase 3 and partnered with Pfizer). We are incorporated in Delaware and our principal executive offices are located in leased offices in Miami, Florida.
Through BioReference, we provide laboratory testing services, primarily to customers in the larger metropolitan areas across New York, New Jersey, Maryland, Pennsylvania, Delaware, Washington, DC, Florida, California, Texas, Illinois and Massachusetts as well as to customers in a number of other states. We offer a comprehensive test menu of clinical diagnostics for blood, urine, and tissue analysis. This includes hematology, clinical chemistry, immunoassay, infectious diseases, serology, hormones, and toxicology assays, as well as Pap smear, anatomic pathology (biopsies) and other types of tissue analysis. We market our laboratory testing services directly to physicians, geneticists, hospitals, clinics, correctional and other health facilities.
We operate established pharmaceutical platforms in Ireland, Chile, Spain, and Mexico, which are generating revenue and which we expect to facilitate future market entry for our products currently in development. In addition, we have a development and commercial supply pharmaceutical company and a global supply chain operation and holding company in Ireland. We own a specialty active pharmaceutical ingredients (“APIs”) manufacturer in Israel, which we expect will facilitate the development of our pipeline of molecules and compounds for our molecular diagnostic and therapeutic products.
102
Note 2 Summary of Significant Accounting Policies
Pre-launch inventories. We may accumulate commercial quantities of certain product candidates prior to the date we anticipate that such products will receive final U.S. FDA approval. The accumulation of such pre-launch inventories involves the risk that such products may not be approved for marketing by the FDA on a timely basis, or ever. This risk notwithstanding, we may accumulate pre-launch inventories of certain products when such action is appropriate in relation to the commercial value of the product launch opportunity. In accordance with our policy, this pre-launch inventory is expensed.
Assets acquired and liabilities assumed in business combinations, licensing and other transactions are generally recognized at the date of acquisition at their respective fair values. We determined the fair value of intangible assets, including IPR&D, using the “income method.”
Goodwill is tested at least annually for impairment, or when events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, by assessing qualitative factors or performing a quantitative analysis in determining whether it is more likely than not that its fair value exceeds the carrying value. Due to the loss of a significant customer in 2018, we recorded a goodwill impairment charge of $11.7 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2018 to write the carrying amount of the FineTech reporting unit down to its estimated fair value due to the loss of a significant customer in 2018. No goodwill impairment was recorded for the years ended December 31, 2017 and 2016 as a result of our testing.
Intangible assets are tested for impairment whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable, although IPR&D is required to be tested at least annually until the project is completed or abandoned. Upon obtaining regulatory approval, the IPR&D asset is then accounted for as a finite-lived intangible asset and amortized on a straight-line basis over its estimated useful life. If the project is abandoned, the IPR&D asset is charged to expense.
We recorded an impairment charge of $10.1 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2018 to write our IPR&D assets for Alpharen and OPK88004 down to their estimated fair value as a result of our testing. We recorded an impairment charge of $13.2 million in Asset impairment charges in our Consolidated Statement of Operations for the year ended December 31, 2017 to write our intangible asset for VARUBI™ down to its estimated fair value.
We amortize intangible assets with definite lives on a straight-line basis over their estimated useful lives, ranging from 3 to 20 years. We use the straight-line method of amortization as there is no reliably determinable pattern in which the economic
103
net deferred tax assets. Our tax accruals are analyzed periodically and adjustments are made as events occur to warrant such
adjustment. Valuation allowances on certain U.S. deferred tax assets and non-U.S. deferred tax assets are established, because
realization of these tax benefits through future taxable income does not meet the more-likely-than-not threshold.
104
On December 22, 2017, the 2017 Tax Cuts and Jobs Act (the “Tax Act”) was enacted into law and the new legislation contains several key tax provisions, including a reduction of the corporate income tax rate from 35% to 21% effective January 1, 2018 and a one-time mandatory transition tax on accumulated foreign earnings, among others. We were required to recognize the effect of the tax law changes in the period of enactment, such as remeasuring our U.S. deferred tax assets and liabilities, as well as reassessing the net realizability of our deferred tax assets and liabilities. In December 2017, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act (SAB 118), which allowed us to record provisional amounts during a measurement period not to extend beyond one year of the enactment date. As of December 22, 2018 we completed our analysis in accordance with SAB 118 and recorded immaterial adjustments.
Effective January 1, 2018, the Tax Act provides for a new global intangible low-taxed income (GILTI) provision. Under the GILTI provision, certain foreign subsidiary earnings in excess of an allowable return on the foreign subsidiary’s tangible assets are included in U.S. taxable income. The Company currently estimates GILTI will be immaterial for the year ended December 31, 2018, although interpretive guidance continues to be issued and future guidance may impact this analysis. The Company has not recorded any deferred taxes for future GILTI inclusions as any future inclusions are expected to be treated as a period expense and offset by net operating loss carryforwards in the U.S.
We operate in various countries and tax jurisdictions globally. For the year ended December 31, 2018, the tax rate differed from the U.S. federal statutory rate of 21% primarily due to the valuation allowance against certain U.S. and non-U.S. deferred tax assets, the relative mix in earnings and losses in the U.S. versus foreign tax jurisdictions, and the impact of certain discrete tax events and operating results in tax jurisdictions which do not result in a tax benefit.
While we have receivables due from federal and state governmental agencies, we do not believe that such receivables represent a credit risk since the related healthcare programs are funded by federal and state governments, and payment is primarily dependent upon submitting appropriate documentation. At December 31, 2018 and 2017, receivable balances (net of contractual adjustments) from Medicare and Medicaid in total were 17 % and 16 %, respectively, of our consolidated Accounts receivable, net.
The portion of our accounts receivable due from individual patients comprises the largest portion of credit risk. At December 31, 2018 and 2017, receivables due from patients represent approximately 3.1 % and 3.2 %, respectively, of our consolidated Accounts receivable, net.
We assess the collectability of accounts receivable balances by considering factors such as historical collection experience, customer credit worthiness, the age of accounts receivable balances, regulatory changes and current economic conditions and trends that may affect a customer’s ability to pay. Actual results could differ from those estimates. Our reported
105
106
Recently adopted accounting pronouncements.
In May 2014, the Financial Accounting Standards Board (the “FASB”) issued Accounting Standards Update (“ASU”) No. 2014-09, “Revenue from Contracts with Customers.” ASU 2014-09, as amended and codified into Topic 606, clarifies the principles for recognizing revenue and develops a common revenue standard for GAAP that removes inconsistencies and weaknesses in revenue requirements, provides a more robust framework for addressing revenue issues, improves comparability of revenue recognition practices across entities, industries, jurisdictions, and capital markets, provides more useful information to users of financial statements through improved disclosure requirements and simplifies the preparation of financial statements by reducing the number of requirements to which an entity must refer. As required, we adopted Topic 606 on January 1, 2018 using the full retrospective approach, and have elected to use the following practical expedients that are permitted under the rules of the adoption, which have been applied consistently to all contracts within all reporting periods presented:
• |
For all reporting periods presented before January 1, 2018, we have not restated revenue from contracts that begin and are completed within the same annual reporting period. |
• |
For all reporting periods presented before January 1, 2018, we have not disclosed the amount of the transaction price allocated to the remaining performance obligations or an explanation of when we expect to recognize that amount as revenue. |
• |
We have applied the practical expedient provided for by Topic 606 by not adjusting the transaction price for significant financing components for periods less than one year. |
As a result of adopting Topic 606 on January 1, 2018 using the full retrospective approach, we revised our comparative financial statements for the prior years as if Topic 606 had been effective for those periods. As a result, the following financial statement line items for 2017 and 2016 were affected:
Consolidated Statement of Operations
|
For the year ended December 31, 2017
(in thousands)
|
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Revenue from services |
$ |
$ |
$ |
( |
) |
||||||
Revenue from transfer of intellectual property and other |
|||||||||||
Selling, general and administrative |
( |
) |
|||||||||
Research and development |
|||||||||||
For the year ended December 31, 2016 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Revenue from services |
$ |
$ |
$ |
( |
) |
||||||
Revenue from transfer of intellectual property and other |
( |
) |
|||||||||
Selling, general and administrative |
( |
) |
|||||||||
Research and development |
|||||||||||
107
Consolidated Balance Sheet
|
December 31, 2017
(in thousands)
|
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Other current assets and prepaid expenses |
$ |
$ |
$ |
||||||||
Accrued expenses |
|||||||||||
Other long-term liabilities, principally contract liabilities, contingent consideration and line of credit |
|||||||||||
Accumulated deficit |
( |
) |
( |
) |
( |
) |
|||||
Consolidated Statement of Cash Flows
For the year ended December 31, 2017 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Net loss |
$ |
( |
) |
$ |
( |
) |
$ |
||||
Other current assets and prepaid expenses |
( |
) |
|||||||||
Contract liabilities |
( |
) |
( |
) |
|||||||
For the year ended December 31, 2016 (in thousands) |
|||||||||||
As adjusted under Topic 606 |
As originally reported |
Effect of change |
|||||||||
Net loss |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Contract liabilities |
( |
) |
( |
) |
|||||||
The most significant change above relates to amounts in our clinical laboratory operations that were historically classified as provision for bad debts, primarily related to patient responsibility, which are now considered an element of variable consideration as an implicit price concession in determining revenues under Topic 606. Accordingly, we report uncollectible balances associated with individual patients as a reduction of the transaction price and therefore as a reduction in Revenue from services when historically these amounts were classified as provision for bad debts within Selling, general and administrative expenses.
In addition, under Topic 606, the upfront consideration received for a license and contract services combined performance obligation is recognized as revenue to the extent of costs incurred based on the length of the expected performance period and the subjectivity in estimating progress towards satisfaction of the performance obligation. Under previous accounting, we recognized revenue over the expected performance period. The adoption of Topic 606 resulted in a cumulative revenue reduction of $41.8 million and an increase of our accumulated deficit balance as of December 31, 2017; with a corresponding increase in our contract liabilities. For the years ended December 31, 2017 and 2016, Revenue from the transfer of intellectual property and other was increased (decreased) by $3.4 million and $(23.3 ) million, respectively, for the change in accounting. For a further discussion of the adoption of Topic 606, refer to Note 14.
In January 2016, the FASB issued ASU No. 2016-01, “Financial Instruments - Overall (Subtopic 825-10),” which addresses certain aspects of recognition, measurement, presentation, and disclosure of financial instruments. The ASU requires equity investments (except those accounted for under the equity method of accounting or those that result in consolidation of the investee) to be measured at fair value with changes in fair value recognized in net income. As a result of the required adoption of ASU 2016-01 on January 1, 2018, we recorded a cumulative-effect adjustment to reclassify our net unrealized gains on our equity securities of $4.9 million as of January 1, 2018 from Accumulated other comprehensive loss to Accumulated deficit in our Consolidated Balance Sheet. Changes in the fair value of certain of our equity securities subsequent to the adoption of ASU 2016-01 on January 1, 2018 will be predominately recognized in net income.
108
In August 2016, the FASB issued ASU No. 2016-15, “Statement of Cash Flows (Topic 230),” which addresses the classification of eight specific cash flow issues with the objective of reducing the existing diversity in practice. The required adoption of ASU 2016-15 in the first quarter of 2018 did not have a significant impact on our Consolidated Financial Statements.
In January 2017, the FASB issued ASU No. 2017-01, “Business Combinations (Topic 805),” which clarifies the definition of a business to assist entities in evaluating whether transactions should be accounted for acquisitions (or disposals) of assets or businesses. The required adoption of ASU 2017-01 in the first quarter of 2018 did not have a significant impact on our Consolidated Financial Statements.
In January 2017, the FASB issued ASU No. 2017-04, “Intangibles - Goodwill and Other (Topic 350),” which simplifies how an entity is required to test for goodwill impairment. Under ASU 2017-04, an impairment loss will reflect the amount by which the carrying amount of a reporting unit exceeds its fair value, not to exceed the carrying amount of goodwill. We early adopted the provisions of ASU 2017-04 prospectively in the fourth quarter of 2018. For a further discussion of the adoption of ASU 2017-04, refer to Goodwill and intangible assets earlier in this Note 2.
Pending accounting pronouncements.
In February 2016, the FASB issued ASU No. 2016-02, “Leases (Topic 842),” which will require organizations that lease assets with lease terms of more than 12 months to recognize assets and liabilities for the rights and obligations created by those leases on their balance sheets. ASU 2016-02, as amended, requires new qualitative and quantitative disclosures to help investors and other financial statement users better understand the amount, timing, and uncertainty of cash flows arising from leases. ASU 2016-02 will be effective for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years, with early adoption permitted. We have begun a process to identify a complete population of our leases. Such process includes reviewing various contracts to identify whether such arrangements convey the right to control the use of an identified asset. The determination of the impact of this new guidance is ongoing and, as such, we are not able to reasonably estimate the effect the adoption of this new standard will have on our financial statements. Based on our preliminary assessment of this ASU, we believe the new standard will have a significant impact on our Consolidated Balance Sheet, which has not yet been quantified. In July 2018, the FASB issued an ASU to provide an additional transition method to adopt the guidance by allowing entities to initially apply the new leases standard at the adoption date and recognize a cumulative effect to the opening balance of retained earnings. We are currently evaluating the choice of transition options.
109
Note 3 Loss Per Share
Basic loss per share is computed by dividing our net loss by the weighted average number of shares outstanding during the period. For diluted earnings per share, the dilutive impact of stock options and warrants is determined by applying the “treasury stock” method. The dilutive impact of the 2033 Senior Notes and 2023 Convertible Notes (each, as defined herein and as discussed in Note 6) has been considered using the “if converted” method. In the periods in which their effect would be antidilutive, no effect has been given to outstanding options, warrants or the potentially dilutive shares issuable pursuant to the 2033 Senior Notes and 2023 Convertible Notes in the dilutive computation. The following table sets forth the computation of basic and diluted earnings (loss) per share:
For the years ended December 31, |
|||||||||||
(In thousands, except per share data) |
2018 |
2017 |
2016 |
||||||||
Numerator |
|||||||||||
Net loss, basic |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Add: Interest on 2033 Senior Notes |
|||||||||||
Change in FV of embedded derivative income |
( |
) |
|||||||||
Net loss, diluted |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Denominator |
|||||||||||
(Shares in thousands) |
|||||||||||
Weighted average common shares outstanding, basic |
|||||||||||
Effect of dilutive securities: |
|||||||||||
2033 Senior Notes |
|||||||||||
Dilutive potential shares |
|||||||||||
Weighted average common shares outstanding, diluted |
|||||||||||
Loss per share, basic |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Loss per share, diluted |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
A total of 16,568,520 , 6,255,624 and 4,736,104 potential shares of Common Stock have been excluded from the calculation of diluted net loss per share for the years ended December 31, 2018, 2017 and 2016, respectively, because their inclusion would be antidilutive.
During the year ended December 31, 2018, 540,000 Common Stock options and Common Stock warrants to purchase shares of our Common Stock were exercised, resulting in the issuance of 353,677 shares of Common Stock. Of the 540,000 Common Stock options and Common Stock warrants exercised, 186,323 shares of Common Stock were surrendered in lieu of a cash payment via the net exercise feature of the agreements.
During the year ended December 31, 2017, 1,720,649 Common Stock options and Common Stock warrants to purchase shares of our Common Stock were exercised, resulting in the issuance of 1,447,792 shares of Common Stock. Of the 1,720,649 Common Stock options and Common Stock warrants exercised, 272,857 shares of Common Stock were surrendered in lieu of a cash payment via the net exercise feature of the agreements.
110
Note 4 Acquisitions, Investments and Licenses
Investments
(in thousands) |
||||||||
Investment type |
Investment Carrying Value |
Underlying Equity in Net Assets |
||||||
Equity method investments |
$ |
$ |
||||||
Variable interest entity, equity method |
||||||||
Equity securities |
||||||||
Equity securities with no readily determinable fair value |
||||||||
Warrants and options |
||||||||
Total carrying value of investments |
$ |
|||||||
Equity method investments
Our equity method investments consist of investments in Pharmsynthez (ownership 9 %), Cocrystal Pharma, Inc. (“COCP”) (9 %), Non-Invasive Monitoring Systems, Inc. (“NIMS”) (1 %), Neovasc Inc. (“Neovasc”)(4 %), InCellDx, Inc. (29 %), BioCardia, Inc. (“BioCardia”) (5 %), and Xenetic Biosciences, Inc. (“Xenetic”) (4 %). The total assets, liabilities, and net losses of our equity method investees as of and for the year ended December 31, 2018 were $234.3 million, $85.9 million, and $179.8 million, respectively. We have determined that we and/or our related parties can significantly influence the success of our equity method investments through our board representation and/or voting power. Accordingly, we account for our investment in these entities under the equity method and record our proportionate share of their losses in Loss from investments in investees in our Consolidated Statement of Operations. Included in Loss from investments in investees for the year ended December 31, 2018 is a charge of $2.9 million to write our investment in InCellDx, Inc. down to its fair value as of December 31, 2018. The aggregate value of our equity method investments based on the quoted market price of their common stock and the number of shares held by us as of December 31, 2018 is $15.3 million.
Equity securities
Our equity securities consist of investments in Phio Pharmaceuticals (“Phio”), previously Rxi Pharmaceuticals, (ownership 0 %), VBI Vaccines Inc. (“VBI”) (7 %), ChromaDex Corporation (0 %), MabVax Therapeutics Holdings, Inc. (“MabVax”) (2 %) and Eloxx Pharmaceuticals, Inc. (“Eloxx”)(4 %). We have determined that our ownership, along with that of our related parties, does not provide us with significant influence over the operations of these investments. Accordingly, we account for our investment in these entities as equity securities, and we record changes in the fair value of these investments in Other income (expense) each reporting period when they have readily determinable fair value. Equity securities without a readily determinable fair value are adjusted to fair value when an observable price change can be identified. Net gains and losses on our equity securities for the year ended December 31, 2018 are as follows:
(in thousands) |
||||
Equity Securities |
For the twelve months ended December 31, 2018 |
|||
Net gains and losses recognized during the period on equity securities |
$ |
|||
Less: Net gains and losses recognized during the period on equity securities sold during the period |
||||
Unrealized net gains recognized during the reporting period on equity securities still held at the reporting date |
$ |
|||
Sales of investments
Gains (losses) included in earnings from sales of our investments are recorded in Other income (expense), net in our Consolidated Statement of Operations. Gains (losses) from sale of our investments for the years ended December 31, 2018 and 2017, was $25.8 thousand and $1.5 million, respectively. No gains (losses) were recognized during the years ended December 31, 2016. The cost of securities sold is based on the specific identification method.
111
Warrants and options
In addition to our equity method investments and equity securities, we hold options to purchase 0.4 million additional shares of BioCardia, 0.2 million of which are vested as of December 31, 2018, and 33 thousand, 0.7 million, 0.5 million, 22 thousand and 29 thousand of warrants to purchase additional shares of COCP, InCellDx, Inc., Xenetic, Phio and Neovasc, respectively. We recorded the changes in the fair value of the options and warrants in Fair value changes of derivative instruments, net in our Consolidated Statement of Operations. We also recorded the fair value of the options and warrants in Investments, net in our Consolidated Balance Sheet. See further discussion of the Company’s options and warrants in Note 18 and Note 19.
Investments in variable interest entities
We have determined that we hold variable interests in Zebra Biologics, Inc. (“Zebra”). We made this determination as a result of our assessment that Zebra does not have sufficient resources to carry out its principal activities without additional financial support.
We own 1,260,000 shares of Zebra Series A-2 Preferred Stock and 900,000 shares of Zebra restricted common stock (ownership 29 % at December 31, 2018). Zebra is a privately held biotechnology company focused on the discovery and development of biosuperior antibody therapeutics and complex drugs. Dr. Richard Lerner, M.D., a member of our Board of Directors, is a founder of Zebra and, along with Dr. Frost, serves as a member of Zebra’s Board of Directors.
In order to determine the primary beneficiary of Zebra, we evaluated our investment and our related parties’ investment, as well as our investment combined with the related party group’s investment to identify if we had the power to direct the activities that most significantly impact the economic performance of Zebra. Based on the capital structure, governing documents and overall business operations of Zebra, we determined that, while a VIE, we do not have the power to direct the activities that most significantly impact Zebra’s economic performance and have no obligation to fund expected losses. We did determine, however, that we can significantly influence the success of Zebra through our board representation and voting power. Therefore, we have the ability to exercise significant influence over Zebra’s operations and account for our investment in Zebra under the equity method.
Other
112
Note 5 Composition of Certain Financial Statement Captions
113
For the years ended December 31, |
|||||||
(In thousands) |
2018 |
2017 |
|||||
Accounts receivable, net |
|||||||
Accounts receivable |
$ |
$ |
|||||
Less: allowance for doubtful accounts |
( |
) |
( |
) |
|||
$ |
$ |
||||||
Inventories, net |
|||||||
Consumable supplies |
$ |
$ |
|||||
Finished products |
|||||||
Work in-process |
|||||||
Raw materials |
|||||||
Less: inventory reserve |
( |
) |
( |
) |
|||
$ |
$ |
||||||
Other current assets and prepaid expenses |
|||||||
Other receivables |
$ |
$ |
|||||
Taxes recoverable |
|||||||
Prepaid supplies |
|||||||
Prepaid insurance |
|||||||
Other |
|||||||
$ |
$ |
||||||
Property, plant and equipment, net: |
|||||||
Machinery, medical and other equipment |
$ |
$ |
|||||
Leasehold improvements |
|||||||
Furniture and fixtures |
|||||||
Automobiles and aircraft |
|||||||
Software |
|||||||
Building |
|||||||
Land |
|||||||
Construction in process |
|||||||
Less: accumulated depreciation |
( |
) |
( |
) |
|||
$ |
$ |
||||||
Intangible assets, net: |
|||||||
Customer relationships |
$ |
$ |
|||||
Technologies |
|||||||
Trade names |
|||||||
Covenants not to compete |
|||||||
Licenses |
|||||||
Product registrations |
|||||||
Other |
|||||||
Less: accumulated amortization |
( |
) |
( |
) |
|||
$ |
$ |
||||||
For the years ended December 31, |
|||||||
(In thousands) |
2018 |
2017 |
|||||
Accrued expenses: |
|||||||
Contract liabilities |
$ |
$ |
|||||
Employee benefits |
|||||||
Taxes payable |
|||||||
Contingent consideration |
|||||||
Clinical trials |
|||||||
Capital leases short-term |
|||||||
Milestone payment |
|||||||
Professional fees |
|||||||
Other |
|||||||
$ |
$ |
||||||
Other long-term liabilities: |
|||||||
Contract liabilities |
$ |
$ |
|||||
Line of credit |
|||||||
Contingent consideration |
|||||||
Capital leases long-term |
|||||||
Mortgages and other debts payable |
|||||||
Other |
|||||||
$ |
$ |
||||||
All of our intangible assets and goodwill acquired relate to our acquisitions of principally OPKO Renal, OPKO Biologics, EirGen Pharma Limited (“EirGen”) and BioReference. We amortize intangible assets with definite lives on a straight-line basis over their estimated useful lives. The estimated useful lives by asset class are as follows: technologies - 5-17 years, customer relationships - 7-20 years, product registrations - 7-10 years, covenants not to compete - 5 years, trade names - 5-10 years, other 9-10 years. We do not anticipate capitalizing the cost of product registration renewals, rather we expect to expense these costs, as incurred. Our goodwill is not tax deductible for income tax purposes in any jurisdiction in which we operate.
The changes in value of the intangible assets and goodwill during the years ended December 31, 2018 and 2017 are primarily due to foreign currency fluctuations between the Chilean Peso, the Euro and the Shekel against the U.S. dollar.
(In thousands) |
Beginning
balance
|
Charged
to
expense
|
Written-off |
Charged
to other
|
Ending
balance
|
|||||||||||
2018 |
||||||||||||||||
Allowance for doubtful accounts |
$ |
( |
) |
( |
) |
$ |
( |
) |
||||||||
Inventory reserve |
$ |
( |
) |
( |
) |
$ |
( |
) |
||||||||
Tax valuation allowance |
$ |
( |
) |
( |
) |
$ |
( |
) |
||||||||
2017 |
||||||||||||||||
Allowance for doubtful accounts |
$ |
( |
) |
( |
) |
$ |
( |
) |
||||||||
Inventory reserve |
$ |
( |
) |
( |
) |
( |
) |
$ |
( |
) |
||||||
Tax valuation allowance |
$ |
( |
) |
( |
) |
( |
) |
$ |
( |
) |
||||||
114
2018 |
2017 |
||||||||||||||||||||||||||
(In thousands) |
Balance at January 1 |
Goodwill impairment |
Foreign exchange and other |
Balance at December 31st |
Balance at January 1 |
Foreign exchange |
Balance at December 31 |
||||||||||||||||||||
Pharmaceuticals |
|||||||||||||||||||||||||||
CURNA |
$ |
$ |
$ |
$ |
$ |
$ |
|||||||||||||||||||||
EirGen |
( |
) |
|||||||||||||||||||||||||
FineTech |
( |
) |
|||||||||||||||||||||||||
OPKO Biologics |
|||||||||||||||||||||||||||
OPKO Chile |
( |
) |
|||||||||||||||||||||||||
OPKO Health Europe |
( |
) |
|||||||||||||||||||||||||
OPKO Renal |
|||||||||||||||||||||||||||
Transition Therapeutics |
( |
) |
|||||||||||||||||||||||||
Diagnostics |
|||||||||||||||||||||||||||
BioReference |
|||||||||||||||||||||||||||
OPKO Diagnostics |
|||||||||||||||||||||||||||
OPKO Lab |
|||||||||||||||||||||||||||
$ |
$ |
( |
) |
$ |
( |
) |
$ |
$ |
$ |
$ |
|||||||||||||||||
115
Note 6 Debt
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. Borrowings under the line of credit will bear interest at a rate of 10 % per annum and may be repaid and reborrowed at any time. The credit agreement includes various customary remedies for the lender following an event of default, including the acceleration of repayment of outstanding amounts under line of credit. The line of credit matures on November 8, 2023. As of as of December 31, 2018, no funds were borrowed under the line of credit, and in February 2019, we repaid amounts borrowed in 2019 and terminated the credit agreement.
In February 2018, we issued a series of 5 % Convertible Promissory Notes (the “2023 Convertible Notes”) in the aggregate principal amount of $55.0 million. The 2023 Convertible Notes mature 5 years from the date of issuance. Each holder of a 2023 Convertible Note has the option, from time to time, to convert all or any portion of the outstanding principal balance of such 2023 Convertible Note, together with accrued and unpaid interest thereon, into shares of our Common Stock, par value $0.01 per share, at a conversion price of $5.00 per share of Common Stock (the “Shares”). We may redeem all or any part of the then issued and outstanding 2023 Convertible Notes, together with accrued and unpaid interest thereon, pro ratably among the holders, upon no fewer than 30 days, and no more than 60 days, notice to the holders. The 2023 Convertible Notes contain customary events of default and representations and warranties of OPKO.
The issuance of the 2023 Convertible Notes and the issuance of the Shares, if any, upon conversion thereof was not, and will not be, respectively, registered under the Securities Act, pursuant to the exemption provided by Section 4(a)(2) thereof, and we have not agreed to register the Shares if or when such Shares are issued. Purchasers of the 2023 Convertible Notes include an affiliate of Dr. Phillip Frost, M.D., our Chairman and Chief Executive Officer, and Dr. Jane H. Hsiao, Ph.D., MBA, our Vice-Chairman and Chief Technical Officer.
In January 2013, we entered into note purchase agreements (the “2033 Senior Notes”) with qualified institutional buyers and accredited investors (collectively, the “Purchasers”) in a private placement in reliance on exemptions from registration under the Securities Act. The 2033 Senior Notes were issued on January 30, 2013. The 2033 Senior Notes, which totaled $175.0 million in original principal amount, bear interest at the rate of 3.0 % per year, payable semiannually on February 1 and August 1 of each year. The 2033 Senior Notes will mature on February 1, 2033, unless earlier repurchased, redeemed or converted. Upon a fundamental change as defined in the Indenture, dated as of January 30, 2013, by and between the Company and Wells Fargo Bank N.A., as trustee, governing the 2033 Senior Notes (the “Indenture”), subject to certain exceptions, the holders may require us to repurchase all or any portion of their 2033 Senior Notes for cash at a repurchase price equal to 100 % of the principal amount of the 2033 Senior Notes being repurchased, plus any accrued and unpaid interest to but not including the related fundamental change repurchase date.
(In thousands) |
2033 Senior Notes |
Discount |
Debt Issuance Cost |
Total |
|||||||||||
Balance at December 31, 2017 |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
|||||||
Amortization of debt discount and debt issuance costs |
— |
||||||||||||||
Balance at December 31, 2018 |
$ |
$ |
( |
) |
$ |
$ |
|||||||||
(In thousands) |
Embedded conversion option |
2033 Senior Notes |
Discount |
Debt Issuance Cost |
Total |
||||||||||||||
Balance at December 31, 2016 |
$ |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
||||||||||
Amortization of debt discount and debt issuance costs |
— |
— |
|||||||||||||||||
Change in fair value of embedded derivative |
( |
) |
— |
— |
— |
( |
) |
||||||||||||
Reclassification of embedded derivatives to equity |
( |
) |
— |
— |
— |
( |
) |
||||||||||||
Balance at December 31, 2017 |
$ |
$ |
$ |
( |
) |
$ |
( |
) |
$ |
||||||||||
The 2033 Senior Notes will be convertible at any time on or after November 1, 2032, through the second scheduled trading day immediately preceding the maturity date, at the option of the holders. Additionally, holders may convert their 2033
116
Senior Notes prior to the close of business on the scheduled trading day immediately preceding November 1, 2032, under the following circumstances: (1) conversion based upon satisfaction of the trading price condition relating to the 2033 Senior Notes; (2) conversion based on the Common Stock price; (3) conversion based upon the occurrence of specified corporate events; or (4) if we call the 2033 Senior Notes for redemption. The 2033 Senior Notes will be convertible into cash, shares of our Common Stock, or a combination of cash and shares of Common Stock, at our election unless we have made an irrevocable election of net share settlement. The initial conversion rate for the 2033 Senior Notes will be 141.48 shares of Common Stock per $1,000 principal amount of 2033 Senior Notes (equivalent to an initial conversion price of approximately $7.07 per share of Common Stock), and will be subject to adjustment upon the occurrence of certain events. In addition, we will, in certain circumstances, increase the conversion rate for holders who convert their 2033 Senior Notes in connection with a make-whole fundamental change (as defined in the Indenture). Holders of the 2033 Senior Notes may require us to repurchase the 2033 Senior Notes for 100 % of their principal amount, plus accrued and unpaid interest, on February 1, 2019, February 1, 2023 and February 1, 2028, or following the occurrence of a fundamental change as defined in the indenture governing the 2033 Senior Notes. On February 1, 2019, approximately $28.8 million aggregate principal amount of 2033 Senior Notes were tendered by holders pursuant to such holders’ option to require us to repurchase the 2033 Senior Notes.
Before February 1, 2019, we may redeem for cash any or all of the 2033 Senior Notes but only if the last reported sale price of our Common Stock exceeds 130 % of the applicable conversion price for at least 20 trading days during the 30 consecutive trading day period ending on the trading day immediately prior to the date on which we deliver the redemption notice. The redemption price will equal 100 % of the principal amount of the 2033 Senior Notes to be redeemed, plus any accrued and unpaid interest to but not including the redemption date. On or after February 1, 2019, we may redeem for cash any or all of the 2033 Senior Notes at a redemption price of 100 % of the principal amount of the 2033 Senior Notes to be redeemed, plus any accrued and unpaid interest up to but not including the redemption date.
The terms of the 2033 Senior Notes, include, among others: (i) rights to convert into shares of our Common Stock, including upon a fundamental change; and (ii) a coupon make-whole payment in the event of a conversion by the holders of the 2033 Senior Notes on or after February 1, 2017 but prior to February 1, 2019. We determined that these specific terms were considered to be embedded derivatives. Embedded derivatives are required to be separated from the host contract, the 2033 Senior Notes, and carried at fair value when: (a) the embedded derivative possesses economic characteristics that are not clearly and closely related to the economic characteristics of the host contract; and (b) a separate, stand-alone instrument with the same terms would qualify as a derivative instrument. We concluded that the embedded derivatives within the 2033 Senior Notes meet these criteria for periods prior to February 1, 2017 and, as such, were valued separate and apart from the 2033 Senior Notes and recorded at fair value each reporting period.
For accounting and financial reporting purposes, we combined these embedded derivatives and valued them together as one unit of accounting. In 2017, certain terms of the embedded derivatives expired pursuant to the original agreement and the embedded derivatives no longer met the criteria to be separated from the host contract and, as a result, the embedded derivatives were no longer required to be valued separate and apart from the 2033 Senior Notes and were reclassified to additional paid in capital. Accordingly, there was no derivative income (loss) for the year ended December 31, 2018.
From 2013 to 2016, holders of the 2033 Senior Notes converted $143.2 million in aggregate principal amount into an aggregate of 21,539,873 shares of the Company’s Common Stock.
On November 5, 2015, BioReference and certain of its subsidiaries entered into a credit agreement with JPMorgan Chase Bank, N.A. (“CB”), as lender and administrative agent, as amended (the “Credit Agreement”). The Credit Agreement provides for a $175.0 million secured revolving credit facility and includes a $20.0 million sub-facility for swingline loans and a $20.0 million sub-facility for the issuance of letters of credit. BioReference may increase the credit facility to up to $275.0 million on a secured basis, subject to the satisfaction of specified conditions. The Credit Agreement matures on November 5, 2020 and is guaranteed by all of BioReference’s domestic subsidiaries. The Credit Agreement is also secured by substantially all assets of BioReference and its domestic subsidiaries, as well as a non-recourse pledge by us of our equity interest in BioReference. Availability under the Credit Agreement is based on a borrowing base comprised of eligible accounts receivables of BioReference and certain of its subsidiaries, as specified therein. As of December 31, 2018, $2.0 million additional funds were available to be borrowed under the Credit Agreement. Principal under the Credit Agreement is due upon maturity on November 5, 2020.
At BioReference’s option, borrowings under the Credit Agreement (other than swingline loans) will bear interest at (i) the CB floating rate (defined as the higher of (a) the prime rate and (b) the LIBOR rate (adjusted for statutory reserve requirements for Eurocurrency liabilities) for an interest period of one month plus 2.50 %) plus an applicable margin of 0.35 % for the first 12 months and 0.50 % thereafter or (ii) the LIBOR rate (adjusted for statutory reserve requirements for Eurocurrency liabilities) plus an applicable margin of 1.35 % for the first 12 months and 1.50 % thereafter. Swingline loans will bear interest at the CB
117
floating rate plus the applicable margin. The Credit Agreement also calls for other customary fees and charges, including an unused commitment fee of 0.50 % of the lending commitments.
On March 17, 2017, BioReference and certain of its subsidiaries entered into Amendment No. 3 to Credit Agreement, which amended the Credit Agreement to permit BioReference and its subsidiaries to dividend cash to the Company in the form of an intercompany loan, in an aggregate amount not to exceed $55.0 million. On August 7, 2017, BioReference and certain of its subsidiaries entered into Amendment No. 4 to Credit Agreement, which amended the Credit Agreement to permit BioReference and its subsidiaries to dividend cash to the Company in the form of an additional intercompany loan, in an aggregate amount not to exceed $35.0 million. On November 8, 2017, BioReference and certain of its subsidiaries entered into Amendment No. 5 to Credit Agreement, which amended the Credit Agreement to, among other things, ease certain thresholds that require increased reporting by BioReference and reduce the pro forma availability condition for BioReference to make certain cash dividends to the Company. On December 22, 2017, BioReference and certain of its subsidiaries entered into Amendment No. 6 to Credit Agreement, which amended the Credit Agreement to, among other things, permit BioReference and its subsidiaries to dividend cash to the Company in the form of intercompany loans, in an aggregate amount not to exceed $45.0 million. The other terms of the Credit Agreement remain unchanged.
In February 2018, BioReference and certain of its subsidiaries entered into Amendment No. 7 to the Credit Agreement, which amended the Credit Agreement to permit BioReference and its subsidiaries to use cash on hand, up to a maximum amount set forth in the amendment, to meet the availability requirements that otherwise would trigger (i) covenants that would require BioReference to maintain a minimum fixed charge coverage ratio and provide certain increased reporting under the Credit Agreement and (ii) CB’s right, as agent for the lenders under the Credit Agreement, to exercise sole dominion over funds held in certain accounts of BioReference. The other terms of the Credit Agreement remain unchanged.
On February 26, 2019, BioReference and certain of its subsidiaries entered into Amendment No. 8 to the Credit Agreement, which amended the Credit Agreement to add back certain cost savings resulting from work force reductions in the 2018 fiscal year to the calculation of EBITDA for purposes of complying with the minimum fixed charge coverage ratio covenant. The other terms of the Credit Agreement remain unchanged.
The Credit Agreement contains customary covenants and restrictions, including, without limitation, covenants that require BioReference and its subsidiaries to maintain a minimum fixed charge coverage ratio if availability under the new credit facility falls below a specified amount and to comply with laws and restrictions on the ability of BioReference and its subsidiaries to incur additional indebtedness or to pay dividends and make certain other distributions to the Company, subject to certain exceptions as specified therein. Failure to comply with these covenants would constitute an event of default under the Credit Agreement, notwithstanding the ability of BioReference to meet its debt service obligations. The Credit Agreement also includes various customary remedies for the lenders following an event of default, including the acceleration of repayment of outstanding amounts under the Credit Agreement and execution upon the collateral securing obligations under the Credit Agreement. Substantially all the assets of BioReference and its subsidiaries are restricted from sale, transfer, lease, disposal or distributions to the Company, subject to certain exceptions. BioReference and its subsidiaries net assets as of December 31, 2018 were approximately $916.4 million, which includes goodwill of $401.8 million and intangible assets of $405.3 million.
118
In addition to the Credit Agreement with CB, we have line of credit agreements with eleven other financial institutions as of December 31, 2018 and 2017 in the U.S., Chile and Spain. These lines of credit are used primarily as a source of working capital for inventory purchases.
(Dollars in thousands) |
Balance Outstanding |
|||||||||||||
Lender |
Interest rate on
borrowings at December 31, 2018
|
Credit line
capacity
|
December 31, 2018 |
December 31,
2017
|
||||||||||
JP Morgan Chase |
$ |
$ |
$ |
|||||||||||
Itau Bank |
||||||||||||||
Bank of Chile |
||||||||||||||
BICE Bank |
||||||||||||||
BBVA Bank |
||||||||||||||
Security Bank |
||||||||||||||
Estado Bank |
||||||||||||||
Santander Bank |
||||||||||||||
Scotiabank |
||||||||||||||
Banco De Sabadell |
||||||||||||||
Banco Bilbao Vizcaya |
||||||||||||||
Santander Bank |
||||||||||||||
Total |
$ |
$ |
$ |
|||||||||||
At December 31, 2018 and 2017, the weighted average interest rate on our lines of credit was approximately 4.7 % and 4.2 %, respectively.
At December 31, 2018 and 2017, we had notes payable and other debt (excluding the 2033 Senior Notes, the 2023 Convertible Notes, the Credit Agreement and amounts outstanding under lines of credit) as follows:
(In thousands) |
December 31, 2018 |
December 31, 2017 |
|||||
Current portion of notes payable |
$ |
$ |
|||||
Other long-term liabilities |
|||||||
Total |
$ |
$ |
|||||
119
Note 7 Shareholders’ Equity
Our authorized capital stock consists of 750,000,000 shares of Common Stock, par value $0.01 per share, and 10,000,000 shares of Preferred Stock, par value $0.01 per share.
Private placements of common stock
On November 8, 2018, we entered into stock purchase agreements with certain investors pursuant to which we agreed to sell to such investors in private placements an aggregate of approximately 26.5 million shares of our Common Stock (the “Shares”) at a purchase price of $3.49 per share, which was the closing bid price of our Common Stock on the NASDAQ Global Select Market (“NASDAQ”) on such date, for an aggregate purchase price of $92.5 million.
The investors in the private placements include an affiliate of Dr. Phillip Frost, our Chairman and Chief Executive Officer ($70 million), and Dr. Jane Hsiao, our Vice Chairman and Chief Technical Officer ($2 million).
The issuance of the Shares were made in reliance on the exemption from registration provided in Section 4(a)(2) of the Securities Act based upon the representations made by the investors that they are “accredited investors” and that they are purchasing the Shares without a present view toward a distribution of the Shares.
Common stock
Subject to the rights of the holders of any shares of Preferred Stock currently outstanding or which may be issued in the future, the holders of the Common Stock are entitled to receive dividends from our funds legally available when, as and if declared by our Board of Directors, and are entitled to share ratably in all of our assets available for distribution to holders of Common Stock upon the liquidation, dissolution or winding-up of our affairs subject to the liquidation preference, if any, of any then outstanding shares of Preferred Stock. Holders of our Common Stock do not have any preemptive, subscription, redemption or conversion rights. Holders of our Common Stock are entitled to one vote per share on all matters which they are entitled to vote upon at meetings of stockholders or upon actions taken by written consent pursuant to Delaware corporate law. The holders of our Common Stock do not have cumulative voting rights, which means that the holders of a plurality of the outstanding shares can elect all of our directors. All of the shares of our Common Stock currently issued and outstanding are fully-paid and nonassessable. No dividends have been paid to holders of our Common Stock since our incorporation, and no cash dividends are anticipated to be declared or paid on our Common Stock in the reasonably foreseeable future.
Preferred Stock
Under our certificate of incorporation, our Board of Directors has the authority, without further action by stockholders, to designate up to 10 million shares of Preferred Stock in one or more series and to fix or alter, from time to time, the designations, powers and rights of each series of Preferred Stock and the qualifications, limitations or restrictions of any series of Preferred Stock, including dividend rights, dividend rate, conversion rights, voting rights, rights and terms of redemption (including sinking fund provisions), redemption price or prices, and the liquidation preference of any wholly issued series of Preferred Stock, any or all of which may be greater than the rights of the Common Stock, and to establish the number of shares constituting any such series.
120
Note 8 Accumulated Other Comprehensive Income (Loss)
(In thousands) |
Foreign currency translation |
Unrealized
gain (loss) in
Accumulated
OCI
|
Total |
||||||||
Balance at December 31, 2017 |
$ |
( |
) |
$ |
$ |
( |
) |
||||
Other comprehensive income (loss) before reclassifications |
( |
) |
( |
) |
|||||||
Reclassification adjustment due to adoption of ASU 2016-01 |
( |
) |
( |
) |
|||||||
Net other comprehensive income (loss) |
( |
) |
( |
) |
( |
) |
|||||
Balance at December 31, 2018 |
$ |
( |
) |
$ |
$ |
( |
) |
||||
(In thousands) |
Foreign currency translation |
Unrealized gain (loss) in Accumulated OCI |
Total |
||||||||
Balance at December 31, 2016 |
$ |
( |
) |
$ |
$ |
( |
) |
||||
Other comprehensive income (loss) before reclassifications |
|||||||||||
Reclassification adjustments for losses included in net loss, net of tax |
( |
) |
( |
) |
|||||||
Net other comprehensive income (loss) |
|||||||||||
Balance at December 31, 2017 |
$ |
( |
) |
$ |
$ |
( |
) |
||||
121
Note 9 Equity-Based Compensation
We maintain six equity-based incentive compensation plans, the 2016 Equity Incentive Plan, the Acuity Pharmaceuticals, Inc. 2003 Equity Incentive Plan, the 2007 Equity Incentive Plan, the 2000 Stock Option Plan, the Modigene Inc. 2005 Stock Incentive Plan and the Modigene Inc. 2007 Equity Incentive Plan that provide for grants of stock options and restricted stock to our directors, officers, key employees and certain outside consultants. Equity awards granted under our 2016 Equity Incentive Plan are exercisable for a period of up to 10 years from the date of grant. Equity awards granted under our 2007 Equity Incentive Plan are exercisable for a period of either 7 years or 10 years from the date of grant. Equity awards granted under our 2000 Stock Option Plan, 2003 Equity Incentive Plan and the two Modigene Plans are exercisable for a period of up to 10 years from date of grant. Vesting periods range from immediate to 5 years.
We classify the cash flows resulting from the tax benefit that arises when the tax deductions exceed the compensation cost recognized for those equity awards (excess tax benefits) as cash flows from operations. There were no excess tax benefits for the years ended December 31, 2018, 2017, and 2016.
Equity-based compensation arrangements to non-employees are accounted for at their fair value on the measurement date. The measurement of equity-based compensation to non-employees is subject to periodic adjustment over the vesting period of the equity instruments.
Valuation and Expense Information
We recorded equity-based compensation expense of $21.8 million, $28.3 million and $42.7 million for the years ended December 31, 2018, 2017, and 2016, respectively, all of which were reflected as operating expenses. Of the $21.8 million of equity based compensation expense recorded in the year ended December 31, 2018, $14.7 million was recorded as selling, general and administrative expenses, $4.2 million was recorded as research and development expenses and $2.8 million was recorded as a cost of revenue. Of the $28.3 million of equity based compensation expense recorded in the year ended December 31, 2017, $21.2 million was recorded as selling, general and administrative expense, $5.1 million was recorded as research and development expenses and $2.0 million was recorded as a cost of revenue. Of the $42.7 million of equity based compensation expense recorded in the year ended December 31, 2016, $33.4 million was recorded as selling, general and administrative expense, $7.5 million was recorded as research and development expenses and 1.8 million was recorded as cost of revenue.
We estimate forfeitures of stock options and recognize compensation cost only for those awards expected to vest. Forfeiture rates are determined for all employees and non-employee directors based on historical experience and our estimate of future vesting. Estimated forfeiture rates are adjusted from time to time based on actual forfeiture experience.
As of December 31, 2018, there was $24.6 million of unrecognized compensation cost related to the stock options granted under our equity-based incentive compensation plans. Such cost is expected to be recognized over a weighted-average period of approximately 1.48 years.
Stock Options
We estimate the fair value of each stock option on the date of grant using the Black-Scholes-Merton Model option-pricing formula and amortize the fair value to expense over the stock option’s vesting period using the straight-line attribution approach for employees and non-employee directors, and for awards issued to non-employees we recognize compensation expense on a graded basis, with most of the compensation expense being recorded during the initial periods of vesting. We apply the following assumptions in our Black-Scholes-Merton Model option-pricing formula:
|
Year Ended
December 31,
2018
|
Year Ended
December 31,
2017
|
Year Ended
December 31,
2016
|
|||
Expected term (in years) |
3.0 - 10.0 |
3.0 - 10.0 |
1.0 - 10.0 |
||
Risk-free interest rate |
2.32% - 3.09% |
1.32% - 2.41% |
0.71% - 2.51% |
||
Expected volatility |
40% - 54% |
38% - 55% |
38% - 64% |
||
Expected dividend yield |
|||||
Expected Term: For the expected term of options granted to employees and non-employee directors, we used an estimate of the expected option life based on historical experience. The expected term of stock options issued to non-employee consultants is the remaining contractual life of the options issued.
122
Risk-Free Interest Rate: The risk-free interest rate is based on the rates paid on securities issued by the U.S. Treasury with a term approximating the expected life of the option.
Expected Volatility: The expected volatility for stock options was based on the historical volatility of our Common Stock.
Expected Dividend Yield: We do not intend to pay dividends on Common Stock for the foreseeable future. Accordingly, we used a dividend yield of zero in the assumptions.
We maintain incentive stock plans that provide for the grants of stock options to our directors, officers, employees and non-employee consultants. As of December 31, 2018, there were 26,668,368 shares of Common Stock reserved for issuance under our 2016 Equity Incentive Plan and our 2007 Equity Incentive Plan. We intend to issue new shares upon the exercise of stock options. Stock options granted under these plans have been granted at an option price equal to the closing market value of the stock on the date of the grant. Stock options granted under these plans to employees typically become exercisable over four years in equal annual installments after the date of grant, and stock options granted to non-employee directors become exercisable in full one-year after the grant date, subject to, in each case, continuous service with us during the applicable vesting period. We assumed stock options to grant Common Stock as part of the mergers with Acuity Pharmaceuticals, Inc., Froptix, Inc., OPKO Biologics and BioReference, which reflected various vesting schedules, including monthly vesting to employees and non-employee consultants.
Options |
Number of
options
|
Weighted
average
exercise
price
|
Weighted
average
remaining
contractual
term (years)
|
Aggregate
intrinsic value
(in thousands)
|
||||||||
Outstanding at December 31, 2017 |
$ |
$ |
||||||||||
Granted |
$ |
|||||||||||
Exercised |
( |
) |
$ |
|||||||||
Forfeited |
( |
) |
$ |
|||||||||
Expired |
( |
) |
$ |
|||||||||
Outstanding at December 31, 2018 |
$ |
$ |
||||||||||
Vested and expected to vest at December 31, 2018 |
$ |
$ |
||||||||||
Exercisable at December 31, 2018 |
$ |
$ |
||||||||||
The total intrinsic value of stock options exercised for the years ended December 31, 2018, 2017, and 2016 was $0.5 million, $6.4 million and $9.9 million, respectively.
123
Note 10 Income Taxes
We operate and are required to file tax returns in the U.S. and various foreign jurisdictions.
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Current |
|||||||||||
Federal |
$ |
$ |
$ |
||||||||
State |
( |
) |
( |
) |
|||||||
Foreign |
( |
) |
( |
) |
( |
) |
|||||
( |
) |
( |
) |
||||||||
Deferred |
|||||||||||
Federal |
( |
) |
|||||||||
State |
( |
) |
|||||||||
Foreign |
( |
) |
|||||||||
( |
) |
||||||||||
Total, net |
$ |
$ |
( |
) |
$ |
||||||
(In thousands) |
December 31, 2018 |
December 31, 2017 |
|||||
Deferred income tax assets: |
|||||||
Federal net operating loss |
$ |
$ |
|||||
State net operating loss |
|||||||
Foreign net operating loss |
|||||||
Research and development expense |
|||||||
Tax credits |
|||||||
Stock options |
|||||||
Accruals |
|||||||
Equity investments |
|||||||
Bad debts |
|||||||
Lease liability |
|||||||
Foreign credits |
|||||||
Equity securities |
|||||||
Other |
|||||||
Deferred income tax assets |
|||||||
Deferred income tax liabilities: |
|||||||
Intangible assets |
( |
) |
( |
) |
|||
Fixed assets |
( |
) |
( |
) |
|||
Other |
( |
) |
( |
) |
|||
Deferred income tax liabilities |
( |
) |
( |
) |
|||
Net deferred income tax assets (liabilities) |
( |
) |
|||||
Valuation allowance |
( |
) |
( |
) |
|||
Net deferred income tax liabilities |
$ |
( |
) |
$ |
( |
) |
|
As of December 31, 2018, we have federal, state and foreign net operating loss carryforwards of approximately $606.6 million, $801.8 million and $152.3 million, respectively, that expire at various dates through 2038 unless indefinite in nature. Included in the foreign net operating losses is $86.1 million related to OPKO Biologics. As of December 31, 2018, we have research and development tax credit carryforwards of approximately $21.7 million that expire in varying amounts through
124
2038. As of each reporting date, management considers new evidence, both positive and negative, that could affect its view of the future realization of deferred tax assets. We have determined a valuation allowance is required against all of our net deferred tax assets that we do not expect to be utilized by the reversing of deferred income tax liabilities.
Under Section 382 of the Internal Revenue Code of 1986, as amended, certain significant changes in ownership may restrict the future utilization of our income tax loss carryforwards and income tax credit carryforwards in the U.S. The annual limitation is equal to the value of our stock immediately before the ownership change, multiplied by the long-term tax-exempt rate (i.e., the highest of the adjusted federal long-term rates in effect for any month in the three-calendar-month period ending with the calendar month in which the change date occurs). This limitation may be increased under the IRC Section 338 Approach (IRS approved methodology for determining recognized Built-In Gain). As a result, federal net operating losses and tax credits may expire before we are able to fully utilize them.
During 2008, we conducted a study to determine the impact of the various ownership changes that occurred during 2007 and 2008. As a result, we have concluded that the annual utilization of our net operating loss carryforwards (“NOLs”) and tax credits is subject to a limitation pursuant to Internal Revenue Code Section 382. Under the tax law, such NOLs and tax credits are subject to expiration from 15 to 20 years after they were generated. As a result of the annual limitation that may be imposed on such tax attributes and the statutory expiration period, some of these tax attributes may expire prior to our being able to use them. There is no current impact on these financial statements as a result of the annual limitation. This study did not conclude whether OPKO’s predecessor, eXegenics, pre-merger NOLs were limited under Section 382. As such, of the $606.6 million of federal net operating loss carryforwards, at least approximately $53.4 million may not be able to be utilized.
We file federal income tax returns in the U.S. and various foreign jurisdictions, as well as with various U.S. states and the Ontario and Nova Scotia provinces in Canada. We are subject to routine tax audits in all jurisdictions for which we file tax returns. Tax audits by their very nature are often complex and can require several years to complete. It is reasonably possible that some audits will close within the next twelve months, which we do not believe would result in a material change to our accrued uncertain tax positions.
U.S. Federal: Under the tax statute of limitations applicable to the Internal Revenue Code, we are no longer subject to U.S. federal income tax examinations by the Internal Revenue Service for years before 2015. However, because we are carrying forward income tax attributes, such as net operating losses and tax credits from 2015 and earlier tax years, these attributes can still be audited when utilized on returns filed in the future.
State: Under the statute of limitations applicable to most state income tax laws, we are no longer subject to state income tax examinations by tax authorities for years before 2014 in states in which we have filed income tax returns. Certain states may take the position that we are subject to income tax in such states even though we have not filed income tax returns in such states and, depending on the varying state income tax statutes and administrative practices, the statute of limitations in such states may extend to years before 2014.
Foreign: Under the statute of limitations applicable to our foreign operations, we are generally no longer subject to tax examination for years before 2013 in jurisdictions where we have filed income tax returns.
Tax Cuts and Jobs Act
On December 22, 2017, the 2017 Tax Cuts and Jobs Act (the “Tax Act”) was enacted into law and the new legislation contains several key tax provisions, including a reduction of the corporate income tax rate from 35% to 21% effective January 1, 2018 and a one-time mandatory transition tax on accumulated foreign earnings, among others. We were required to recognize the effect of the tax law changes in the period of enactment, such as remeasuring our U.S. deferred tax assets and liabilities, as well as reassessing the net realizability of our deferred tax assets and liabilities. In December 2017, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act (SAB 118), which allowed us to record provisional amounts during a measurement period not to extend beyond one year of the enactment date. As of December 22, 2018 we completed our analysis in accordance with SAB 118 and recorded immaterial adjustments.
Effective January 1, 2018, the Tax Act provides for a new global intangible low-taxed income (GILTI) provision. Under the GILTI provision, certain foreign subsidiary earnings in excess of an allowable return on the foreign subsidiary’s tangible assets are included in U.S. taxable income. The Company currently estimates GILTI will be immaterial for the year ended December 31, 2018, although interpretive guidance continues to be issued and future guidance may impact this analysis. The Company has not recorded any deferred taxes for future GILTI inclusions as any future inclusions are expected to be treated as a period expense and offset by net operating loss carryforwards in the U.S.
The Tax Act affects the tax treatment of foreign earnings and profits (“E&P”) and results in a one-time transition tax on our post-1986 foreign E&P that we previously deferred from U.S. income tax expense. We determined that we did not owe any
125
transition tax and we have not provided for additional income taxes on any remaining undistributed foreign E&P not subject to the transition tax, or any outside tax basis differences inherent in our foreign subsidiaries. On January 15, 2019, the U.S. Department of Treasury released final regulations related to the one-time transition tax. Although the Company's assessment of these final rules is not complete, they are not expected to materially impact the Company’s financial statements.
Unrecognized Tax Benefits
As of December 31, 2018, 2017, and 2016, the total amount of gross unrecognized tax benefits was approximately $17.5 million, $21.3 million, and $27.5 million, respectively. As of December 31, 2018, the total gross unrecognized tax benefit of $17.5 million consisted of increases of $8.4 million as a result of current year activity, and decreases of $4.6 million as a result of the lapse of statutes of limitations. As of December 31, 2018, the total amount of unrecognized tax benefits that, if recognized, would affect our effective income tax rate was $(14.2 ) million. We account for any applicable interest and penalties on uncertain tax positions as a component of income tax expense and we recognized $(1.9 ) million and $0.4 million of interest expense for the years ended December 31, 2018 and 2017, respectively. As of December 31, 2017 and 2016, $(12.4 ) million and $6.1 million of the unrecognized tax benefits, if recognized, would have affected our effective income tax rate. We believe it is reasonably possible that approximately $1.3 million of unrecognized tax benefits may be recognized within the next twelve months, mainly due to anticipated statute of limitations lapses in various jurisdictions.
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Unrecognized tax benefits at beginning of period |
$ |
$ |
$ |
||||||||
Gross increases – tax positions in prior period |
|||||||||||
Gross increases – tax positions in current period |
|||||||||||
Gross decreases – tax positions in prior period |
( |
) |
( |
) |
( |
) |
|||||
Lapse of Statute of Limitations |
( |
) |
( |
) |
( |
) |
|||||
Unrecognized tax benefits at end of period |
$ |
$ |
$ |
||||||||
Other Income Tax Disclosures
For the years ended December 31, |
||||||||
2018 |
2017 |
2016 |
||||||
Federal statutory rate |
% |
% |
% |
|||||
State income taxes, net of federal benefit |
% |
% |
% |
|||||
Foreign income tax |
( |
)% |
( |
)% |
( |
)% |
||
Income Tax Refunds |
% |
% |
% |
|||||
Research and development tax credits |
% |
% |
% |
|||||
Non-Deductible components of Convertible Debt |
( |
)% |
% |
% |
||||
Valuation allowance |
( |
)% |
( |
)% |
% |
|||
Rate change effect |
% |
( |
)% |
% |
||||
Non-deductible items |
( |
)% |
( |
)% |
( |
)% |
||
Unrecognized tax benefits |
( |
)% |
( |
)% |
( |
)% |
||
Other |
( |
)% |
( |
)% |
( |
)% |
||
Total |
% |
( |
)% |
% |
||||
126
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Pre-tax income (loss): |
|||||||||||
U.S. |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Foreign |
( |
) |
( |
) |
( |
) |
|||||
Total |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Note 11 Related Party Transactions
On November 8, 2018, we entered into stock purchase agreements with certain investors pursuant to which we agreed to sell to such investors in private placements an aggregate of approximately 26.5 million shares of our Common Stock (the “Shares”) at a purchase price of $3.49 per share, which was the closing bid price of our Common Stock on the NASDAQ on such date, for an aggregate purchase price of $92.5 million. The investors in the private placements include an affiliate of Dr. Phillip Frost, our Chairman and Chief Executive Officer ($70 million), and Dr. Jane Hsiao, our Vice Chairman and Chief Technical Officer ($2 million).
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. Borrowings under the line of credit will bear interest at a rate of 10 % per annum and may be repaid and reborrowed at any time. The credit agreement includes various customary remedies for the lender following an event of default, including the acceleration of repayment of outstanding amounts under line of credit. The line of credit matures on November 8, 2023. As of as of December 31, 2018, no funds were borrowed under the line of credit. We repaid amounts borrowed in 2019 and terminated the line of credit on or around February 20, 2019.
In February 2018, we issued the 2023 Convertible Notes in the aggregate principal amount of $55.0 million. Refer to Note 6. Purchasers of the 2023 Convertible Notes include Dr. Hsiao and an affiliate of Dr. Frost.
We hold investments in Zebra (ownership 29 %), Neovasc (4 %), ChromaDex Corporation (0 %), MabVax (2 %), COCP (9 %), NIMS (1 % ) and BioCardia (5 %). These investments were considered related party transactions as a result of our executive management’s ownership interests and/or board representation in these entities. See further discussion of our investments in Note 4.
In February 2018, we invested an additional $1.0 million in COCP for a convertible note, which was converted into 538,544 shares of its common stock in May 2018. In April 2017, we invested an additional $1.0 million in COCP for 138,889 shares of its common stock, and in August 2016, we invested an additional $2.0 million in COCP for 162,602 shares of its common stock.
In November 2017, we invested an additional $3.0 million in Neovasc for 20,547 shares of its common stock, 20,547 Series A warrants, 20,547 Series B warrants and 8,221 Series C warrants, after adjusting for a 1-for-100 reverse stock split in 2018. In April 2018, we exercised our Series B warrants in a cashless exercise and received 1,069,090 shares of Neovasc common stock.
In July 2017, we invested an additional $0.1 million in MabVax for 50,714 shares of common stock and in May 2017, we invested an additional $0.5 million in MabVax for 1,667 shares of Series L Preferred Stock and 107,607 shares of Series I Preferred Stock. We had also invested an additional $1.0 million in MabVax in August 2016 for 69,300 shares of its common stock and warrants to purchase 138,600 shares of its common stock.
In October 2016, we entered into a consulting agreement to provide strategic advisory services to BioCardia. In connection with the consulting agreement, BioCardia granted us 418,977 common stock options, after adjusting for a 1-for-12 reverse stock split in 2017. In December 2016, we purchased 1,602,564 shares of BioCardia, after adjusting for the reverse stock split, from Dr. Frost for $2.5 million. We have also purchased shares of BioCardia in the open market. BioCardia is a
127
related party as a result of our executive management’s ownership interest and board representation in BioCardia and its predecessor, Tiger X Medical, Inc. In October 2016, BioCardia completed its merger with Tiger X Medical, Inc., to which Tiger X Medical, Inc. was the surviving entity and the name of the issuer was changed to BioCardia.
In November 2016, we entered into a Pledge Agreement with the Museum of Science, Inc. and the Museum of Science Endowment Fund, Inc. pursuant to which we will contribute an aggregate of $1.0 million over a four-year period for constructing, equipping and the general operation of the Frost Science Museum. Dr. Frost and Mr. Pfenniger serve on the Board of Trustees of the Frost Science Museum and Mr. Pfenniger is the Vice Chairman of the Board of Trustees.
We lease office space from Frost Real Estate Holdings, LLC (“Frost Holdings”) in Miami, Florida, where our principal executive offices are located. Effective January 1, 2017, we entered into an amendment to our lease agreement with Frost Holdings. The lease, as amended, is for approximately 29,500 square feet of space. The lease provides for payments of approximately $81 thousand per month in the first year increasing annually to $86 thousand per month in the third year, plus applicable sales tax. The rent is inclusive of operating expenses, property taxes and parking.
Our wholly-owned subsidiary, BioReference, purchases and uses certain products acquired from InCellDx, Inc., a company in which we hold a 29 % minority interest.
128
Note 12 Employee Benefit Plans
Note 13 Commitments and Contingencies
In connection with our acquisitions of CURNA, OPKO Diagnostics and OPKO Renal, we agreed to pay future consideration to the sellers upon the achievement of certain events. As a result, as of December 31, 2018, we have recorded $24.6 million as contingent consideration, with $2.4 million recorded within Accrued expenses and $22.2 million recorded within Other long-term liabilities in the accompanying Consolidated Balance Sheets. Refer to Note 5.
As previously disclosed, on September 7, 2018, the Securities and Exchange Commission (“SEC”) filed a lawsuit in the Southern District of New York (the “SEC Complaint”) against a number of individuals and entities (the “Defendants”), including the Company and its CEO and Chairman, Phillip Frost (“Dr. Frost”). The SEC alleged, among other things, that the Company (i) aided and abetted an illegal “pump and dump” scheme perpetrated by a number of the Defendants, and (ii) failed to file required Schedules 13D or 13G with the SEC. On December 27, 2018, the Company announced that the Company and Dr. Frost entered into settlement agreements with the Securities and Exchange Commission (the “Commission”), which upon approval of the court would resolve the SEC Complaint against each of them. The settlement was approved by the Court in January 2019. Pursuant to the settlement, and without admitting or denying any of the allegations of the Complaint, the Company is enjoined from violating Section 13(d) of the Exchange Act and paid a $100,000 penalty. Liability under Section 13(d) can be established without any showing of wrongful intent or negligence.
Following the SEC’s announcement of the SEC Complaint, we have been named in seven class action lawsuits and more than a dozen derivative suits relating to the allegations in the SEC Complaint among other matters. The Company intends to vigorously defend itself against the claims. Based on the early stages of these legal proceedings, at this time, the Company is not able to reasonably estimate a possible range of loss, if any, that may result from these allegations. For a more detailed discussion of pending matters, please see Part II, Item 1, “Legal Proceedings.”
In August 2017, we entered into a Commitment Letter (the “Commitment Letter”) with Veterans Accountable Care Group, LLC (“VACG”) in connection with the submission of a bid by its affiliate, the Veterans Accountable Care Organization, LLC (“VACO”) in response to a request for proposal (“RFP”) from the Veterans Health Administration (“VA”) regarding its Community Care Network. We were notified in January 2019 that the bid was awarded to a third party. If VACO were to have been successful in its bid, we would have acquired a fifteen percent (15 %) membership interest in VACO. In addition, BioReference, our wholly-owned subsidiary, would have provided laboratory services for the Community Care Network, a region which currently includes approximately 2,133,000 veterans in the states of Massachusetts, Maine, New Hampshire, Vermont, New York, Pennsylvania, New Jersey, Rhode Island, Connecticut, Maryland, Virginia, West Virginia, and North Carolina.
Pursuant to the Commitment Letter and had VACO been successful in its bid, we committed to provide, or to arrange from a third party lender, a line of credit for VACG in the amount of $50.0 million (the “Facility”). Funds drawn under the Facility would be contributed by VACG to VACO in order to satisfy the financial stability requirement of VACO in connection with its submission of the RFP. VACG would not be permitted to draw down on the Facility unless and until the VHA awards a contract to VACO. The Facility would have a maturity of five (5 ) years. Interest on the Facility would be payable at a rate equal to 6.5 % per annum, payable quarterly in arrears. The Facility would be subject to the negotiation of definitive documentation conditions customary for transactions of such type and otherwise acceptable to VACG and the lender under the Facility.
We accrue a liability for legal contingencies when we believe that it is both probable that a liability has been incurred and that we can reasonably estimate the amount of the loss. We review established accruals and adjust them to reflect ongoing negotiations, settlements, rulings, advice of legal counsel and other relevant information. To the extent new information is obtained and our views on the probable outcomes of claims, suits, assessments, investigations or legal proceedings change, changes in our accrued liabilities would be recorded in the period in which such determination is made. For the matters referenced in the paragraph below, the amount of liability is not probable or the amount cannot be reasonably estimated; and, therefore, accruals have not been made. In addition, in accordance with the relevant authoritative guidance, for matters which the likelihood of material loss is at least reasonably possible, we provide disclosure of the possible loss or range of loss; however, if a reasonable estimate cannot be made, we will provide disclosure to that effect.
129
From time to time, we may receive inquiries, document requests, Civil Investigative Demands (“CIDs”) or subpoenas from the Department of Justice, the Office of Inspector General and Office for Civil Rights (“OCR”) of the Department of Health and Human Services, the Centers for Medicare and Medicaid Services, various payors and fiscal intermediaries, and other state and federal regulators regarding investigations, audits and reviews. In addition to the matters discussed in this note, we are currently responding to CIDs, subpoenas or document requests for various matters relating to our laboratory operations. Some pending or threatened proceedings against us may involve potentially substantial amounts as well as the possibility of civil, criminal, or administrative fines, penalties, or other sanctions, which could be material. Settlements of suits involving the types of issues that we routinely confront may require monetary payments as well as corporate integrity agreements. Additionally, qui tam or “whistleblower” actions initiated under the civil False Claims Act may be pending but placed under seal by the court to comply with the False Claims Act’s requirements for filing such suits. Also, from time to time, we may detect issues of non-compliance with federal healthcare laws pertaining to claims submission and reimbursement practices and/or financial relationships with physicians, among other things. We may avail ourselves of various mechanisms to address these issues, including participation in voluntary disclosure protocols. Participating in voluntary disclosure protocols can have the potential for significant settlement obligations or even enforcement action. The Company generally has cooperated, and intends to continue to cooperate, with appropriate regulatory authorities as and when investigations, audits and inquiries arise.
In April 2017, the Civil Division of the United States Attorney’s Office for the Southern District of New York (the “SDNY”) informed BioReference that it believes that, from 2006 to the present, BioReference had, in violation of the False Claims Act, improperly billed Medicare and TRICARE (both are federal government healthcare programs) for clinical laboratory services provided to hospital inpatient beneficiaries at certain hospitals. BioReference is reviewing and assessing the allegations made by the SDNY, and, at this point, BioReference has not determined whether there is any merit to the SDNY’s claims nor can it determine the extent of any potential liability. While management cannot predict the outcome of these matters at this time, the ultimate outcome could be material to our business, financial condition, results of operations, and cash flows.
We are a party to other litigation in the ordinary course of business. We do not believe that any such litigation will have a material adverse effect on our business, financial condition, results of operations or cash flows.
We expect to continue to incur substantial research and development expenses, including expenses related to the hiring of personnel and additional clinical trials. We expect that selling, general and administrative expenses will also increase as we expand our sales, marketing and administrative staff and add infrastructure, particularly as it relates to the launch of Rayaldee. We do not anticipate that we will generate substantial revenue from the sale of proprietary pharmaceutical products or certain of our diagnostic products for some time and we have generated only limited revenue from our pharmaceutical operations in Chile, Mexico, Israel, Spain, and Ireland, and from sale of the 4Kscore test. If we acquire additional assets or companies, fail to generate expected cash flow from BioReference, accelerate our product development programs or initiate additional clinical trials, we will need additional funds. If we are not able to secure additional funding when needed, we may have to delay, reduce the scope of, or eliminate one or more of our clinical trials or research and development programs or possible acquisitions.
We have employment agreements with certain executives of BioReference which provide for compensation and certain other benefits and for severance payments under certain circumstances. During the years ended December 31, 2018, 2017 and 2016, we recognized $4.9 million, $5.8 million and $17.9 million, respectively, of severance costs pursuant to these employment agreements as a component of Selling, general and administrative expense.
Note 14 Revenue Recognition
Effective January 1, 2018, we adopted Accounting Standards Codification Topic 606, Revenue from Contracts with Customers. We generate revenues from services, products and intellectual property as follows:
Revenue from services
Revenue for laboratory services is recognized at the time test results are reported, which approximates when services are provided and the performance obligations are satisfied. Services are provided to patients covered by various third-party payor programs including various managed care organizations, as well as the Medicare and Medicaid programs. Billings for services are included in revenue net of allowances for contractual discounts, allowances for differences between the amounts billed and estimated program payment amounts, and implicit price concessions provided to uninsured patients which are all elements of variable consideration.
130
The following are descriptions of our payors for laboratory services:
Healthcare Insurers. Reimbursements from healthcare insurers are based on negotiated fee-for-service schedules. Revenues consist of amounts billed, net of contractual allowances for differences between amounts billed and the estimated consideration we expect to receive from such payors, which considers historical denial and collection experience and the terms of our contractual arrangements. Adjustments to the allowances, based on actual receipts from the third-party payors, are recorded upon settlement.
Government Payors. Reimbursements from government payors are based on fee-for-service schedules set by governmental authorities, including traditional Medicare and Medicaid. Revenues consist of amounts billed, net of contractual allowances for differences between amounts billed and the estimated consideration we expect to receive from such payors, which considers historical denial and collection experience and the terms of our contractual arrangements. Adjustments to the allowances, based on actual receipts from the government payors, are recorded upon settlement.
Client Payors. Client payors include physicians, hospitals, employers, and other institutions for which services are performed on a wholesale basis, and are billed and recognized as revenue based on negotiated fee schedules.
Patients. Uninsured patients are billed based on established patient fee schedules or fees negotiated with physicians on behalf of their patients. Insured patients (including amounts for coinsurance and deductible responsibilities) are billed based on fees negotiated with healthcare insurers. Collection of billings from patients is subject to credit risk and ability of the patients to pay. Revenues consist of amounts billed net of discounts provided to uninsured patients in accordance with our policies and implicit price concessions. Implicit price concessions represent differences between amounts billed and the estimated consideration that we expect to receive from patients, which considers historical collection experience and other factors including current market conditions. Adjustments to the estimated allowances, based on actual receipts from the patients, are recorded upon settlement.
The complexities and ambiguities of billing, reimbursement regulations and claims processing, as well as issues unique to Medicare and Medicaid programs, require us to estimate the potential for retroactive adjustments as an element of variable consideration in the recognition of revenue in the period the related services are rendered. Actual amounts are adjusted in the period those adjustments become known. For the years ended December 31, 2018 and 2017, revenue reductions due to changes in estimates of implicit price concessions for performance obligations satisfied in prior periods of $22.8 million and $66.0 million, respectively, were recognized. No material revenue reductions due to changes in estimates of implicit price concessions for performance obligations satisfied in prior periods were recognized during the year ended December 31, 2016.
Third-party payors, including government programs, may decide to deny payment or recoup payments for testing they contend were improperly billed or not medically necessary, against their coverage determinations, or for which they believe they have otherwise overpaid (including as a result of their own error), and we may be required to refund payments already received. Our revenues may be subject to retroactive adjustment as a result of these factors among others, including without limitation, differing interpretations of billing and coding guidance and changes by government agencies and payors in interpretations, requirements, and “conditions of participation” in various programs. We have processed requests for recoupment from third-party payors in the ordinary course of our business, and it is likely that we will continue to do so in the future. If a third-party payer denies payment for testing or recoups money from us in a later period, reimbursement revenue for our testing could decline.
As an integral part of our billing compliance program, we periodically assess our billing and coding practices, respond to payor audits on a routine basis, and investigate reported failures or suspected failures to comply with federal and state healthcare reimbursement requirements, as well as overpayment claims which may arise from time to time without fault on the part of the Company. We may have an obligation to reimburse Medicare, Medicaid, and third-party payors for overpayments regardless of fault. We have periodically identified and reported overpayments, reimbursed payors for overpayments and taken appropriate corrective action.
Settlements with third-party payors for retroactive adjustments due to audits, reviews or investigations are also considered variable consideration and are included in the determination of the estimated transaction price for providing services. These settlements are estimated based on the terms of the payment agreement with the payor, correspondence from the payor and our historical settlement activity, including an assessment of the probability a significant reversal of cumulative revenue recognized will occur when the uncertainty is subsequently resolved. Estimated settlements are adjusted in future periods as adjustments become known (that is, new information becomes available), or as years are settled or are no longer subject to such audits, reviews, and investigations.
During 2017, a payor informed us it had overpaid BioReference due to an error on its part over a period of approximately ten years, including multiple years prior to the acquisition of BioReference by OPKO in August 2015. As of December 31,
131
2018 and 2017, we have liabilities of approximately $35.9 million and $30.0 million within Accrued expenses and Other long-term liabilities related to reimbursements for payor overpayments.
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Healthcare insurers |
$ |
$ |
$ |
||||||||
Government payors |
|||||||||||
Client payors |
|||||||||||
Patients |
|||||||||||
Total |
$ |
$ |
$ |
||||||||
Revenue from products
We recognize revenue from product sales when a customer obtains control of promised goods or services. The amount of revenue that is recorded reflects the consideration that we expect to receive in exchange for those goods or services. Our estimates for sales returns and allowances are based upon the historical patterns of product returns and allowances taken, matched against the sales from which they originated, and our evaluation of specific factors that may increase or decrease the risk of product returns. Product revenues are recorded net of estimated rebates, chargebacks, discounts, co-pay assistance and other deductions (collectively, “Sales Deductions”) as well as estimated product returns which are all elements of variable consideration. Allowances are recorded as a reduction of revenue at the time product revenues are recognized. The actual amounts of consideration ultimately received may differ from our estimates. If actual results in the future vary from our estimates, we will adjust these estimates, which would affect Revenue from products in the period such variances become known.
We launched Rayaldee in the U.S. through our dedicated renal sales force in November 2016. Rayaldee is distributed in the U.S. principally through the retail pharmacy channel, which initiates with the largest wholesalers in the U.S. (collectively, “Rayaldee Customers”). In addition to distribution agreements with Rayaldee Customers, we have entered into arrangements with many healthcare providers and payors that provide for government-mandated and/or privately-negotiated rebates, chargebacks and discounts with respect to the purchase of Rayaldee.
We recognize revenue for shipments of Rayaldee at the time of delivery to customers after estimating Sales Deductions and product returns as elements of variable consideration utilizing historical information and market research projections. For the years ended December 31, 2018, and 2017, we recognized $20.3 million and $9.1 million in net product revenue from sales of Rayaldee.
(In thousands) |
Chargebacks, discounts, rebates and fees |
Governmental |
Returns |
Total |
||||||||||||
Balance at December 31, 2017 |
$ |
$ |
$ |
$ |
||||||||||||
Provision related to current period sales |
||||||||||||||||
Credits or payments made |
( |
) |
( |
) |
( |
) |
( |
) |
||||||||
Balance at December 31, 2018 |
$ |
$ |
$ |
$ |
||||||||||||
Total gross Rayaldee sales
|
$ |
|||||||||||||||
Provision for Rayaldee sales allowances and accruals as a percentage of gross Rayaldee sales
|
% |
|||||||||||||||
Taxes collected from customers related to revenues from services and revenues from products are excluded from revenues.
Revenue from intellectual property
132
We recognize revenues from the transfer of intellectual property generated through license, development, collaboration and/or commercialization agreements. The terms of these agreements typically include payment to us for one or more of the following: non-refundable, up-front license fees; development and commercialization milestone payments; funding of research and/or development activities; and royalties on sales of licensed products. Revenue is recognized upon satisfaction of a performance obligation by transferring control of a good or service to the customer.
For research, development and/or commercialization agreements that result in revenues, we identify all material performance obligations, which may include a license to intellectual property and know-how, and research and development activities. In order to determine the transaction price, in addition to any upfront payment, we estimate the amount of variable consideration at the outset of the contract either utilizing the expected value or most likely amount method, depending on the facts and circumstances relative to the contract. We constrain (reduce) our estimates of variable consideration such that it is probable that a significant reversal of previously recognized revenue will not occur throughout the life of the contract. When determining if variable consideration should be constrained, we consider whether there are factors outside of our control that could result in a significant reversal of revenue. In making these assessments, we consider the likelihood and magnitude of a potential reversal of revenue. These estimates are re-assessed each reporting period as required.
Upfront License Fees: If a license to our intellectual property is determined to be functional intellectual property distinct from the other performance obligations identified in the arrangement, we recognize revenue from nonrefundable, upfront license fees based on the relative value prescribed to the license compared to the total value of the arrangement. The revenue is recognized when the license is transferred to the customer and the customer is able to use and benefit from the license. For licenses that are not distinct from other obligations identified in the arrangement, we utilize judgment to assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time. If the combined performance obligation is satisfied over time, we apply an appropriate method of measuring progress for purposes of recognizing revenue from nonrefundable, upfront license fees. We evaluate the measure of progress each reporting period and, if necessary, adjust the measure of performance and related revenue recognition.
Development and Regulatory Milestone Payments: Depending on facts and circumstances, we may conclude that it is appropriate to include the milestone in the estimated transaction price or that it is appropriate to fully constrain the milestone. A milestone payment is included in the transaction price in the reporting period that we conclude that it is probable that recording revenue in the period will not result in a significant reversal in amounts recognized in future periods. We may record revenues from certain milestones in a reporting period before the milestone is achieved if we conclude that achievement of the milestone is probable and that recognition of revenue related to the milestone will not result in a significant reversal in amounts recognized in future periods. We record a corresponding contract asset when this conclusion is reached. Milestone payments that have been fully constrained are not included in the transaction price to date. These milestones remain fully constrained until we conclude that achievement of the milestone is probable and that recognition of revenue related to the milestone will not result in a significant reversal in amounts recognized in future periods. We re-evaluate the probability of achievement of such development milestones and any related constraint each reporting period. We adjust our estimate of the overall transaction price, including the amount of revenue recorded, if necessary.
Research and Development Activities: If we are entitled to reimbursement from our customers for specified research and development expenses, we account for them as separate performance obligations if distinct. We also determine whether the research and development funding would result in revenues or an offset to research and development expenses in accordance with provisions of gross or net revenue presentation. The corresponding revenues or offset to research and development expenses are recognized as the related performance obligations are satisfied.
Sales-based Milestone and Royalty Payments: Our customers may be required to pay us sales-based milestone payments or royalties on future sales of commercial products. We recognize revenues related to sales-based milestone and royalty payments upon the later to occur of (i) achievement of the customer’s underlying sales or (ii) satisfaction of any performance obligation(s) related to these sales, in each case assuming the license to our intellectual property is deemed to be the predominant item to which the sales-based milestones and/or royalties relate.
Other Potential Products and Services: Arrangements may include an option for license rights, future supply of drug substance or drug product for either clinical development or commercial supply at the licensee’s election. We assess if these options provide a material right to the licensee and if so, they are accounted for as separate performance obligations at the inception of the contract and revenue is recognized only if the option is exercised and products or services are subsequently delivered or when the rights expire. If the promise is based on market terms and not considered a material right, the option is accounted for if and when exercised. If we are entitled to additional payments when the licensee exercises these options, any additional payments are generally recorded in license or other revenues when the licensee obtains control of the goods, which is upon delivery.
133
Note 15 Strategic Alliances
Japan Tobacco Inc.
On October 12, 2017, EirGen, our wholly-owned subsidiary, and Japan Tobacco Inc. (“JT”) entered into a Development and License Agreement (the “JT Agreement”) granting JT the exclusive rights for the development and commercialization of Rayaldee in Japan (the “JT Territory”). The license grant to JT covers the therapeutic and preventative use of the product for (i) SHPT in non-dialysis and dialysis patients with CKD, (ii) rickets, and (iii) osteomalacia (the “JT Initial Indications”), as well as such additional indications as may be added to the scope of the license subject to the terms of the JT Agreement (the JT Additional Indications” and together with the JT Initial Indications, the “JT Field”).
In connection with the license, OPKO received an initial upfront payment of $6 million and received another $6 million upon the initiation of OPKO’s phase 2 study for Rayaldee in dialysis patients in the U.S. in September 2018. OPKO is also eligible to receive up to an additional aggregate amount of $31 million upon the achievement of certain regulatory and development milestones by JT for Rayaldee in the JT Territory, and $75 million upon the achievement of certain sales based milestones by JT in the JT Territory. OPKO will also receive tiered, double digit royalty payments at rates ranging from low double digits to mid-teens on net sales of Rayaldee within the JT Territory. JT will, at its sole cost and expense, be responsible for performing all development activities necessary to obtain all regulatory approvals for Rayaldee in Japan and for all commercial activities pertaining to Rayaldee in Japan.
The JT Agreement provides for the following: (1) an exclusive license in the JT Territory in the JT Field for the development and commercialization of Rayaldee; and (2) at JT’s option, EirGen will supply products to support the development, sale and commercialization of the products to JT in the JT Territory.
The initial consideration primarily includes the non-refundable $6 million upfront payment and the $6 million we received upon the initiation of our phase 2 study for Rayaldee in dialysis patients in the U.S. The initial consideration will be recognized over the performance period through 2021, when we anticipate completing the transfer of license materials specified in the JT Agreement and our performance obligation is complete.
We are also eligible to receive up to $31 million in regulatory and development milestones and $75 million in sales milestones. Payments received for regulatory, development and sales milestones are non-refundable. The milestones are payable if and when the associated milestone is achieved and will be recognized as revenue in the period in which the associated milestone is achieved, assuming all other revenue recognition criteria are met. To date, no revenue has been recognized related to these milestones.
Vifor Fresenius Medical Care Renal Pharma Ltd
In May 2016, EirGen, our wholly-owned subsidiary, and Vifor Fresenius Medical Care Renal Pharma Ltd (“VFMCRP”), entered into a Development and License Agreement (the “VFMCRP Agreement”) for the development and commercialization of Rayaldee (the “Product”) worldwide, except for (i) the U.S., (ii) any country in Central America or South America (excluding Mexico), (iii) Russia, (iv) China, (v) Japan, (vi) Ukraine, (vii) Belorussia, (viii) Azerbaijan, (ix) Kazakhstan, and (x) Taiwan (the “VFMCRP Territory”). The license to VFMCRP potentially covers all therapeutic and prophylactic uses of the Product in human patients (the “VFMCRP Field”), provided that initially the license is for the use of the Product for the treatment or prevention of SHPT related to patients with stage 3 or 4 CKD and vitamin D insufficiency/deficiency (the “VFMCRP Initial Indication”).
Under the terms of the VFMCRP Agreement, EirGen granted to VFMCRP an exclusive license in the VFMCRP Territory in the VFMCRP Field to use certain EirGen patents and technology to make, have made, use, sell, offer for sale, and import Products and to develop, commercialize, have commercialized, and otherwise exploit the Product. EirGen received a non-refundable and non-creditable initial payment of $50 million, which was recognized in Revenue from the transfer of intellectual
134
property and other in our Consolidated Statement of Operations in 2016. EirGen also received a $2.0 million payment triggered by the approval of Rayaldee in Canada for the treatment of SHPT in adults with stage 3 or 4 CKD and vitamin D insufficiency in July 2018. EirGen is also eligible to receive up to an additional $35 million in regulatory milestones (“Regulatory Milestones”) and $195 million in launch and sales-based milestones (“Sales Milestones”), and will receive tiered royalties on sales of the product at percentage rates that range from the mid-teens to the mid-twenties or a minimum royalty, whichever is greater, upon the commencement of sales of the Product within the VFMCRP Territory and in the VFMCRP Field.
We plan to share responsibility with VFMCRP for the conduct of trials specified within an agreed-upon development plan, with each company leading certain activities within the plan. EirGen will lead the manufacturing activities within and outside the VFMCRP Territory and the commercialization activities outside the VFMCRP Territory and outside the VFMCRP Field in the VFMCRP Territory and VFMCRP will lead the commercialization activities in the VFMCRP Territory and the VFMCRP Field. For the initial development plan, the companies have agreed to certain cost sharing arrangements. VFMCRP will be responsible for all other development costs that VFMCRP considers necessary to develop the Product for the use of the Product for the VFMCRP Initial Indication in the VFMCRP Territory in the VFMCRP Field except as otherwise provided in the VFMCRP Agreement. The first of the clinical studies provided for in the development activities commenced in September 2018.
In connection with the VFMCRP Agreement, the parties entered into a letter agreement pursuant to which EirGen granted to VFMCRP an exclusive option (the “Option”) to acquire an exclusive license under certain EirGen patents and technology to use, import, offer for sale, sell, distribute and commercialize the Product in the U.S. solely for the treatment of SHPT in dialysis patients with CKD and vitamin D insufficiency (the “Dialysis Indication”). Upon exercise of the Option, VFMCRP will reimburse EirGen for all of the development costs incurred by EirGen with respect to the Product for the Dialysis Indication in the U.S. VFMCRP would also pay EirGen up to an additional aggregate amount of $555 million of sales-based milestones upon the achievement of certain milestones and would be obligated to pay royalties at percentage rates that range from the mid-teens to the mid-twenties on sales of the Product in the U.S. for the Dialysis Indication. To date, VFMCRP has not exercised its option.
Payments received for Regulatory Milestones and Sales Milestones are non-refundable. The Regulatory Milestones are payable if and when VFMCRP obtains approval from certain regulatory authorities and will be recognized as revenue in the period in which the associated milestone is achieved, assuming all other revenue recognition criteria are met. We account for the Sales Milestones as royalties and Sales Milestones payments will be recognized as revenue in the period in which the associated milestone is achieved or sales occur, assuming all other revenue recognition criteria are met.
Pfizer Inc.
In December 2014, we entered into an exclusive worldwide agreement with Pfizer Inc. (“Pfizer”) for the development and commercialization of our long-acting hGH-CTP for the treatment of growth hormone deficiency (“GHD”) in adults and children, as well as for the treatment of growth failure in children born small for gestational age (“SGA”) (the “Pfizer Transaction”).
The Pfizer Transaction closed in January 2015 following the termination of the waiting period under the Hart-Scott-Rodino Act. Under the terms of the Pfizer Transaction, we received non-refundable and non-creditable upfront payments of $295.0 million and are eligible to receive up to an additional $275.0 million upon the achievement of certain regulatory milestones. Pfizer received the exclusive license to commercialize hGH-CTP worldwide. In addition, we are eligible to receive initial tiered royalty payments associated with the commercialization of hGH-CTP for Adult GHD with percentage rates ranging from the high teens to mid-twenties. Upon the launch of hGH-CTP for Pediatric GHD in certain major markets, the royalties will transition to regional, tiered gross profit sharing for both hGH-CTP and Pfizer’s Genotropin®.
The agreement with Pfizer will remain in effect until the last sale of the licensed product, unless earlier terminated as permitted under the agreement. In addition to termination rights for material breach and bankruptcy, Pfizer is permitted to terminate the Agreement in its entirety, or with respect to one or more world regions, without cause after a specified notice period. If the Agreement is terminated by us for Pfizer’s uncured material breach, or by Pfizer without cause, provision has been made for transition of product and product responsibilities to us for the terminated regions, as well as continued supply of product by Pfizer or transfer of supply to us in order to support the terminated regions.
We are recognizing the non-refundable $295.0 million upfront payments as the research and development services are completed and had contract liabilities related to the Pfizer Transactions of $83.1 million at December 31, 2018, of which $60.6 million was classified in Accrued expenses and $22.5 million was classified in Other long-term liabilities.
The Pfizer Transaction includes milestone payments of $275.0 million upon the achievement of certain milestones. The milestones range from $20.0 million to $90.0 million each and are based on achievement of regulatory approval in the U.S. and
135
regulatory approval and price approval in other major markets. The milestone payments will be recognized as revenue in the period in which the associated milestone is achieved, assuming all other revenue recognition criteria are met. To date, no revenue has been recognized related to the achievement of the milestones.
TESARO
In November 2009, we entered into an asset purchase agreement (the “NK-1 Agreement”) under which we acquired VARUBI™ (rolapitant) and other neurokinin-1 (“NK-1”) assets from Merck. In December 2010, we entered into an exclusive license agreement with TESARO, in which we out-licensed the development, manufacture, commercialization and distribution of our lead NK-1 candidate, VARUBI™ (the “TESARO License”). Under the terms of the license, we received a $6.0 million upfront payment from TESARO and we received $30.0 million of milestone payments from TESARO upon achievement of certain regulatory and commercial sale milestones and we are eligible to receive additional commercial milestone payments of up to $85.0 million if specified levels of annual net sales are achieved. The sales based milestone payments will be recognized as revenue in full in the period in which the associated sales occur. During year ended December 31, 2017, $10.0 million of revenue was recognized related to the achievement of the milestones under the TESARO License. During the years ended December 31, 2018 and 2016, no revenue was recognized related to the achievement of the milestones under the TESARO License.
TESARO is also obligated to pay us tiered royalties on annual net sales achieved in the U.S. and Europe at percentage rates that range from the low double digits to the low twenties, and outside of the U.S. and Europe at low double-digit percentage rates. Royalties will be recognized in the period the sales occur. TESARO assumed responsibility for clinical development and commercialization of licensed products at its expense. Under the NK-1 Agreement, we will continue to receive royalties on a country-by-country and product-by-product basis until the later of the date that all of the patent rights licensed from us and covering VARUBI™ expire, are invalidated or are not enforceable and 12 years from the first commercial sale of the product.
If TESARO elects to develop and commercialize VARUBI™ in Japan through a third-party licensee, TESARO will share equally with us all amounts it receives in connection with such activities, subject to certain exceptions and deductions.
The term of the license will remain in force until the expiration of the royalty term in each country, unless we terminate the license earlier for TESARO’s material breach of the license or bankruptcy. TESARO has a right to terminate the license at any time during the term for any reason on three months’ written notice. TESARO announced during the first quarter of 2018 that it has elected to suspend further distribution of Varubi IV.
TESARO assigned its rights and obligations under the agreement to TerSera Therapeutics LLC (“TerSera”) in June 2018 pursuant to an asset purchase agreement. Under the asset purchase agreement, TerSera is responsible for VARUBI in the U.S. and Canada and TESARO can continue to commercialize VARUBY® in Europe and the rest of the world though a sublicense with TerSera.
Pharmsynthez
In April 2013, we entered into a series of concurrent transactions with Pharmsynthez, a Russian pharmaceutical company traded on the Moscow Stock Exchange pursuant to which we acquired an equity method investment in Pharmsynthez (ownership 9 %). We also granted rights to certain technologies in the Russian Federation, Ukraine, Belarus, Azerbaijan and Kazakhstan (the “Pharmsynthez Territories”) to Pharmsynthez and agreed to perform certain development activities. We will receive from Pharmsynthez royalties on net sales of products incorporating the technologies in the Pharmsynthez Territories, as well as a percentage of any sublicense income from third parties for the technologies in the Pharmsynthez Territories.
RXi Pharmaceuticals Corporation
In March 2013, we completed the sale to RXi (now known as Phio Pharmaceuticals Corp.) of substantially all of our assets in the field of RNA interference (the “RNAi Assets”) (collectively, the “Asset Purchase Agreement”). Pursuant to the Asset Purchase Agreement, RXi will be required to pay us up to $50.0 million in milestone payments upon the successful development and commercialization of each drug developed by RXi, certain of its affiliates or any of its or their licensees or sublicensees utilizing patents included within the RNAi Assets (each, a “Qualified Drug”). In addition, RXi will also be required to pay us royalties equal to: (a) a mid single-digit percentage of “Net Sales” (as defined in the Asset Purchase Agreement) with respect to each Qualified Drug sold for an ophthalmologic use during the applicable “Royalty Period” (as defined in the Asset Purchase Agreement); and (b) a low single-digit percentage of net sales with respect to each Qualified Drug sold for a non-ophthalmologic use during the applicable Royalty Period.
136
Other
137
Note 16 Leases
Operating leases
We conduct certain of our operations under operating lease agreements. Rent expense under operating leases was approximately $18.9 million, $18.9 million, and $18.8 million for the years ended December 31, 2018, 2017, and 2016, respectively.
Year Ending |
(In thousands) |
||
2019 |
$ |
||
2020 |
|||
2021 |
|||
2022 |
|||
2023 |
|||
Thereafter |
|||
Total minimum operating lease commitments |
$ |
||
Capital leases
Capital leases |
Year ended December 31, 2018 |
||
Automobiles |
$ |
||
Less: Accumulated Depreciation |
( |
) |
|
Net capital leases in Property, plant and equipment |
$ |
||
Year Ending |
(In thousands) |
||
2019 |
$ |
||
2020 |
|||
2021 |
|||
2022 |
|||
2023 |
|||
Total minimum capital lease commitments |
|||
Less: Amounts representing interest |
|||
Net capital liability |
$ |
||
Current |
$ |
||
Long-term |
$ |
||
138
Note 17 Segments
We manage our operations in two reportable segments, pharmaceutical and diagnostics. The pharmaceutical segment consists of our pharmaceutical operations we acquired in Chile, Mexico, Ireland, Israel and Spain, Rayaldee product sales and our pharmaceutical research and development. The diagnostics segment primarily consists of our clinical laboratory operations we acquired through the acquisition of BioReference and our point-of-care operations. There are no significant inter-segment sales. We evaluate the performance of each segment based on operating profit or loss. There is no inter-segment allocation of interest expense and income taxes.
139
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Revenue from services: |
|||||||||||
Pharmaceutical |
$ |
$ |
$ |
||||||||
Diagnostics |
|||||||||||
Corporate |
|||||||||||
$ |
$ |
$ |
|||||||||
Revenue from products: |
|||||||||||
Pharmaceutical |
$ |
$ |
$ |
||||||||
Diagnostics |
— |
— |
— |
||||||||
Corporate |
— |
— |
— |
||||||||
$ |
$ |
$ |
|||||||||
Revenue from transfer of intellectual property and other: |
|||||||||||
Pharmaceutical |
$ |
$ |
$ |
||||||||
Diagnostics |
|||||||||||
Corporate |
|||||||||||
$ |
$ |
$ |
|||||||||
Operating loss: |
|||||||||||
Pharmaceutical |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Diagnostics |
( |
) |
( |
) |
( |
) |
|||||
Corporate |
( |
) |
( |
) |
( |
) |
|||||
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
|||
Depreciation and amortization: |
|||||||||||
Pharmaceutical |
$ |
$ |
$ |
||||||||
Diagnostics |
|||||||||||
Corporate |
|||||||||||
$ |
$ |
$ |
|||||||||
Income (loss) from investment in investees: |
|||||||||||
Pharmaceutical |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
||
Diagnostics |
( |
) |
( |
) |
|||||||
Corporate |
|||||||||||
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
|||
Revenues: |
|||||||||||
United States |
$ |
$ |
$ |
||||||||
Ireland |
|||||||||||
Chile |
|||||||||||
Spain |
|||||||||||
Israel |
|||||||||||
Mexico |
|||||||||||
Other |
|||||||||||
$ |
$ |
$ |
|||||||||
140
(In thousands) |
December 31, 2018 |
December 31, 2017 |
|||||
Assets: |
|||||||
Pharmaceutical |
$ |
$ |
|||||
Diagnostics |
|||||||
Corporate |
|||||||
$ |
$ |
||||||
Goodwill: |
|||||||
Pharmaceutical |
$ |
$ |
|||||
Diagnostics |
|||||||
Corporate |
|||||||
$ |
$ |
||||||
No customer represented more than 10% of our total consolidated revenue during the years ended December 31, 2018, 2017 and 2016. As of December 31, 2018 and 2017, no customer represented more than 10% of our accounts receivable balance.
(In thousands) |
December 31, 2018 |
December 31, 2017 |
|||||
PP&E: |
|||||||
U.S. |
$ |
$ |
|||||
Foreign |
|||||||
Total |
$ |
$ |
|||||
Note 18 Fair Value Measurements
We record fair values at an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement determined based on assumptions that market participants would use in pricing an asset or liability. We utilize a three-tier fair value hierarchy, which prioritizes the inputs used in measuring fair value. These tiers include: Level 1, defined as observable inputs such as quoted prices in active markets; Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions.
As of December 31, 2018, we have equity securities (refer to Note 4), forward foreign currency exchange contracts for inventory purchases (refer to Note 19) and contingent consideration related to the acquisitions of CURNA, OPKO Diagnostics and OPKO Renal that are required to be measured at fair value on a recurring basis. In addition, in connection with our investment and our consulting agreement with BioCardia, we record the related BioCardia options at fair value as well as the warrants from COCP, InCellDx, Inc., Xenetic, Phio and Neovasc.
141
Fair value measurements as of December 31, 2018 |
|||||||||||||||
(In thousands) |
Quoted
prices in
active
markets for
identical
assets
(Level 1)
|
Significant
other
observable
inputs
(Level 2)
|
Significant
unobservable
inputs
(Level 3)
|
Total |
|||||||||||
Assets: |
|||||||||||||||
Money market funds |
$ |
$ |
$ |
$ |
|||||||||||
Equity securities |
|||||||||||||||
Common stock options/warrants |
|||||||||||||||
Forward contracts |
|||||||||||||||
Total assets |
$ |
$ |
$ |
$ |
|||||||||||
Liabilities: |
|||||||||||||||
Contingent consideration: |
$ |
$ |
$ |
||||||||||||
Total liabilities |
$ |
$ |
$ |
$ |
|||||||||||
Fair value measurements as of December 31, 2017 |
|||||||||||||||
(In thousands) |
Quoted
prices in
active
markets for
identical
assets
(Level 1)
|
Significant
other
observable
inputs
(Level 2)
|
Significant
unobservable
inputs
(Level 3)
|
Total |
|||||||||||
Assets: |
|||||||||||||||
Money market funds |
$ |
$ |
$ |
$ |
|||||||||||
Equity securities |
|||||||||||||||
Common stock options/warrants |
|||||||||||||||
Total assets |
$ |
$ |
$ |
$ |
|||||||||||
Liabilities: |
|||||||||||||||
Forward contracts |
$ |
$ |
$ |
$ |
|||||||||||
Contingent consideration: |
|||||||||||||||
Total liabilities |
$ |
$ |
$ |
$ |
|||||||||||
There have been no transfers between Level 1 and Level 2 and no transfers to or from Level 3 of the fair value hierarchy.
As of December 31, 2018 and 2017, the carrying value of our other financial instrument assets approximates their fair value due to their short-term nature or variable rate of interest.
December 31, 2018 |
|||
(In thousands) |
Contingent
consideration
|
||
Balance at December 31, 2017 |
$ |
||
Total losses (gains) for the period: |
|||
Included in results of operations |
( |
) |
|
Balance at December 31, 2018 |
$ |
||
142
December 31, 2017 |
|||||||
(In thousands) |
Contingent consideration |
Embedded conversion option |
|||||
Balance at December 31, 2016 |
$ |
$ |
|||||
Total losses (gains) for the period: |
|||||||
Included in results of operations |
( |
) |
( |
) |
|||
Foreign currency impact |
|||||||
Payments |
( |
) |
|||||
Reclassification of embedded derivatives to equity |
( |
) |
|||||
Balance at December 31, 2017 |
$ |
$ |
|||||
The estimated fair values of our financial instruments have been determined by using available market information and what we believe to be appropriate valuation methodologies. We use the following methods and assumptions in estimating fair value:
143
Note 19 Derivative Contracts
(In thousands) |
Balance Sheet Component |
December 31, 2018 |
December 31, 2017 |
||||||
Derivative financial instruments: |
|||||||||
Common stock options/warrants |
Investments, net |
$ |
$ |
||||||
Forward contracts |
Unrealized gains on forward contracts are recorded in Other current assets and prepaid expenses. Unrealized (losses) on forward contracts are recorded in Accrued expenses. |
$ |
$ |
( |
) |
||||
We enter into foreign currency forward exchange contracts to cover the risk of exposure to exchange rate differences arising from inventory purchases on letters of credit. Under these forward contracts, for any rate above or below the fixed rate, we receive or pay the difference between the spot rate and the fixed rate for the given amount at the settlement date.
For the years ended December 31, |
|||||||||||
(In thousands) |
2018 |
2017 |
2016 |
||||||||
Derivative gain (loss): |
|||||||||||
Common stock options/warrants |
$ |
$ |
( |
) |
$ |
( |
) |
||||
2033 Senior Notes |
|||||||||||
Forward contracts |
$ |
$ |
( |
) |
$ |
||||||
Total |
$ |
$ |
$ |
||||||||
144
Note 20 Selected Quarterly Financial Data (Unaudited)
For the 2018 Quarters Ended |
|||||||||||||||
(In thousands, except per share data) |
March 31 |
June 30 |
September 30 |
December 31 |
|||||||||||
Total revenues |
$ |
$ |
$ |
$ |
|||||||||||
Total costs and expenses |
|||||||||||||||
Net income (loss) |
( |
) |
( |
) |
( |
) |
( |
) |
|||||||
Earnings (loss) per share, basic and diluted |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
|||
For the 2017 Quarters Ended |
|||||||||||||||
(In thousands, except per share data) |
March 31 |
June 30 |
September 30 |
December 31 |
|||||||||||
Total revenues |
$ |
$ |
$ |
$ |
|||||||||||
Total costs and expenses |
|||||||||||||||
Net income (loss) |
( |
) |
( |
) |
( |
) |
( |
) |
|||||||
Earnings (loss) per share, basic |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
|||
Earnings (loss) per share, diluted |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
$ |
( |
) |
|||
Note 21 Subsequent Events
In February 2019, we issued $200.0 million aggregate principal amount of Convertible Senior Notes due 2025 (the “2025 Notes”) in an underwritten public offering. The 2025 Notes will bear interest at a rate of 4.50 % per year, payable semiannually in arrears on February 15 and August 15 of each year, beginning on August 15, 2019. The notes mature on February 15, 2025, unless earlier repurchased, redeemed or converted.
Holders may convert their 2025 Notes at their option at any time prior to the close of business on the business day immediately preceding November 15, 2024 only under the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2019 (and only during such calendar quarter), if the last reported sale price of our common stock for at least 20 trading days (whether or not consecutive) during a period of 30 consecutive trading days ending on the last trading day of the immediately preceding calendar quarter is greater than or equal to 130 % of the conversion price on each applicable trading day; (2) during the five business day period after any five consecutive trading day period (the “measurement period”) in which the trading price per $1,000 principal amount of 2025 Notes for each trading day of the measurement period was less than 98 % of the product of the last reported sale price of our common stock and the conversion rate on each such trading day; (3) if we call any or all of the 2025 Notes for redemption, at any time prior to the close of business on the scheduled trading day immediately preceding the redemption date; or (4) upon the occurrence of specified corporate events. On or after November 15, 2024, until the close of business on the business day immediately preceding the maturity date, holders of the 2025 Notes may convert their notes at any time, regardless of the foregoing circumstances. Upon conversion, we will pay or deliver, as the case may be, cash, shares of our common stock, or a combination of cash and shares of our common stock, at our election.
The conversion rate for the notes will initially be 236.7424 shares of common stock per $1,000 principal amount of 2025 Notes (equivalent to an initial conversion price of approximately $4.22 per share of common stock). The conversion rate for the 2025 Notes will be subject to adjustment in some events but will not be adjusted for any accrued and unpaid interest. In addition, following certain corporate events that occur prior to the maturity date of the notes or if we deliver a notice of redemption, in certain circumstances we will increase the conversion rate of the 2025 Notes for a holder who elects to convert its notes in connection with such a corporate event or notice of redemption as the case may be.
We may not redeem the 2025 Notes prior to February 15, 2022. We may redeem for cash any or all of the notes, at our option, on or after February 15, 2022, if the last reported sale price of our common stock has been at least 130 % of the conversion price for the notes then in effect for at least 20 trading days (whether or not consecutive) during any 30 consecutive trading day period (including the last trading day of such period) ending on, and including, the trading day immediately preceding the date on which we provide notice of redemption at a redemption price equal to 100 % of the principal amount of the notes to be redeemed, plus accrued and unpaid interest to, but excluding, the redemption date. No sinking fund is provided for the 2025 Notes.
145
If we undergo a fundamental change prior to the maturity date of the 2025 Notes, holders may require us to repurchase for cash all or any portion of their notes at a repurchase price equal to 100 % of the principal amount of the notes to be repurchased, plus accrued and unpaid interest to, but excluding, the fundamental change repurchase date. The 2025 Notes will be our senior unsecured obligations and will rank senior in right of payment to any of our indebtedness that is expressly subordinated in right of payment to the 2025 Notes; equal in right of payment to any of our existing and future liabilities that are not so subordinated; effectively junior in right of payment to any of our secured indebtedness to the extent of the value of the assets securing such indebtedness; and structurally junior to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries.
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. The credit agreement was terminated on or around February 20, 2019 and amounts borrowed during 2019 were repaid from the proceeds of the 2025 Convertible Notes offering.
On February 1, 2019, approximately $28.8 million aggregate principal amount of 2033 Senior Notes were tendered by holders pursuant to such holders’ option to require us to repurchase the 2033 Senior Notes. Holders of the 2033 Senior Notes issued in January 2013 may require us to repurchase the 2033 Senior Notes for 100 % of their principal amount, plus accrued and unpaid interest, again on February 1, 2023 and February 1, 2028, or following the occurrence of a fundamental change as defined in the indenture governing the 2033 Senior Notes.
On February 26, 2019, BioReference and certain of its subsidiaries entered into Amendment No. 8 to the Credit Agreement, which amended the Credit Agreement to add back certain cost savings resulting from work force reductions in the 2018 fiscal year to the calculation of EBITDA for purposes of complying with the minimum fixed charge coverage ratio covenant. The other terms of the Credit Agreement remain unchanged.
In December 2018, we and Dr. Frost entered into settlements with the SEC, which, upon approval by the court in January 2019, resolved the claims against us and Dr. Frost raised in the SEC complaint. Pursuant to the settlement between us and the SEC, and without admitting or denying any of the allegations of the complaint, we agreed to an injunction from violations of Section 13(d) of the Securities Exchange Act of 1934 (the “Exchange Act”), a strict liability claim, and to pay a $100,000 penalty, which has been paid. We also agreed to, within certain stipulated time periods: (i) establish a Management Investment Committee (“MIC”) that will make recommendations to an Independent Investment Committee (“IIC”) of our Board of Directors in connection with existing and future strategic minority investments; and (ii) retain an Independent Compliance Consultant (“ICC”) to (a) advise us on whether filings pursuant to Section 13(d) of the Exchange Act for previous strategic investments made at the suggestion of or in tandem with Dr. Frost should be amended or made to reflect group membership with Dr. Frost and his related entities; (b) review our existing policies and procedures relating to compliance with Section 13(d) of the Exchange Act; and (c) review the independence of the MIC and IIC of our Board of Directors solely for purposes of the handling of strategic minority investments. The ICC is required to report its findings (including recommendations as to filings, amendments, improvements to policies and procedures, and improvement to the composition of the MIC and the IIC to our Board of Directors) to the SEC within 15 days of completion of its work, and we are required to implement the ICC’s recommendations, and to certify our compliance with these undertakings in writing.
Under the terms of the settlement between the SEC and Dr. Frost, and without admitting or denying any of the allegations in the Complaint, Dr. Frost agreed to injunctions from violations of Sections 5(a) and (c) and 17(a)(2) of the Securities Act, claims which may be satisfied by strict liability and negligence, respectively, and Section 13(d) of the Exchange Act, also a strict liability claim; to pay approximately $5.5 million in penalty, disgorgement and pre-judgment interest, which has been paid; and to be prohibited, with certain exceptions, from trading in penny stocks. The settlements include no restriction on Dr. Frost’s ability to continue to serve as our CEO and Chairman.
146
ITEM 9. |
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
ITEM 9A. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and our Chief Financial Officer, have evaluated the effectiveness of the Company’s disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”)) as of December 31, 2018. Our disclosure controls and procedures are designed to provide reasonable assurance that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the rules and forms of the Securities and Exchange Commission. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company’s management, including its principal executive and principal financial officers, as appropriate to allow timely decisions regarding required disclosure. Based on this evaluation, management concluded that our disclosure controls and procedures were effective as of December 31, 2018.
Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended. Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. All internal control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined effective could provide only reasonable assurance with respect to financial statement preparation and presentation.
Our management conducted an evaluation of the effectiveness of our internal control over financial reporting as of December 31, 2018, based on the framework in the Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (the “2013 Internal Control-Integrated Framework”). Based on our evaluation under the 2013 Internal Control-Integrated Framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2018.
The effectiveness of the Company’s internal control over financial reporting as of December 31, 2018 has been audited by Ernst & Young LLP, our independent registered public accounting firm, who also audited our Consolidated Financial Statements included in this Annual Report on Form 10-K, as stated in their report which appears with our accompanying Consolidated Financial Statements.
Changes to the Company’s Internal Control Over Financial Reporting
We have implemented new controls as part of our effort to adopt Accounting Standards Update (“ASU”) No. 2014-09, Revenue from Contracts with Customers. The adoption of the ASU required the implementation of new accounting processes which necessitated changes to our internal controls over financial reporting.
These changes to the Company’s internal control over financial reporting that occurred since the beginning of 2018 have materially affected, or are reasonably likely to materially affect, the Company’s internal control over financial reporting.
147
ITEM 9B. OTHER INFORMATION
On February 26, 2019, BioReference and certain of its subsidiaries entered into Amendment No. 8 to the Credit Agreement, which amended the Credit Agreement to add back certain cost savings resulting from work force reductions in the 2018 fiscal year to the calculation of EBITDA for purposes of complying with the minimum fixed charge coverage ratio covenant. The other terms of the Credit Agreement remain unchanged.
148
PART III
The information required in Items 10 (Directors, Executive Officers and Corporate Governance), Item 11 (Executive Compensation), Item 12 (Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters), Item 13 (Certain Relationships and Related Transactions, and Director Independence), and Item 14 (Principal Accounting Fees and Services) is incorporated by reference to the Company’s definitive proxy statement for the 2019 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of December 31, 2018.
149
PART IV.
Item 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES.
(a) |
(1) |
Financial Statements: See Part II, Item 8 of this report. |
Schedule I - Condensed Financial Information of Registrant. Additionally, the financial statement schedule entitled “Schedule II – Valuation and Qualifying Accounts” has been omitted since the information required is included in the consolidated financial statements and notes thereto. Other schedules are omitted because they are not required. |
||
(2) |
Exhibits: See below. |
|
150
151
152
153
101.PRE |
XBRL Taxonomy Extension Presentation Linkbase Document |
* |
Denotes management contract or compensatory plan or arrangement. |
** |
Filed herewith. |
+ |
Certain confidential material contained in the document has been omitted and filed separately with the Securities and Exchange Commission. |
(1) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on April 2, 2007, and incorporated herein by reference. |
(2) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 31, 2008 and incorporated herein by reference. |
(3) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 8, 2008 for the Company’s three-month period ended June 30, 2008, and incorporated herein by reference. |
(4) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 12, 2008 for the Company’s three-month period ended September 30, 2008, and incorporated herein by reference. |
(5) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on May 8, 2009 for the Company’s three-month period ended March 31, 2009, and incorporated herein by reference. |
(6) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 7, 2009 for the Company’s three-month period ended June 30, 2009, and incorporated herein by reference. |
(7) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on September 24, 2009, and incorporated herein by reference. |
(8) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 9, 2009 for the Company’s three-month period ended September 30, 2009, and incorporated herein by reference. |
(9) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on May 10, 2010 for the Company’s three-month period ended March 31, 2010, and incorporated herein by reference. |
(10) |
Filed with the Company’s Amendment to Annual Report on Form 10-K filed with the Securities and Exchange Commission on February 3, 2011. |
(11) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 17, 2010. |
(12) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on March 10, 2011, and incorporated herein by reference. |
(13) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on May 10, 2011 for the Company’s three-month period ended March 31, 2011, and incorporated herein by reference. |
(14) |
Filed with the Company’s Quarterly Report on Form 10-Q/A filed with the Securities and Exchange Commission on July 5, 2011, and incorporated herein by reference. |
(15) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 9, 2011 for the Company’s three-month period ended September 30, 2011, and incorporated herein by reference. |
(16) |
Filed with the Company’s Annual Report on Form 10-K/A filed with the Securities and Exchange Commission on July 28, 2011. |
(17) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 15, 2012. |
(18) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on May 10, 2012 for the Company’s three-month period ended March 31, 2012, and incorporated herein by reference. |
(19) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 9, 2012 for the Company’s three-month period ended September 30, 2012, and incorporated herein by reference. |
(20) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on January 29, 2013, and incorporated herein by reference. |
(21) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 18, 2013. |
(22) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on May 10, 2013 for the Company’s three-month period ended March 31, 2013, and incorporated herein by reference. |
(23) |
Filed with the Company’s Schedule 13D filed with the Securities and Exchange Commission on March 22, 2013, and incorporated herein by reference. |
(24) |
Filed as Annex A to the Company’s Preliminary Joint Proxy Statement/Prospectus, Form S-4, with the Securities Exchange Commission on June 27, 2013, as amended, and incorporated herein by reference. |
154
(25) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on February 5, 2013, and incorporated herein by reference. |
(26) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 12, 2013 for the Company’s three month period ended September 30, 2013, and incorporated herein by reference. |
(27) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 5, 2015 for the Company’s three month period ended June 30, 2015, and incorporated herein by reference. |
(28) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on June 4, 2015, and incorporated herein by reference. |
(29) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on February 27, 2015, and incorporated herein by reference. |
(30) |
Filed under Part II, Item 8, of the Bio-Reference Laboratories, Inc. Form 10-K filed with the Securities and Exchange Commission on January 13, 2015 (File No. 0-15266), and incorporated herein by reference. |
(31) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on June 30, 2016 and incorporated herein by reference. |
(32) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on February 29, 2016, and incorporated herein by reference. |
(33) |
Filed with the Company’s Definitive Proxy Statement on Schedule 14A filed with the Securities and Exchange Commission on March 25, 2016, and incorporated herein by reference. |
(34) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 8, 2016 for the Company’s three month period ended June 30, 2016, and incorporated herein by reference. |
(35) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on March 23, 2017 and incorporated herein by reference. |
(36) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on November 8, 2017 and incorporated herein by reference. |
(37) |
Filed with the Company’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 1, 2018 and incorporated herein by reference. |
(38) |
Filed with the Company’s Quarterly Report on Form 10-Q filed with the Securities and Exchange Commission on August 7, 2018 for the Company’s three month period ended June 30, 2018, and incorporated herein by reference |
(39) |
Filed with the Company’s Current Report on Form 8-K filed with the Securities and Exchange Commission on February 7, 2019 and incorporated herein by reference.
|
155
Schedule I - Condensed Financial Information of Registrant
OPKO Health, Inc.
PARENT COMPANY CONDENSED BALANCE SHEETS
(In thousands, except share and per share data)
December 31, |
|||||||
2018 |
2017 |
||||||
ASSETS |
|||||||
Current assets: |
|||||||
Cash and cash equivalents |
$ |
14,724 |
$ |
21,385 |
|||
Other current assets and prepaid expenses |
2,545 |
4,586 |
|||||
Total current assets |
17,269 |
25,971 |
|||||
Property, plant and equipment, net |
59 |
150 |
|||||
Investments |
1,873,009 |
1,851,616 |
|||||
Other assets |
113 |
146 |
|||||
Total assets |
$ |
1,890,450 |
$ |
1,877,883 |
|||
LIABILITIES AND EQUITY |
|||||||
Current liabilities: |
|||||||
Accounts payable |
$ |
1,085 |
$ |
1,077 |
|||
Accrued expenses |
8,213 |
3,023 |
|||||
Current portion of 2033 Senior Notes |
31,562 |
— |
|||||
Current portion of notes payable |
521 |
521 |
|||||
Total current liabilities |
41,381 |
4,621 |
|||||
2033 Senior Notes, net of discount |
57,299 |
29,160 |
|||||
Deferred tax liabilities, net |
479 |
479 |
|||||
Total long-term liabilities |
57,778 |
29,639 |
|||||
Total liabilities |
99,159 |
34,260 |
|||||
Equity: |
|||||||
Common Stock - $0.01 par value, 750,000,000 shares authorized; 560,023,745 and 560,023,745 shares issued at December 31, 2018 and 2017, respectively |
5,869 |
5,600 |
|||||
Treasury Stock, at cost - 549,907 shares at December 31, 2018 and 2017, respectively |
(1,791 |
) |
(1,791 |
) |
|||
Additional paid-in capital |
3,004,422 |
2,889,256 |
|||||
Accumulated other comprehensive income (loss) |
(20,131 |
) |
(528 |
) |
|||
Accumulated deficit |
(1,197,078 |
) |
(1,048,914 |
) |
|||
Total shareholders’ equity |
1,791,291 |
1,843,623 |
|||||
Total liabilities and equity |
$ |
1,890,450 |
$ |
1,877,883 |
|||
The accompanying Notes to Parent Company Condensed Financial Statements are an integral part of these statements.
156
OPKO Health, Inc.
PARENT COMPANY CONDENSED STATEMENTS OF INCOME
(In thousands)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Revenues: |
|||||||||||
Revenue from products |
$ |
— |
$ |
— |
$ |
— |
|||||
Revenue from transfer of intellectual property and other |
1,069 |
1,069 |
— |
||||||||
Total revenues |
1,069 |
1,069 |
— |
||||||||
Costs and expenses: |
|||||||||||
Costs of revenue |
2,358 |
1,438 |
875 |
||||||||
Selling, general and administrative |
52,397 |
57,410 |
60,819 |
||||||||
Research and development |
4,184 |
4,426 |
3,791 |
||||||||
Total costs and expenses |
58,939 |
63,274 |
65,485 |
||||||||
Operating loss |
(57,870 |
) |
(62,205 |
) |
(65,485 |
) |
|||||
Other income and (expense), net: |
|||||||||||
Interest income |
631 |
260 |
440 |
||||||||
Interest expense |
(8,608 |
) |
(4,426 |
) |
(3,585 |
) |
|||||
Fair value changes of derivative instruments, net |
1,991 |
652 |
2,738 |
||||||||
Other income (expense), net |
3,906 |
5,177 |
(2,387 |
) |
|||||||
Other income and (expense), net |
(2,080 |
) |
1,663 |
(2,794 |
) |
||||||
Loss before income taxes and investment losses |
(59,950 |
) |
(60,542 |
) |
(68,279 |
) |
|||||
Income tax benefit (provision) |
(11 |
) |
(247 |
) |
(686 |
) |
|||||
Net loss before investment losses |
(59,961 |
) |
(60,789 |
) |
(68,965 |
) |
|||||
Loss from investments in investees |
(10,822 |
) |
(12,646 |
) |
(7,665 |
) |
|||||
Net income (loss) from subsidiaries, net of taxes |
(82,257 |
) |
(231,815 |
) |
28,271 |
||||||
Net loss attributable to common shareholders |
$ |
(153,040 |
) |
$ |
(305,250 |
) |
$ |
(48,359 |
) |
||
The accompanying Notes to Parent Company Condensed Financial Statements are an integral part of these statements.
157
OPKO Health, Inc.
PARENT COMPANY CONDENSED STATEMENTS OF COMPREHENSIVE INCOME
(In thousands)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Net loss |
$ |
(153,040 |
) |
$ |
(305,250 |
) |
$ |
(48,359 |
) |
||
Other comprehensive income (loss), net of tax: |
|||||||||||
Change in foreign currency translation and other comprehensive income (loss) |
(14,727 |
) |
22,724 |
(4,955 |
) |
||||||
Investments: |
|||||||||||
Change in unrealized gain (loss), net of tax |
— |
3,790 |
(3,810 |
) |
|||||||
Reclassification adjustments due to adoption of ASU 2016-01 |
(4,876 |
) |
— |
— |
|||||||
Reclassification adjustments for losses included in net loss, net of tax |
— |
(33 |
) |
4,293 |
|||||||
Comprehensive loss |
$ |
(172,643 |
) |
$ |
(278,769 |
) |
$ |
(52,831 |
) |
||
The accompanying Notes to Parent Company Condensed Financial Statements are an integral part of these statements.
158
OPKO Health, Inc.
PARENT COMPANY CONDENSED STATEMENTS OF CASH FLOWS
(In thousands)
For the years ended December 31, |
|||||||||||
2018 |
2017 |
2016 |
|||||||||
Cash flows from operating activities: |
|||||||||||
Net loss |
$ |
(153,040 |
) |
$ |
(305,250 |
) |
$ |
(48,359 |
) |
||
Adjustments to reconcile net loss to net cash used in operating activities: |
|||||||||||
Depreciation and amortization |
91 |
138 |
89 |
||||||||
Non-cash interest |
4,564 |
2,049 |
1,866 |
||||||||
Amortization of deferred financing costs |
551 |
574 |
149 |
||||||||
Losses from investments in investees |
10,822 |
12,646 |
7,665 |
||||||||
(Income) loss from subsidiaries |
82,257 |
231,815 |
(28,271 |
) |
|||||||
Equity-based compensation – employees and non-employees |
21,761 |
28,308 |
42,693 |
||||||||
Realized loss (gain) on equity securities and disposal of fixed assets |
208 |
(652 |
) |
(2,738 |
) |
||||||
Loss (gain) on conversion of 3.00% convertible senior notes |
— |
— |
284 |
||||||||
Change in fair value of derivative instruments |
(6,124 |
) |
(4,953 |
) |
2,347 |
||||||
Changes in other assets and liabilities |
6,847 |
4,258 |
(6,844 |
) |
|||||||
Net cash used in operating activities |
(32,063 |
) |
(31,067 |
) |
(31,119 |
) |
|||||
Cash flows from investing activities: |
|||||||||||
Investments in investees |
(1,000 |
) |
(9,625 |
) |
(14,424 |
) |
|||||
Subsidiary financing |
(123,787 |
) |
41,990 |
(44,568 |
) |
||||||
Proceeds from sale of equity securities |
1,516 |
2,211 |
— |
||||||||
Capital expenditures |
— |
— |
(368 |
) |
|||||||
Net cash provided by (used in) investing activities |
(123,271 |
) |
34,576 |
(59,360 |
) |
||||||
Cash flows from financing activities: |
|||||||||||
Proceeds from the exercise of Common Stock options and warrants |
1,173 |
2,132 |
8,576 |
||||||||
Issuance of common stock |
92,500 |
— |
— |
||||||||
Issuance convertible notes, net |
55,000 |
— |
— |
||||||||
Net cash provided by financing activities |
148,673 |
2,132 |
8,576 |
||||||||
Net increase (decrease) in cash and cash equivalents |
(6,661 |
) |
5,641 |
(81,903 |
) |
||||||
Cash and cash equivalents at beginning of period |
21,385 |
15,744 |
97,647 |
||||||||
Cash and cash equivalents at end of period |
$ |
14,724 |
$ |
21,385 |
$ |
15,744 |
|||||
SUPPLEMENTAL INFORMATION: |
|||||||||||
Interest paid |
$ |
956 |
$ |
956 |
$ |
966 |
|||||
Income taxes paid, net of refunds |
$ |
(578 |
) |
$ |
327 |
$ |
— |
||||
Non-cash financing: |
|||||||||||
Shares issued upon the conversion of: |
|||||||||||
2033 Senior Notes |
$ |
— |
$ |
— |
$ |
583 |
|||||
Common Stock options and warrants, surrendered in net exercise |
$ |
806 |
$ |
1,546 |
$ |
350 |
|||||
Issuance of capital stock to acquire or contingent consideration settlement: |
$ |
— |
|||||||||
Transition Therapeutics, Inc. |
$ |
— |
$ |
— |
$ |
58,530 |
|||||
OPKO Renal |
$ |
— |
$ |
— |
$ |
25,986 |
|||||
OPKO Health Europe |
$ |
— |
$ |
304 |
$ |
313 |
|||||
Issuance of stock for investment in Xenetic |
$ |
— |
$ |
— |
$ |
4,856 |
|||||
The accompanying Notes to Parent Company Condensed Financial Statements are an integral part of these statements.
159
OPKO Health, Inc.
Notes to Parent Company Condensed Financial Statements
Note 1. Organization and Basis of Presentation
We are a diversified healthcare company that seeks to establish industry-leading positions in large and rapidly growing medical markets. The parent company condensed financial statements included in this Schedule I represent the financial statements of OPKO Health, Inc., the parent company (or “OPKO”), on a stand-alone basis and do not include results of operations from our consolidated subsidiaries. The Parent Company Condensed Financial Statements should be read in conjunction with our audited consolidated financial statements included in Item 8 of Part II of this Form 10-K. As of December 31, 2018 and 2017, approximately $1.9 billion and $1.9 billion, respectively, of our Investments, net have not been eliminated in the parent company condensed financial statements.
The Parent Company Condensed Financial Statements included herein have been prepared in accordance with Rule 12-04, Schedule I of Regulation S-X, as substantially all the assets of BioReference, a wholly-owned subsidiary, and its subsidiaries are restricted from sale, transfer, lease, disposal or distributions to OPKO under the credit agreement with JPMorgan Chase Bank, N.A. (the “Credit Agreement”), subject to certain exceptions. BioReference and its subsidiaries’ net assets as of December 31, 2018 were approximately $0.9 billion, which includes goodwill of $401.8 million and intangible assets of $405.3 million. BioReference’s restricted net assets exceeds 25% of OPKO’s consolidated net assets of $2.5 billion as of December 31, 2018.
Note 2 Debt
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. Borrowings under the line of credit will bear interest at a rate of 10% per annum and may be repaid and reborrowed at any time. The credit agreement includes various customary remedies for the lender following an event of default, including the acceleration of repayment of outstanding amounts under line of credit. The line of credit matures on November 8, 2023. As of as of December 31, 2018, no funds were borrowed under the line of credit, and in February 2019, we repaid amounts borrowed in 2019 and terminated the credit agreement.
In February 2018, we issued a series of 5% Convertible Promissory Notes (the “2023 Convertible Notes”) in the aggregate principal amount of $55.0 million. The 2023 Convertible Notes mature 5 years from the date of issuance. Each holder of a 2023 Convertible Note has the option, from time to time, to convert all or any portion of the outstanding principal balance of such 2023 Convertible Note, together with accrued and unpaid interest thereon, into shares of our Common Stock, par value $0.01 per share, at a conversion price of $5.00 per share of Common Stock (the “Shares”). We may redeem all or any part of the then issued and outstanding 2023 Convertible Notes, together with accrued and unpaid interest thereon, pro ratably among the holders, upon no fewer than 30 days, and no more than 60 days, notice to the holders. The 2023 Convertible Notes contain customary events of default and representations and warranties of OPKO.
The issuance of the 2023 Convertible Notes and the issuance of the Shares, if any, upon conversion thereof was not, and will not be, respectively, registered under the Securities Act, pursuant to the exemption provided by Section 4(a)(2) thereof, and we have not agreed to register the Shares if or when such Shares are issued. Purchasers of the 2023 Convertible Notes include an affiliate of Dr. Phillip Frost, M.D., our Chairman and Chief Executive Officer, and Dr. Jane H. Hsiao, Ph.D., MBA, our Vice-Chairman and Chief Technical Officer.
In January 2013, we entered into note purchase agreements (the “2033 Senior Notes”) with qualified institutional buyers and accredited investors (collectively, the “Purchasers”) in a private placement in reliance on exemptions from registration under the Securities Act. The 2033 Senior Notes were issued on January 30, 2013. The 2033 Senior Notes, which totaled $175.0 million in original principal amount, bear interest at the rate of 3.0% per year, payable semiannually on February 1 and August 1 of each year. The 2033 Senior Notes will mature on February 1, 2033, unless earlier repurchased, redeemed or converted. Upon a fundamental change as defined in the Indenture, dated as of January 30, 2013, by and between the Company and Wells Fargo Bank N.A., as trustee, governing the 2033 Senior Notes (the “Indenture”), subject to certain exceptions, the holders may require us to repurchase all or any portion of their 2033 Senior Notes for cash at a repurchase price equal to 100% of the principal amount of the 2033 Senior Notes being repurchased, plus any accrued and unpaid interest to but not including the related fundamental change repurchase date.
160
The following table sets forth information related to the 2033 Senior Notes which is included in our Consolidated Balance Sheet as of December 31, 2018:
(In thousands) |
2033 Senior Notes |
Discount |
Debt Issuance Cost |
Total |
|||||||||||
Balance at December 31, 2017 |
$ |
31,850 |
$ |
(2,565 |
) |
$ |
(125 |
) |
$ |
29,160 |
|||||
Amortization of debt discount and debt issuance costs |
— |
2,277 |
125 |
2,402 |
|||||||||||
Balance at December 31, 2018 |
$ |
31,850 |
$ |
(288 |
) |
$ |
— |
$ |
31,562 |
||||||
The following table sets forth information related to the 2033 Senior Notes which is included in our Consolidated Balance Sheet as of December 31, 2017:
(In thousands) |
Embedded conversion option |
2033 Senior Notes |
Discount |
Debt Issuance Cost |
Total |
||||||||||||||
Balance at December 31, 2016 |
$ |
16,736 |
$ |
31,850 |
$ |
(4,612 |
) |
$ |
(273 |
) |
$ |
43,701 |
|||||||
Amortization of debt discount and debt issuance costs |
— |
— |
2,047 |
148 |
2,195 |
||||||||||||||
Change in fair value of embedded derivative |
(3,185 |
) |
— |
— |
— |
(3,185 |
) |
||||||||||||
Reclassification of embedded derivatives to equity |
(13,551 |
) |
— |
— |
— |
(13,551 |
) |
||||||||||||
Balance at December 31, 2017 |
$ |
— |
$ |
31,850 |
$ |
(2,565 |
) |
$ |
(125 |
) |
$ |
29,160 |
|||||||
The 2033 Senior Notes will be convertible at any time on or after November 1, 2032, through the second scheduled trading day immediately preceding the maturity date, at the option of the holders. Additionally, holders may convert their 2033 Senior Notes prior to the close of business on the scheduled trading day immediately preceding November 1, 2032, under the following circumstances: (1) conversion based upon satisfaction of the trading price condition relating to the 2033 Senior Notes; (2) conversion based on the Common Stock price; (3) conversion based upon the occurrence of specified corporate events; or (4) if we call the 2033 Senior Notes for redemption. The 2033 Senior Notes will be convertible into cash, shares of our Common Stock, or a combination of cash and shares of Common Stock, at our election unless we have made an irrevocable election of net share settlement. The initial conversion rate for the 2033 Senior Notes will be 141.48 shares of Common Stock per $1,000 principal amount of 2033 Senior Notes (equivalent to an initial conversion price of approximately $7.07 per share of Common Stock), and will be subject to adjustment upon the occurrence of certain events. In addition, we will, in certain circumstances, increase the conversion rate for holders who convert their 2033 Senior Notes in connection with a make-whole fundamental change (as defined in the Indenture). Holders of the 2033 Senior Notes may require us to repurchase the 2033 Senior Notes for 100% of their principal amount, plus accrued and unpaid interest, on February 1, 2019, February 1, 2023 and February 1, 2028, or following the occurrence of a fundamental change as defined in the indenture governing the 2033 Senior Notes. On February 1, 2019, approximately $28.8 million aggregate principal amount of 2033 Senior Notes were tendered by holders pursuant to such holders’ option to require us to repurchase the 2033 Senior Notes.
Before February 1, 2019, we may redeem for cash any or all of the 2033 Senior Notes but only if the last reported sale price of our Common Stock exceeds 130% of the applicable conversion price for at least 20 trading days during the 30 consecutive trading day period ending on the trading day immediately prior to the date on which we deliver the redemption notice. The redemption price will equal 100% of the principal amount of the 2033 Senior Notes to be redeemed, plus any accrued and unpaid interest to but not including the redemption date. On or after February 1, 2019, we may redeem for cash any or all of the 2033 Senior Notes at a redemption price of 100% of the principal amount of the 2033 Senior Notes to be redeemed, plus any accrued and unpaid interest up to but not including the redemption date.
The terms of the 2033 Senior Notes, include, among others: (i) rights to convert into shares of our Common Stock, including upon a fundamental change; and (ii) a coupon make-whole payment in the event of a conversion by the holders of the 2033 Senior Notes on or after February 1, 2017 but prior to February 1, 2019. We determined that these specific terms were considered to be embedded derivatives. Embedded derivatives are required to be separated from the host contract, the 2033 Senior Notes, and carried at fair value when: (a) the embedded derivative possesses economic characteristics that are not clearly and closely related to the economic characteristics of the host contract; and (b) a separate, stand-alone instrument with the same terms would qualify as a derivative instrument. We concluded that the embedded derivatives within the 2033 Senior Notes meet these criteria for periods prior to February 1, 2017 and, as such, were valued separate and apart from the 2033 Senior Notes and recorded at fair value each reporting period.
161
For accounting and financial reporting purposes, we combined these embedded derivatives and valued them together as one unit of accounting. In 2017, certain terms of the embedded derivatives expired pursuant to the original agreement and the embedded derivatives no longer met the criteria to be separated from the host contract and, as a result, the embedded derivatives were no longer required to be valued separate and apart from the 2033 Senior Notes and were reclassified to additional paid in capital. Accordingly, there was no derivative income (loss) for the year ended December 31, 2018.
From 2013 to 2016, holders of the 2033 Senior Notes converted 143.2 million in aggregate principal amount into an aggregate of 21,539,873 shares of the Company’s Common Stock.
In November 2015, BioReference and certain of its subsidiaries entered into the Credit Agreement with JPMorgan Chase Bank, which provides for a $175.0 million secured revolving credit facility and includes a $20.0 million sub-facility for swingline loans and a $20.0 million sub-facility for the issuance of letters of credit. The Credit Agreement matures on November 5, 2020 and is secured by substantially all assets of BioReference and its domestic subsidiaries, as well as a non-recourse pledge by us of our equity interest in BioReference.
Note 3 Commitments and Contingencies
We have no significant direct commitments and contingencies, but our subsidiaries do. See Note 13 of our Consolidated Financial Statements in Item 8 of Part II of this Form 10-K.
Note 4 Dividends
We did not receive any dividend payments from our consolidated subsidiaries for the years ended December 31, 2018, 2017 and 2016.
Note 5 Income Taxes
The Parent Company Condensed Financial Statements recognize the current and deferred income tax consequences that result from our activities during the current and preceding periods pursuant to the provisions of Accounting Standards Codification Topic 740, Income Taxes (ASC 740), as if we were a separate taxpayer rather than a member of the consolidated income tax return group. The tax expense and benefit recorded in OPKO’s consolidated financial statements was the result of activity at the subsidiaries and therefore all tax benefit and expense was reported in the Net income (loss) from subsidiaries, net of taxes line in the Condensed Statement of Income.
Note 6 Subsequent Events
In February 2019, we issued $200.0 million aggregate principal amount of Convertible Senior Notes due 2025 (the “2025 Notes”) in an underwritten public offering. The 2025 Notes will bear interest at a rate of 4.50% per year, payable semiannually in arrears on February 15 and August 15 of each year, beginning on August 15, 2019. The notes mature on February 15, 2025, unless earlier repurchased, redeemed or converted.
Holders may convert their 2025 Notes at their option at any time prior to the close of business on the business day immediately preceding November 15, 2024 only under the following circumstances: (1) during any calendar quarter commencing after the calendar quarter ending on March 31, 2019 (and only during such calendar quarter), if the last reported sale price of our common stock for at least 20 trading days (whether or not consecutive) during a period of 30 consecutive trading days ending on the last trading day of the immediately preceding calendar quarter is greater than or equal to 130% of the conversion price on each applicable trading day; (2) during the five business day period after any five consecutive trading day period (the “measurement period”) in which the trading price per $1,000 principal amount of 2025 Notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price of our common stock and the conversion rate on each such trading day; (3) if we call any or all of the 2025 Notes for redemption, at any time prior to the close of business on the scheduled trading day immediately preceding the redemption date; or (4) upon the occurrence of specified corporate events. On or after November 15, 2024, until the close of business on the business day immediately preceding the maturity date, holders of the 2025 Notes may convert their notes at any time, regardless of the foregoing circumstances. Upon conversion, we will pay or deliver, as the case may be, cash, shares of our common stock, or a combination of cash and shares of our common stock, at our election.
The conversion rate for the notes will initially be 236.7424 shares of common stock per $1,000 principal amount of 2025 Notes (equivalent to an initial conversion price of approximately $4.22 per share of common stock). The conversion rate for the 2025 Notes will be subject to adjustment in some events but will not be adjusted for any accrued and unpaid interest. In addition, following certain corporate events that occur prior to the maturity date of the notes or if we deliver a notice of
162
redemption, in certain circumstances we will increase the conversion rate of the 2025 Notes for a holder who elects to convert its notes in connection with such a corporate event or notice of redemption as the case may be.
We may not redeem the 2025 Notes prior to February 15, 2022. We may redeem for cash any or all of the notes, at our option, on or after February 15, 2022, if the last reported sale price of our common stock has been at least 130% of the conversion price for the notes then in effect for at least 20 trading days (whether or not consecutive) during any 30 consecutive trading day period (including the last trading day of such period) ending on, and including, the trading day immediately preceding the date on which we provide notice of redemption at a redemption price equal to 100% of the principal amount of the notes to be redeemed, plus accrued and unpaid interest to, but excluding, the redemption date. No sinking fund is provided for the 2025 Notes.
If we undergo a fundamental change prior to the maturity date of the 2025 Notes, holders may require us to repurchase for cash all or any portion of their notes at a repurchase price equal to 100% of the principal amount of the notes to be repurchased, plus accrued and unpaid interest to, but excluding, the fundamental change repurchase date. The 2025 Notes will be our senior unsecured obligations and will rank senior in right of payment to any of our indebtedness that is expressly subordinated in right of payment to the 2025 Notes; equal in right of payment to any of our existing and future liabilities that are not so subordinated; effectively junior in right of payment to any of our secured indebtedness to the extent of the value of the assets securing such indebtedness; and structurally junior to all indebtedness and other liabilities (including trade payables) of our current or future subsidiaries.
On November 8, 2018, we entered into a credit agreement with an affiliate of Dr. Frost, pursuant to which the lender committed to provide us with an unsecured line of credit in the amount of $60 million. The credit agreement was terminated on or around February 20, 2019 and amounts borrowed during 2019 were repaid from the proceeds of the 2025 Convertible Notes offering.
On February 1, 2019, approximately $28.8 million aggregate principal amount of 2033 Senior Notes were tendered by holders pursuant to such holders’ option to require us to repurchase the 2033 Senior Notes. Holders of the 2033 Senior Notes issued in January 2013 may require us to repurchase the 2033 Senior Notes for 100% of their principal amount, plus accrued and unpaid interest, again on February 1, 2023 and February 1, 2028, or following the occurrence of a fundamental change as defined in the indenture governing the 2033 Senior Notes.
On February 26, 2019, BioReference and certain of its subsidiaries entered into Amendment No. 8 to the Credit Agreement, which amended the Credit Agreement to add back certain cost savings resulting from work force reductions in the 2018 fiscal year to the calculation of EBITDA for purposes of complying with the minimum fixed charge coverage ratio covenant. The other terms of the Credit Agreement remain unchanged.
In December 2018, we and Dr. Frost entered into settlements with the SEC, which, upon approval by the court in January 2019, resolved the claims against us and Dr. Frost raised in the SEC complaint. Pursuant to the settlement between us and the SEC, and without admitting or denying any of the allegations of the complaint, we agreed to an injunction from violations of Section 13(d) of the Securities Exchange Act of 1934 (the “Exchange Act”), a strict liability claim, and to pay a $100,000 penalty, which has been paid. We also agreed to, within certain stipulated time periods: (i) establish a Management Investment Committee (“MIC”) that will make recommendations to an Independent Investment Committee (“IIC”) of our Board of Directors in connection with existing and future strategic minority investments; and (ii) retain an Independent Compliance Consultant (“ICC”) to (a) advise us on whether filings pursuant to Section 13(d) of the Exchange Act for previous strategic investments made at the suggestion of or in tandem with Dr. Frost should be amended or made to reflect group membership with Dr. Frost and his related entities; (b) review our existing policies and procedures relating to compliance with Section 13(d) of the Exchange Act; and (c) review the independence of the MIC and IIC of our Board of Directors solely for purposes of the handling of strategic minority investments. The ICC is required to report its findings (including recommendations as to filings, amendments, improvements to policies and procedures, and improvement to the composition of the MIC and the IIC to our Board of Directors) to the SEC within 15 days of completion of its work, and we are required to implement the ICC’s recommendations, and to certify our compliance with these undertakings in writing.
Under the terms of the settlement between the SEC and Dr. Frost, and without admitting or denying any of the allegations in the Complaint, Dr. Frost agreed to injunctions from violations of Sections 5(a) and (c) and 17(a)(2) of the Securities Act, claims which may be satisfied by strict liability and negligence, respectively, and Section 13(d) of the Exchange Act, also a strict liability claim; to pay approximately $5.5 million in penalty, disgorgement and pre-judgment interest, which has been paid; and to be prohibited, with certain exceptions, from trading in penny stocks. The settlements include no restriction on Dr. Frost’s ability to continue to serve as our CEO and Chairman.
We have reviewed all subsequent events and transactions that occurred after the date of our December 31, 2018 Consolidated Balance Sheet date, through the time of filing this Annual Report on Form 10-K.
163
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Date: March 1, 2019 |
OPKO HEALTH, INC. |
|
By: |
/s/ Phillip Frost, M.D. |
|
Phillip Frost, M.D. |
||
Chairman of the Board and |
||
Chief Executive Officer |
||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
Signature |
Title |
Date |
||
/s/ Phillip Frost, M.D. |
Chairman of the Board and Chief Executive |
March 1, 2019 |
||
Phillip Frost, M.D. |
Officer (Principal Executive Officer) |
|||
/s/ Jane H. Hsiao, Ph.D., MBA |
Vice Chairman and Chief Technical Officer |
March 1, 2019 |
||
Jane H. Hsiao, Ph.D., MBA |
||||
/s/ Steven D. Rubin |
Director and Executive Vice President – |
March 1, 2019 |
||
Steven D. Rubin |
Administration |
|||
/s/ Adam Logal |
Senior Vice President, Chief Financial Officer, |
March 1, 2019 |
||
Adam Logal |
Chief Accounting Officer and Treasurer
(Principal Financial Officer)
|
|||
/s/ Richard Krasno, Ph.D. |
Director |
March 1, 2019 |
||
Richard Krasno, Ph.D. |
||||
/s/ Richard A. Lerner, M.D. |
Director |
March 1, 2019 |
||
Richard A. Lerner, M.D. |
||||
/s/ John A. Paganelli |
Director |
March 1, 2019 |
||
John A. Paganelli |
||||
/s/ Richard C. Pfenniger, Jr. |
Director |
March 1, 2019 |
||
Richard C. Pfenniger, Jr. |
||||
/s/ Alice Lin-Tsing Yu, M.D., Ph.D. |
Director |
March 1, 2019 |
||
Alice Lin-Tsing Yu, M.D., Ph.D. |
||||
/s/ Robert S. Fishel, M.D. |
Director |
March 1, 2019 |
||
Robert S. Fishel, M.D. |
||||
164
Exhibit Number |
Description |
Certification by Phillip Frost, Chief Executive Officer, pursuant to Rule 13a-14(a) and 15d-14(a) of the Securities and Exchange Act of 1934 as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 for the year ended December 31, 2018. |
|
Certification by Adam Logal, Chief Financial Officer, pursuant to Rule 13a-14(a) and 15d-14(a) of the Securities and Exchange Act of 1934 as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 for the year ended December 31, 2018. |
|
Certification by Phillip Frost, Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 for the year ended December 31, 2018. |
|
Certification by Adam Logal, Chief Financial Officer, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 for the year ended December 31, 2018. |
|
101.INS |
XBRL Instance Document |
101.SCH |
XBRL Taxonomy Extension Schema Document |
101.CAL |
XBRL Taxonomy Extension Calculation Linkbase Document |
101.DEF |
XBRL Taxonomy Extension Definition Linkbase Document |
101.LAB |
XBRL Taxonomy Extension Label Linkbase Document |
101.PRE |
XBRL Taxonomy Extension Presentation Linkbase Document |
165